More than 10,000 Americans are waiting for a liver transplant. Many more never make the list, because they are too sick to handle a major surgery. That gap is why an idea that sounds a little strange at first keeps coming up in liver research: What if you could add liver function without replacing the liver?
MIT engineers are pushing that concept with what they call “satellite livers,” small pockets of liver tissue that can be injected and left to do some of the liver’s work while the damaged organ stays in place.
“We think of these as satellite livers. If we could deliver these cells into the body, while leaving the sick organ in place, that would provide booster function,” says Sangeeta Bhatia, the John and Dorothy Wilson Professor of Health Sciences and Technology and of Electrical Engineering and Computer Science at MIT. She is also a member of MIT’s Koch Institute for Integrative Cancer Research and the Institute for Medical Engineering and Science (IMES).
In a mouse study, Bhatia’s team showed that injected liver cells could stay viable for at least two months and keep producing liver proteins during that time. The work appears today in the journal Cell Biomaterials. MIT postdoc Vardhman Kumar is the lead author.
Your liver does a lot of things quietly, until it cannot. The study notes that the human liver plays a role in about 500 essential functions. It helps regulate blood clotting, removes bacteria from the bloodstream, and metabolizes drugs. Most of this work is carried out by hepatocytes, the main functional cells of the liver.
For more than a decade, Bhatia’s lab has worked on ways to restore hepatocyte function without doing a full transplant. One route is to put hepatocytes into a biomaterial, such as a hydrogel, and implant that material. But implantation still means surgery.
So the group took aim at a less invasive idea: injection. The problem is that liver cells injected alone typically do not settle into a stable home inside the body. In the new work, the MIT team tried to give the cells something like a ready-made neighborhood, one that could be delivered through a syringe and help the transplanted cells plug into the body’s blood supply.
The researchers injected hepatocytes together with hydrogel microspheres, tiny building blocks that can pack together tightly. When they are jammed together, the microspheres behave in a useful way: they can act like a liquid when pushed through a needle, then regain a solid structure once the stress is removed.
Kumar describes what the microspheres do for the cells.
“What we did is use this technology to create an engineered niche for cell transplantation,” he says. “If the cells are injected in the absence of these spheres, they would not integrate efficiently with the host, but these microspheres provide the hepatocytes with a niche where they can stay localized and become connected to the host circulation much faster.”
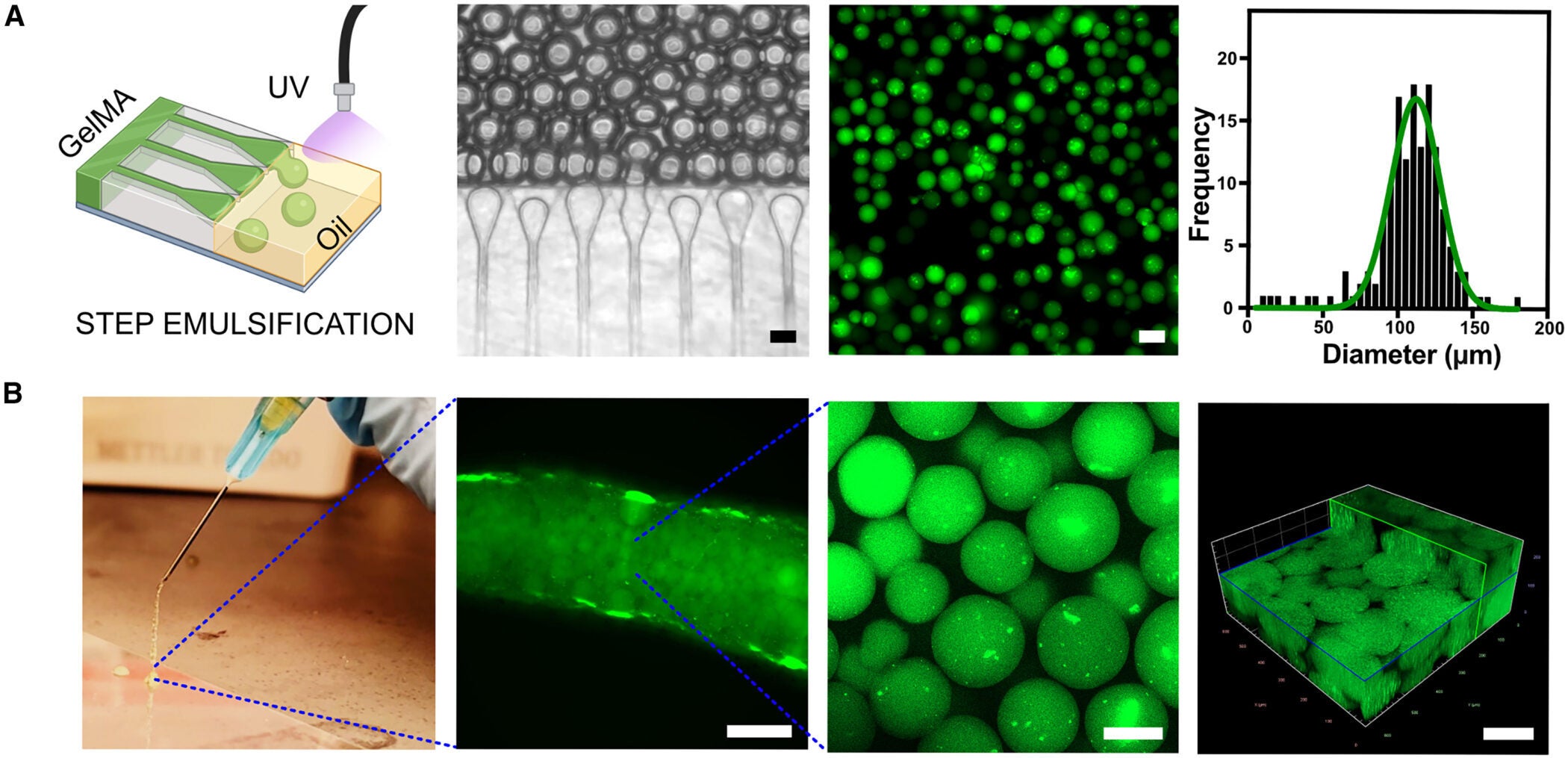
The team used gelatin methacryloyl, or GelMA, to make the microspheres. They created droplets using a microfluidic device and then crosslinked the droplets with UV exposure, producing microspheres with a mean diameter of about 110 micrometers. When the packed microspheres were pushed through a 25-gauge needle, they still preserved void spaces between them, a feature that matters because those spaces can allow cells and blood vessels to move in.
The group compared these packed microspheres to bulk, monolithic GelMA gels in mice. One week after implantation in the fat pad near the reproductive organs, bulk gels stayed mostly avascular, with a clear boundary between gel and host tissue.
The packed microspheres looked different. Blood vessels infiltrated the spaces between spheres, and histology showed higher cellularity. In other words, the microsphere scaffold behaved less like a sealed plug and more like something the body could grow into.
That early vascular integration matters, because liver cells need fast access to nutrients and oxygen.
The research also leans on a practical clinical tool: ultrasound.
Working with Nicole Henning, an ultrasound research specialist at the Koch Institute, the team developed an ultrasound-guided injection approach. Ultrasound helped them place the material where they wanted it, and it also gave them a way to track whether the graft stayed put over time.
The microsphere bolus showed up as a moderately echogenic structure on ultrasound, distinguishable from surrounding fat. By contrast, saline created a dark bolus that quickly dispersed. The microsphere scaffold remained detectable for weeks, which the researchers attribute to acoustic scattering from the many interfaces in the microsphere structure. They also used contrast-enhanced ultrasound with injected microbubbles, detecting perfusion signals in the graft region consistent with vascular connectivity.
The study’s main implant site was fat tissue in the belly, specifically perigonadal adipose tissue. The researchers write that future grafts might be delivered to other locations such as the spleen or near the kidneys. Kumar adds that for many liver disorders, the graft does not need to sit close to the liver.
“For a vast majority of liver disorders, the graft does not need to sit close to the liver,” he says.
A second ingredient turned out to matter: supportive cells.
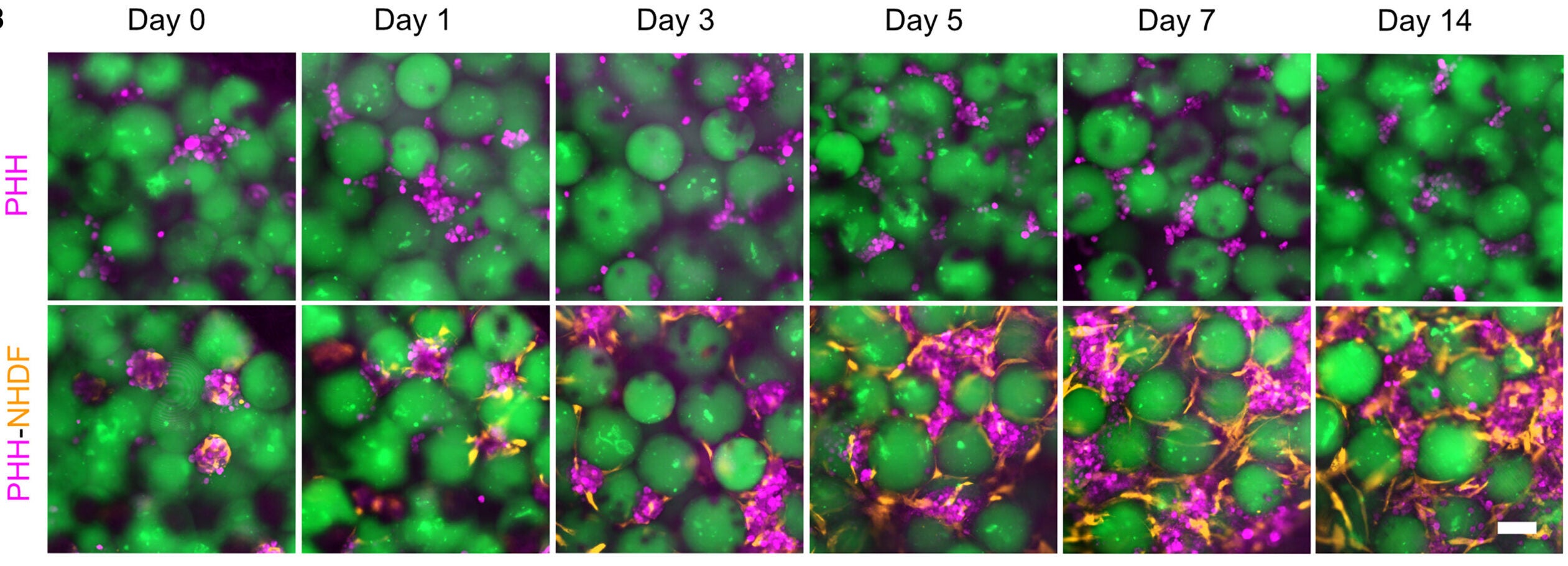
The researchers combined primary human hepatocytes with neonatal human dermal fibroblasts, creating aggregates for three days before mixing them into the microsphere slurry. The paper notes that earlier work has shown that close-contact signals from stromal cells like fibroblasts help stabilize hepatocyte phenotype and function.
In lab culture, hepatocyte-only aggregates did not hold together well. They fragmented, forming smaller clusters and single cells. In co-cultures, fibroblasts migrated outward and spread across the microsphere surfaces, forming a network that bridged aggregates and spheres.
After 14 days in vitro, hepatocytes expressed arginase-1, a mature hepatocyte marker. Albumin secretion in the media was significantly higher when fibroblasts were present. Urea production was similar in both conditions at day 14, reported as 49.34 ± 25.53 versus 46.57 ± 11.57 micrograms per milliliter per day per million cells, with five constructs in each condition. The team also measured drug-metabolizing capacity using CYP3A4 activity after rifampicin induction. CYP3A4 activity was detectable in both groups, but higher with fibroblasts.
For the in vivo test, the researchers compared three injections in NSG mice: hepatocyte aggregates alone, hepatocyte-fibroblast aggregates, and hepatocyte-fibroblast aggregates delivered with GelMA microspheres.
The difference showed up quickly on ultrasound. Aggregates in media dispersed through the fat tissue and were not detectable after the injection. Microsphere-containing grafts stayed as a localized structure and remained visible for the full eight-week study period, allowing the team to track graft volume.
At the endpoint, histology matched the imaging. In the groups without microspheres, only a few scattered clusters of hepatocytes could be found. In the microsphere group, the grafts formed compact, tissue-like niches that localized the hepatocytes. The team reports that circulating human albumin levels were consistently higher in the microsphere group than in the others, and at week eight they remained significantly elevated.
Staining showed arginase-1 expression in the grafts. Hepatocytes also showed localization of the bile salt export pump, described as consistent with mature lineage and polarity, in the microsphere group and in the hepatocyte-fibroblast group, but not in the hepatocyte-only group.
The researchers also looked for cell death using TUNEL staining. They report minimal cell death, under 1.2% TUNEL-positive within the graft, and no overlap between the TUNEL signal and hepatocytes.
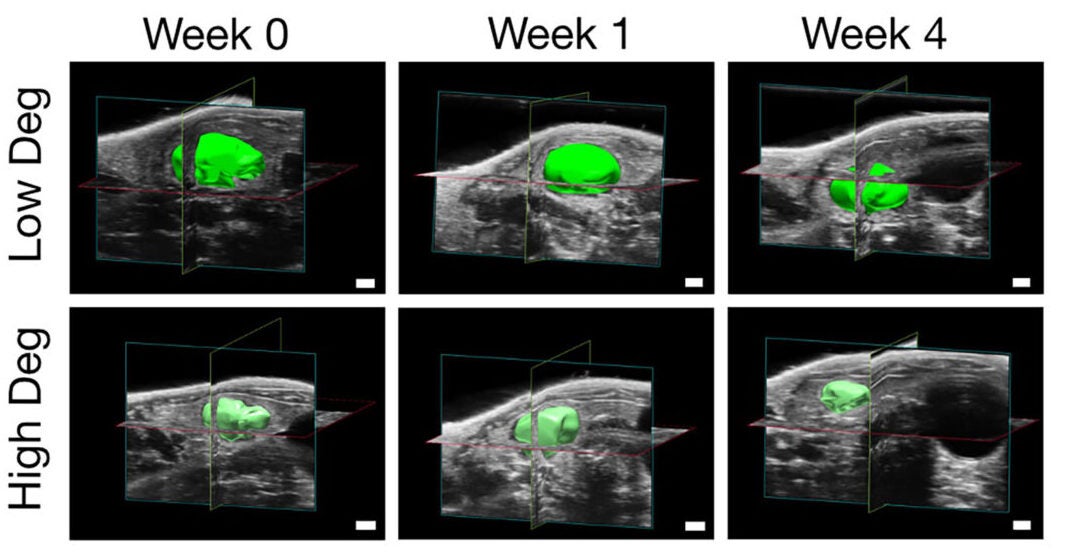
The team also experimented with how long the microspheres stick around.
They made high-degradability and low-degradability microspheres by changing the degree of crosslinking. In mice, high-degradability gels lost detectable volume by four weeks, while low-degradability gels only slightly decreased over the same period.
The tissue response looked different too. In high-degradability grafts, vessels had larger average luminal cross-sectional area and extended beyond the original void spaces into the remodeled niche. In low-degradability grafts, vessels stayed confined to the voids between microspheres.
Macrophage staining suggested different spatial patterns and trends in markers linked to pro-inflammatory and pro-regenerative phenotypes, though the study emphasizes caution in interpreting immune patterns in immunodeficient mouse models.
Function shifted along with remodeling. At week four, serum albumin secretion was higher in the high-degradability group, suggesting improved graft function.
The paper flags several limitations and next steps.
One limitation is imaging. Ultrasound can track the microsphere scaffold because of its natural contrast, but it cannot distinguish microspheres from hepatocyte aggregates inside the graft. The authors suggest future versions could incorporate defined contrast agents in the microspheres and separately labeled cells to track each component.
Another issue is scale. The team says future work will explore the maximum niche size this approach can support, since larger grafts raise questions about diffusion, vascularization, and stability. Cell supply is also a challenge. The study notes that stem-cell-derived hepatocyte-like cells often fail to reach full maturity, and it points to advances in expanding primary human hepatocytes in different formats as a possible route to larger grafts.
The authors also underline a major caution: the immune environment. Their mouse model does not replicate the complexity of immune responses in an intact system, so questions about macrophage polarization, scaffold remodeling, and foreign body responses need testing in immunocompetent or humanized immune models.
If this strategy translates beyond mice, it points to a different kind of liver support, one that could be delivered with a needle rather than an operating room. Ultrasound-guided injection is already a routine tool in many clinical settings, and the work suggests that engineered cell niches could be placed, monitored, and potentially adjusted over time without repeated open surgeries.
The study also frames these grafts as flexible. They could act as an alternative to surgery for some patients, or as temporary support while someone waits for a donor organ. The approach is not limited to the liver, either. The authors highlight pancreatic islet transplantation as another area where an image-guided, localized niche might address problems seen with cell dispersion and poor engraftment.
For patients facing liver failure, the long-term promise is not a full replacement organ in a lab, at least not yet. It is a smaller, injectable add-on that buys time and restores some of what the failing organ can no longer do.
Research findings are available online in the journal Cell Biomaterials.
The original story “MIT engineers built injectable ‘satellite livers’ as an alternative to liver transplants” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post MIT engineers built injectable ‘satellite livers’ as an alternative to liver transplants appeared first on The Brighter Side of News.