One participant pointed to her chest. That, she explained, is where she felt her foot hit the treadmill. Not the foot itself, not the ground beneath it, but a sensation somewhere above the injury that her brain had learned to translate into something useful. “It wasn’t like I could feel my foot hit the treadmill or anything like that,” she said, “but it was close.”
Close is a word that carries real weight in spinal cord injury research. For people who have lost all sensation and movement below the waist, close to normal is not a consolation. It is a clinical milestone.
A team from Brown University, Rhode Island Hospital, and VA Providence Healthcare has now reported results from a small but significant clinical trial, the first to demonstrate simultaneous motor control and sensory feedback in people with complete spinal cord injuries. The findings were published in Nature Biomedical Engineering.

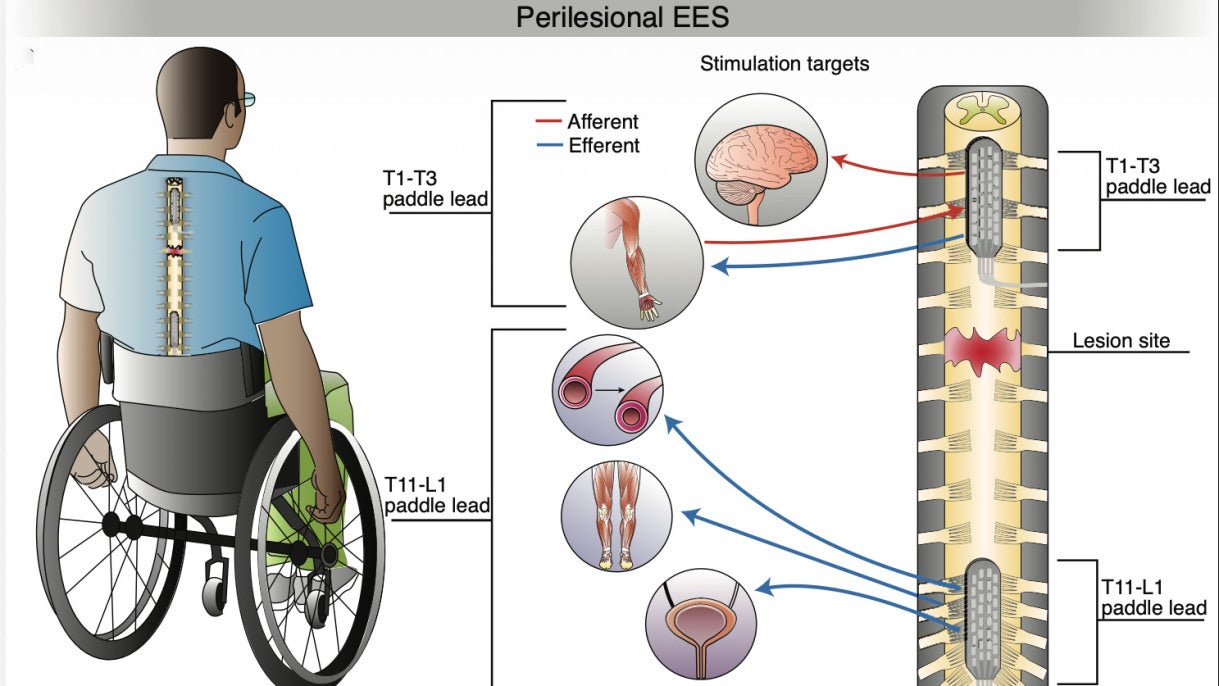
Three volunteers participated. Each had lost the use of their legs following complete thoracic spinal cord injuries. Surgeons implanted two small electrode arrays along each participant’s spine, one positioned above the injury site and one below it. Both arrays delivered patterned electrical pulses designed to mimic the signals that normally travel through an intact spinal cord.
The study ran for 12 to 14 days per participant, conducted entirely in a hospital setting. At the end, the electrode arrays were removed.
Prior research had established that stimulation below a spinal injury can activate the nerve networks responsible for leg movement. What had never been attempted was combining that motor stimulation with a simultaneous strategy for restoring sensory awareness of limb position, targeting both sides of the injury at once.
“This is the first time that simultaneous motor stimulation and sensory feedback have been demonstrated in people with complete spinal cord injuries,” said David Borton, an associate professor of engineering at Brown and a biomedical engineer at the VA Center for Neurorestoration and Neurotechnology. “By providing both motor activation and simultaneous sensory feedback, we are making progress toward restoring coordinated movements and functional independence.”
Before any walking experiments, the team needed to figure out which electrical patterns would actually produce useful leg movements in each person. The spinal cord is not a simple system. The parameters that activate one person’s muscles may do nothing in another. Finding the right settings across a 32-channel electrode array, with adjustable amplitude, frequency, and location, involves an enormous number of possible combinations.
To navigate that complexity, the researchers built what they called the DJ board: a physical control panel of knobs and sliders that let participants directly adjust their own stimulation in real time while watching their legs respond. Participants were given target leg positions and poses and dialed the settings until their legs reached them.
“Participants told us that using the DJ board was actually a lot of fun,” said study lead author Jonathan Calvert, an assistant professor of neurological surgery at UC Davis who worked on the project as a postdoctoral researcher at Brown. “They really enjoyed being able to see their legs move again and having their own control through the interface.”
Data from those sessions trained machine learning models developed by Thomas Serre, a professor of cognitive and linguistic sciences at Brown. The algorithms found optimal matches between target muscle patterns and stimulation parameters far more efficiently than trial and error alone could manage.
“The space of possible stimulations is huge, far too large to be efficiently searched by trial and error,” said co-author Lakshmi Narasimhan Govindarajan, a recent Ph.D. graduate from Serre’s lab now at MIT. “Machine learning provides an opportunity to more efficiently search and personalize stimulation patterns.”
The sensory challenge required a different strategy altogether. Below a complete spinal injury, the nerve pathways that normally carry sensation from the legs to the brain are severed. There is no way to stimulate those pathways directly. The researchers instead targeted the spinal cord above the injury, where connections to the brain remain intact, but where those connections map to the chest, shoulders, and arms rather than the legs.
The approach they used is called sensory substitution. Instead of restoring the original sensation, it creates a new one that the brain can learn to interpret. Stimulation intensity above the injury was synchronized with the angle of the participants’ knee joints, so that as the leg moved further from a neutral position, the feeling in the chest or arm grew stronger. Participants practiced while blindfolded, using one arm to indicate where they thought their leg was positioned based solely on what they felt from the stimulation.
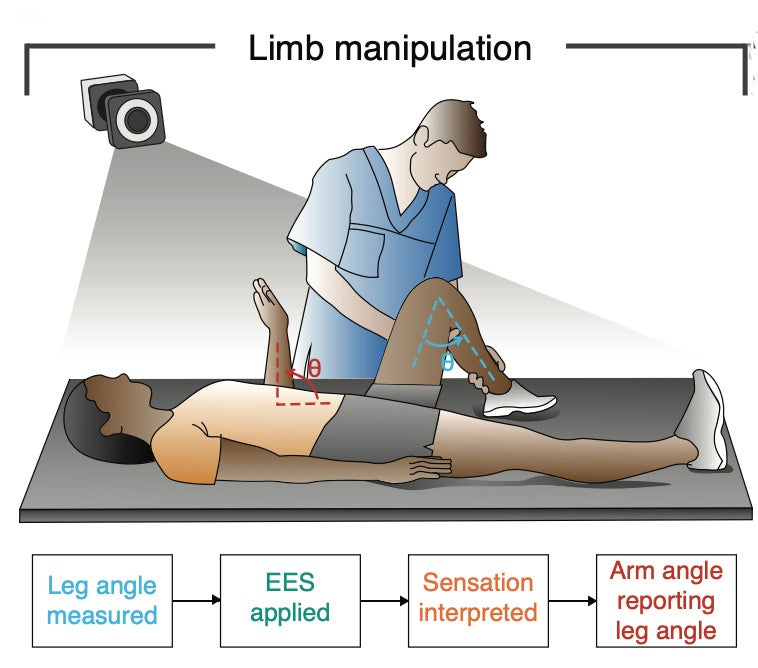
Two participants achieved correlations of 0.9 or above between their actual leg position and their reported perception of it. A third participant showed significantly better performance with the stimulation active than with it off, even though that person retained some residual sensation below the injury.
“We found that the participants could report the angle of their knee with high accuracy based on the intensity of sensations generated by stimulation,” Calvert said. “Participants indicated that this type of sensory feedback could be very useful in their daily life, such as transferring in and out of their wheelchair.”
One participant described learning the system over time: “I know that’s a pointed left foot, or I know that’s a flexed right foot. The more you did the stimulation, the more I learned.”
The central experiment brought both systems together. Supported by an overhead harness and assisted by physical therapists, the participants performed stepping movements on a treadmill while both electrode arrays delivered stimulation simultaneously.
The lower array activated the muscle groups needed for walking. The upper array provided real-time sensory feedback tied to foot position during the gait cycle, pulsing to signal when each foot struck the ground. Participants were not relying on their eyes.
One participant correctly identified foot strikes on both sides more than 87 percent of the time, with most correct responses occurring within about half a second of actual contact. A second participant showed significantly higher accuracy when the upper array was active than when it was off.
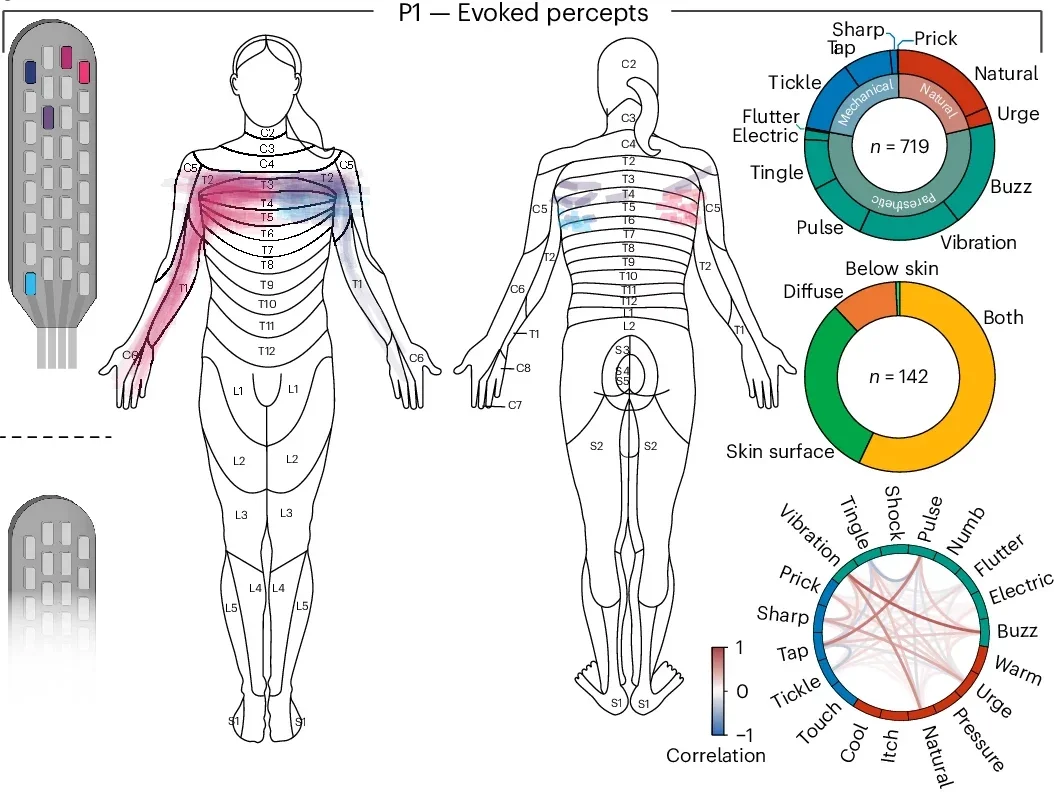
The chest sensation the first participant described, the one that told her where her foot was, was not a perfect replica of normal feeling. Participants described their sensations in varied terms, ranging from a light flutter just beneath the skin to something deeper that felt like it came from within the muscle. But they could use it. That distinction between accurate and natural is central to understanding what this research actually achieved.
The trial was brief by design. The electrode arrays were externalized, meaning the connecting wires exited the skin, which required participants to stay hospitalized throughout the study. That constraint prevented any exploration of long-term rehabilitation effects, real-world use, or how the system might perform outside a controlled lab setting.
The researchers also noted that stimulation above the injury, while effective for sensory substitution, produced sensations in body areas unrelated to the legs. The brain can learn to reinterpret those signals, as participants demonstrated, but the process takes time. A cochlear implant provides an instructive parallel: the sounds it produces initially feel unnatural, but over months of use, most recipients integrate them into normal hearing.
No device-related adverse effects were reported across all three participants, clearing the way for longer and more ambitious studies. The team plans to recruit new participants for trials conducted outside the hospital, testing whether the system holds up in daily life.
“There’s reason to believe that coordinated stimulation across an injury site could produce positive rehabilitation effects,” Borton said. “That’s not something we were able to fully explore in this study, but that we plan to pursue in future work.”
Dr. Jared Fridley, chief of spinal neurosurgery at the University of Texas at Austin, who contributed to the research during his time at Rhode Island Hospital, framed the significance of the two-way approach directly: “By simultaneously restoring motor activation and meaningful sensory feedback, we’re moving beyond isolated function toward coordinated, purposeful movement. That’s a critical step if neurotechnology is going to translate into real-world independence for people living with severe spinal cord injury.”
For the roughly 300,000 Americans living with spinal cord injuries, current technology can partially restore movement in some cases, but restoring sensation has remained an unsolved problem. This research demonstrates that both functions can be targeted at once in people with complete injuries, using hardware that is already common in clinical neurosurgery for pain management.
The machine learning approach developed here could reduce the time and clinical resources needed to configure these systems for individual patients, lowering one of the main barriers to broader adoption. A fully implantable version of this technology, without the externalized wires that restricted this trial to a hospital setting, is technically feasible and represents the logical next step.
Beyond walking, participants pointed to everyday situations where sensing limb position matters, feet slipping off wheelchair footrests, transferring from a chair, knowing whether a leg is in a safe position without looking. The technology demonstrated here would not need to restore natural feeling to be genuinely useful. It would only need to be reliable, learnable, and wearable.
Research findings are available online in the journal Nature Biomedical Engineering.
The original story “Electrical stimulation can restore ability to move limbs after spinal cord injury” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Electrical stimulation can restore ability to move limbs after spinal cord injury appeared first on The Brighter Side of News.