Some fish, it turns out, are morning people. They swim hard during daylight, sleep mostly at night, and tend to live longer. Others drift into daytime napping early in life, move less vigorously, and die sooner. The remarkable thing is that this divergence shows up well before middle age, in animals that are genetically similar and raised under identical conditions.
A study published in Science has mapped the full arc of aging in individual vertebrates for the first time, tracking dozens of small fish continuously from adolescence to natural death and finding that behavior, observed early enough, can predict how much time an animal has left.
The research was led by postdoctoral scholars Claire Bedbrook and Ravi Nath at Stanford’s Wu Tsai Neurosciences Institute, working in collaboration between the labs of geneticist Anne Brunet and bioengineer Karl Deisseroth. It was supported by the Knight Initiative for Brain Resilience.

Most aging studies work by comparing groups of young animals against groups of old ones. That approach yields useful data, but it misses something: the lived trajectory of a single individual through time, the moment aging begins to tilt in one direction, and what that tilt looks like in daily behavior before it becomes obvious.
To watch aging unfold continuously, the researchers needed an animal with a compressed lifespan. They chose the African turquoise killifish, a small vertebrate with a typical adult life of four to eight months that shares key biological features with longer-lived species, including a complex brain. The Brunet lab has spent years developing the killifish as a model for aging research.
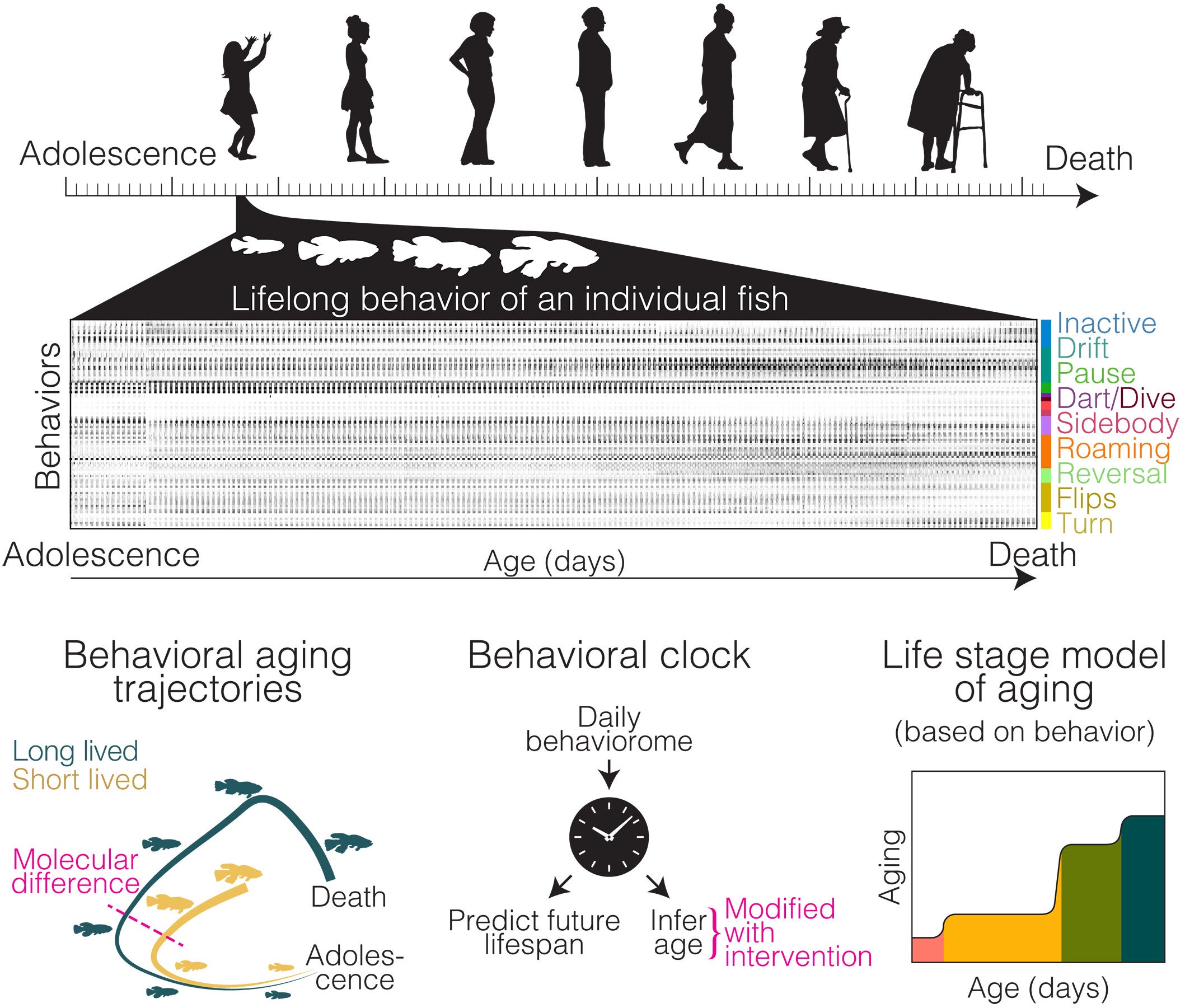
Eighty-one fish were placed in individual camera-monitored tanks at puberty and filmed at 20 frames per second until they died. Every moment was recorded. In total, the system generated billions of video frames, which a deep-learning algorithm then translated into 100 distinct behavioral patterns, short recurring movements and rest states that captured how each animal spent its time throughout the day and night.
“Behavior is a wonderfully integrated readout, reflecting what’s happening across the brain and body,” said Brunet, the Michele and Timothy Barakett Professor of Genetics at Stanford Medicine. “Molecular markers are essential, but they capture only slices of biology. With behavior, you see the whole organism, continuously and non-invasively.”
When the team compared fish that ultimately lived longer against those that died sooner, they found the two groups had already started diverging by around 70 to 100 days of age, well before what counts as midlife for this species. The short-lived fish were not eating less or growing differently. The differences showed up in how they moved and when they slept.

Fish headed toward shorter lives began sleeping increasingly during the day as young adults. Their longer-lived counterparts kept sleep tightly confined to nighttime hours. The longer-lived fish also moved with greater vigor during the day, reaching higher peak speeds when darting around the tank.
Machine-learning classifiers trained on just a few days of behavioral data from middle-aged fish predicted future lifespan with an accuracy exceeding 70 percent.
“Behavioral changes pretty early on in life are telling us about future health and future lifespan,” Bedbrook said.
That predictive signal is not merely behavioral. When the team sacrificed a separate cohort of fish at middle age and sequenced gene activity across eight organs, the liver showed the clearest molecular separation between animals on longer versus shorter trajectories. Fish destined for shorter lives had higher activity in genes linked to ribosome production and protein synthesis. Those same gene signatures intensify with age more broadly, suggesting they may be driving the divergence rather than simply reflecting it.
One of the study’s more unexpected findings involved the shape of aging itself. The common assumption is that decline accumulates gradually, a slow dimming rather than a series of steps. The killifish data told a different story.
Most animals underwent two to six abrupt behavioral transitions across their lives, each lasting only a few days, followed by long stretches of relative stability that could persist for weeks. Animals progressed through these stages in sequence, rarely cycling back. The pattern held across the cohort.
“We expected aging to be a slow, gradual process,” said Bedbrook. “Instead, animals stay stable for long periods and then transition very quickly into a new stage. Seeing this staged architecture appear from continuous behavior alone was one of the most exciting discoveries.”

The researchers suggest that aging may work more like a Jenga tower than a melting ice cube. Stresses accumulate without visible consequence until a threshold is crossed and the system suddenly reorganizes into a new, stable configuration. Six distinct life stages emerged from the data. Young animals clustered together at the start, then fanned out into diverging midlife and late-life stages, with the transition into old-life stages marked by a loss of tight circadian timing and the spread of inactivity into daytime hours.
Patterns from human molecular biology echo this picture. Protein and transcriptomic studies in people have found waves of biological change concentrated in the fourth, sixth, seventh, and eighth decades of life, consistent with discrete transitions rather than continuous drift.
The team also tested how dietary restriction affected behavioral aging. A separate cohort of 39 fish was fed only three times per day, all in the morning, rather than the standard seven feedings spread across daylight hours. These fish woke earlier, slept more at night, and moved with higher peak speeds than normally fed animals.
When the behavioral aging clock was applied to the dietary restriction group, it estimated their biological age as approximately 42 days younger than their true chronological age. The restricted fish were aging along a similar path as their counterparts but traversing it more slowly.
That result aligns with findings in calorie-restricted mice and primates, which tend to die of similar diseases as controls but at older ages. The implication is that dietary restriction does not redirect the trajectory of aging so much as it slows the pace of travel along it.
The team also tracked female killifish and found they progressed through a distinct and accelerated aging path compared with males, consistent with the sexual dimorphism in aging seen across the animal kingdom. The behavioral lifespan classifier, trained primarily on male data, still correctly predicted short lifespan in 74 percent of females tested, suggesting some predictive features cross sex lines.
Both Bedbrook and Nath will continue this research as they open their own laboratories at Princeton University, carrying the killifish system and its questions into a new setting.
The parallels to human aging are not lost on the researchers. Nath is particularly interested in whether sleep itself can be manipulated to slow aging, and whether intervening before decline begins can alter an individual’s trajectory. Deisseroth’s lab is pursuing tools to continuously monitor neural activity alongside behavior in the same animals, which could reveal whether the brain leads or follows the rest of the body in aging transitions.
Wearable devices already collect continuous movement and sleep data from millions of people. Whether the same early behavioral signals that predicted lifespan in killifish could be extracted from that data in humans remains an open question, but one the researchers consider worth asking seriously.
“We now have the tools to map aging continuously in a vertebrate,” Bedbrook said. “With the rise of wearables and long-term tracking in humans, I’m excited to see whether the same principles, early predictors, staged aging, divergent trajectories, hold true in people.”
If behavioral signals captured early in life reliably forecast how aging unfolds, that knowledge could reshape preventive medicine. Clinicians might eventually use continuous data from wearable sensors to identify individuals whose activity and sleep patterns resemble early-aging trajectories, prompting intervention before the biological stages that predict poor health have taken hold.
Dietary approaches that demonstrably slow the behavioral pace of aging in animal models offer concrete targets for human translation. The staged model of aging also has implications for drug development: if aging proceeds through discrete transitions rather than gradual decline, therapeutic windows may exist just before each transition, when the system is most vulnerable to being tipped in either direction.
Sleep, circadian timing, and peak physical activity emerge from this study as measurable, modifiable signals tied to longevity, accessible without invasive testing and continuously trackable with technology already on millions of wrists.
Research findings are available online in the journal Science.
The original story “Daily movement and sleep patterns can predict lifespan with striking accuracy” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Daily movement and sleep patterns can predict lifespan with striking accuracy appeared first on The Brighter Side of News.