Platinum works beautifully. It speeds up chemical reactions, holds up under punishment, and sits reliably at the center of industrial processes worth billions of dollars annually. It also costs roughly 20,000 times more than the most abundant metal in the Earth’s crust.
That gap has driven decades of searching. If chemists could coax a cheaper, more common element into behaving like platinum and its chemical cousins, the implications for manufacturing, sustainability, and cost would be enormous. A team at King’s College London has now taken a significant step in that direction, and the element they’re working with is aluminum.
Dr Clare Bakewell and her laboratory have created a new form of aluminum, published in Nature Communications, that can break apart tough chemical bonds and generate molecular structures never previously observed. The discovery opens a line of chemistry that the researchers say goes beyond simply mimicking precious metals.
“We chose aluminum as it’s super abundant, making it approximately 20,000 times less expensive than precious metals such as platinum and palladium,” Bakewell said.

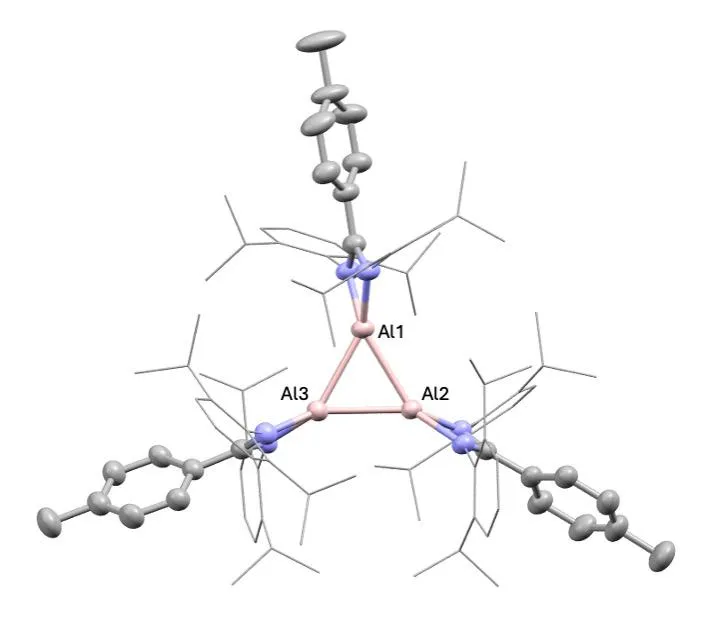
The centerpiece of the work is a molecule called a cyclotrialumane: three aluminum atoms arranged in a triangular ring, held together by aluminum-aluminum bonds. That arrangement sounds simple. Getting there wasn’t.
Aluminum almost always behaves predictably, sitting in a stable, well-understood chemical state. For it to do the kinds of things that make transition metals useful in catalysis and synthesis, it needs to be pushed into a different, much less stable condition. Bakewell’s lab works with aluminum in that unusual state, where it carries a lower charge than normal and becomes highly reactive as a result.
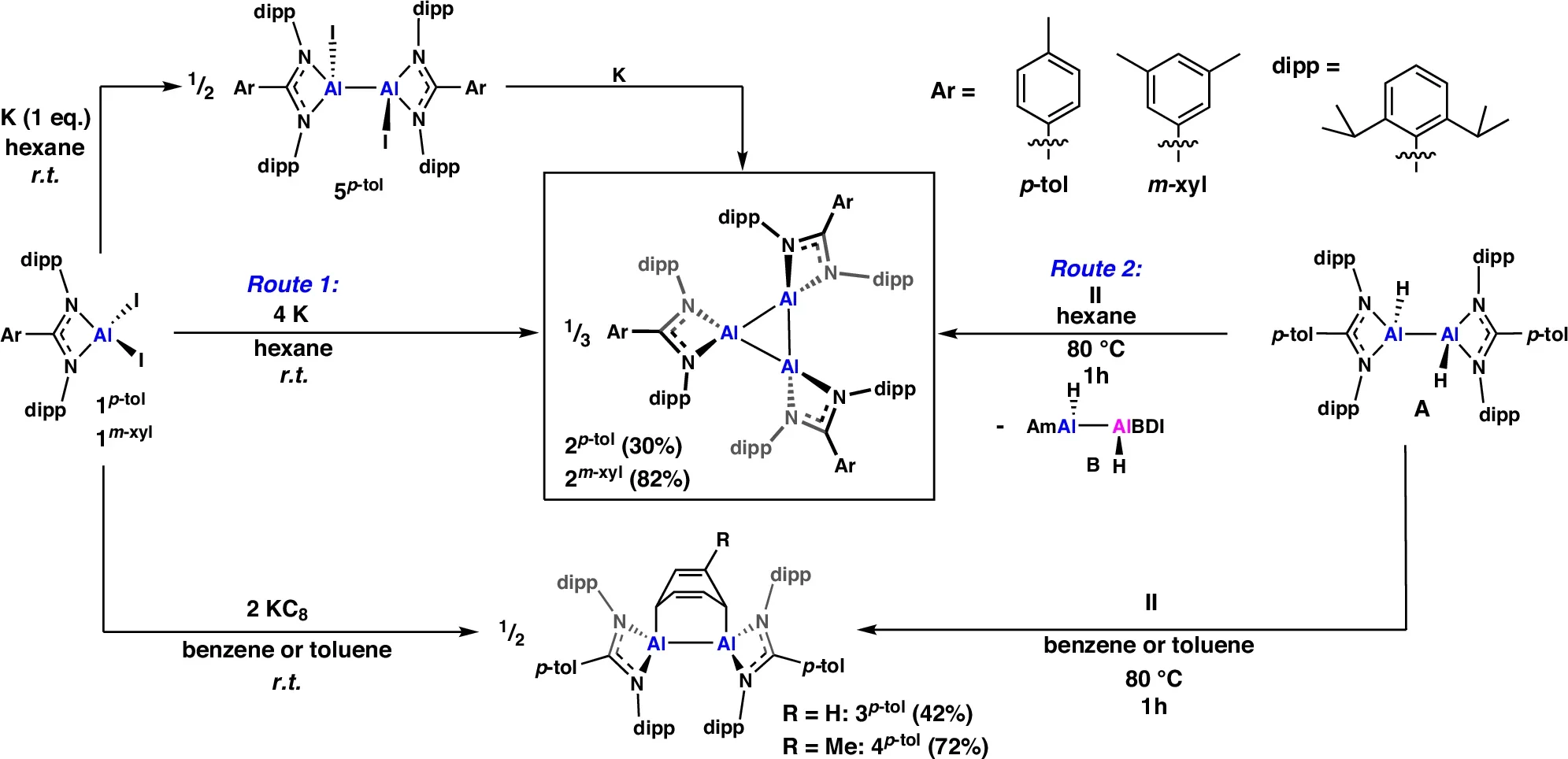
Monomeric versions of reactive aluminum, single atoms stabilized by surrounding molecular scaffolding, have been known since 2000. Dimers, pairs of aluminum atoms bonded together, followed in 2017. Tetramers, clusters of four, were actually the first reported back in 1991. Trimers, three atoms in a ring, had a conspicuous absence in the catalogue. No one had isolated a neutral cyclic version.
Bakewell’s team filled that gap by reducing aluminum compounds using potassium metal, a process that strips away charge and produces the reactive aluminum species needed to form the trimeric ring. The reaction produced dark red crystalline solids; X-ray analysis confirmed the triangular aluminum core.
One structural detail stood out: the trimer holds its shape when dissolved in solution. Many related compounds fragment as soon as they leave the solid state. For a molecule to be genuinely useful in chemistry, it needs to survive dissolving. This one does.
“What’s special about this work is that we’re pushing the boundaries of chemical knowledge,” Bakewell said. “Most excitingly, we can use this aluminum trimer to build completely new compounds with levels of reactivity that have never been observed before.”
Testing what the cyclotrialumane actually does revealed behaviour that had no prior precedent.
At room temperature, exposing the trimer to ethylene, a simple two-carbon gas and one of the most important feedstocks in industrial chemistry, produced an immediate color change and a new product. A molecule of ethylene had inserted itself into the aluminum core, creating a five-membered ring containing both aluminum and carbon atoms. That structure, a five-membered ring of this type, had never been seen for any metal, transition or main group.
Left in excess ethylene, the reaction continued. A seven-membered ring formed next, also without precedent. Further reaction produced additional products through what the researchers describe as stepwise chain growth, each ethylene molecule adding in sequence to build larger and larger structures.
The trimer also split dihydrogen, the simple molecule of two hydrogen atoms, at room temperature. That reaction is one of the standard benchmarks for testing whether a main group element compound can perform like a transition metal catalyst. The cyclotrialumane passed.
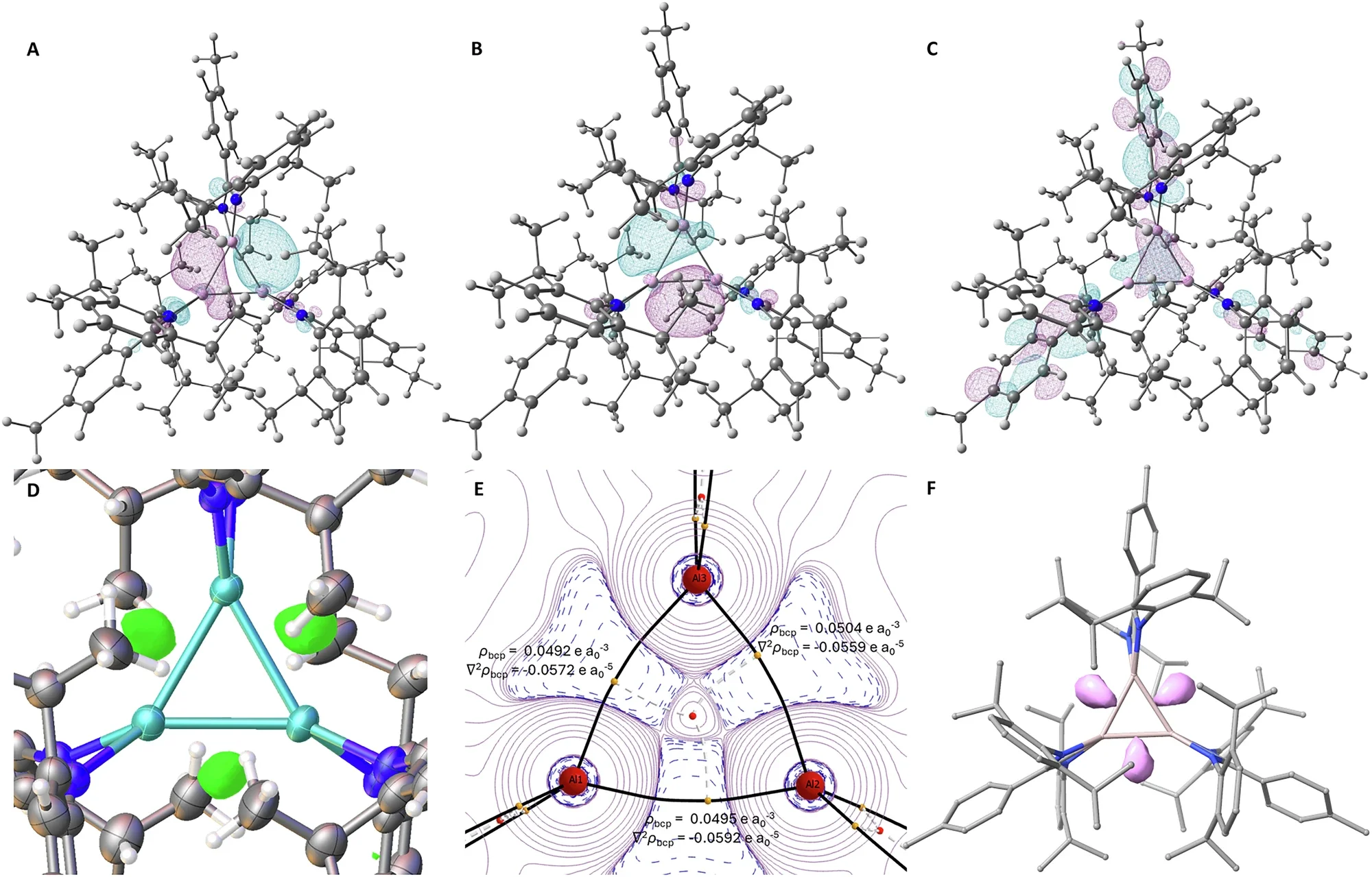
Computational modelling, running detailed calculations of how electrons are distributed across the aluminum triangle, revealed that the three aluminum atoms share bonding electrons in a way that combines covalent character with additional stabilization from interactions between the bonds themselves. The geometry of the ring creates significant strain, which in turn drives the high reactivity.
The research sits firmly in fundamental chemistry. Bakewell is explicit about where the work stands: “We’re very much in the exploratory phase and we’re just at the start of beginning to unlock the capability of these earth-abundant materials.”
No industrial process runs on cyclotrialumanes today. The synthesis, while reproducible and reasonably high-yielding, is carried out in carefully controlled laboratory conditions using inert atmospheres. The path from here to a practical catalyst or industrial reagent involves many further steps that the paper doesn’t claim to have taken.
The study also acknowledges that aluminum extraction, while far cheaper than platinum mining, is itself energy-intensive. The sustainability case rests on relative abundance and cost, not on a zero-impact supply chain.
What the work does establish is a previously missing piece of aluminum chemistry. Neutral trimeric aluminum in this reactive state had simply not existed in the chemical literature. Its reactivity patterns with ethylene, dihydrogen, and other molecules now create a foundation for understanding what this class of compound can do and where it might eventually be useful.
“From what we’ve seen already, this chemistry could support a transition to cleaner, greener and cheaper chemical production, whilst making new discoveries along the way,” Bakewell said.
The significance here is both immediate and long-range. In the short term, the cyclotrialumane fills a genuine gap in the periodic table of reactive molecular structures. Chemists now have access to an aluminum compound with a reactivity profile that overlaps with, and in some respects surpasses, what transition metals can achieve in specific reactions.
Over a longer horizon, the ability to perform chemical transformations using earth-abundant, inexpensive aluminum rather than scarce and geopolitically sensitive metals like platinum, palladium, or rhodium addresses a real and growing concern in industrial chemistry. Many of the most useful transition metals are concentrated in politically unstable regions, and their extraction carries significant environmental cost. A versatile aluminum-based chemistry platform wouldn’t eliminate that dependency overnight, but research like this builds the scientific case for alternatives.
The ligand system that stabilizes the cyclotrialumane is also described as readily modifiable, meaning the same basic architecture can be tuned to produce different variants with different properties. That tunability is precisely what makes a new compound class valuable as a research platform rather than a one-off curiosity.
Research findings are available online in the journal Nature Communications.
The original story “New triangular form of aluminum may be more valuable than platinum” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post New triangular form of aluminum may be more valuable than platinum appeared first on The Brighter Side of News.