A small molecule inside your body quietly fuels life every day. It helps your cells turn fat into energy, keeping your heart beating and your muscles moving. Now, scientists have found a way to watch that process unfold in real time, offering a rare glimpse into how diseases like cancer reshape the body’s energy use.
Researchers at King’s College London have developed a new imaging tool that tracks how cells use fats for fuel. Their work introduces a tracer that reveals how tumors and healthy tissues rely on a molecule called carnitine.
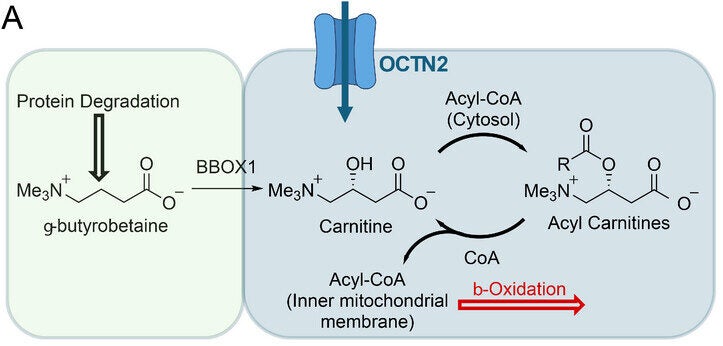
Carnitine plays a simple but vital role. It carries fatty acids into the mitochondria, the part of the cell that produces energy. Without it, your body would struggle to use fat as fuel. Until now, scientists could not easily observe how this process works inside living organisms.
“Using this novel tracer, we can look at carnitine metabolism in living subjects for the very first time,” said Professor Tim Witney, Professor of Molecular Imaging at the School of Biomedical Engineering & Imaging Sciences. “Understanding how the body uses different fuel types, both in health and across a range of diseases, could enable the development of new metabolic drugs and help us optimise healthy living.”
Your body depends on a balance of nutrients to function. Under normal conditions, it switches between fuels like glucose and fat based on need. Disease can disrupt that balance.
In cancer, cells often consume large amounts of glucose. That pattern has shaped how doctors detect tumors for years. However, this new study shows that some aggressive cancers also rely heavily on fats.
To uncover this hidden behavior, the research team focused on carnitine. This molecule acts as a shuttle, moving fatty acids into the mitochondria. Once inside, those fats are broken down to generate energy.
Carnitine also helps maintain chemical balance inside cells. It removes harmful compounds and supports normal metabolism. When this system breaks down, it can contribute to diseases such as cancer and heart conditions.
Despite its importance, scientists have struggled to study carnitine directly inside living systems. Traditional tools could not track it with enough precision.
To solve this problem, the team created modified versions of carnitine that could be detected in imaging scans. They introduced fluorine, an element that is rare in the human body. This makes it easier to track using advanced techniques.
The researchers developed two versions. One is fluoromethylcarnitine, known as FMC. The other is a radioactive form called [18F]FMC, designed for use in positron emission tomography, or PET imaging.
PET scans allow scientists to visualize how molecules move through the body. They are widely used to track glucose in cancer. This new tracer brings that same level of insight to fat metabolism.
![In vitro evaluation of [18/19F]FMC.](https://www.thebrighterside.news/uploads/2026/03/fat-3-e1774797401731.jpg)
Creating the tracer required careful design. The team needed to modify carnitine without disrupting its function. They placed the fluorine tag in a part of the molecule that would not interfere with its role in energy production.
After several steps, they produced a stable compound that could be used in imaging studies. Tests confirmed that the tracer remained active long enough for detailed scans.
The next step was to see how the tracer behaves in living cells. Researchers used a human lung cancer cell line to test whether the modified molecule entered cells like natural carnitine.
The results were clear. Cancer cells absorbed the tracer quickly. When researchers added excess natural carnitine, uptake dropped sharply. This confirmed that both molecules compete for the same transport system.
They also used a drug called meldonium, which blocks the carnitine transporter. This reduced tracer uptake even further. These findings showed that the new tracer follows the same biological pathways as natural carnitine.
The team then examined what happens after the tracer enters the cell. Inside, carnitine normally forms compounds called acyl-carnitines. These are part of the process that breaks down fats for energy.
The tracer behaved in the same way. Within minutes, it converted into different forms inside the cell. Over time, it produced compounds that matched normal metabolic patterns.
![Imaging carnitine utilisation in vivo with [18F]FMC PET.](https://www.thebrighterside.news/uploads/2026/03/fat--e1774797425380.jpg)
This confirmed that the tracer does more than enter cells. It participates in real metabolic processes, making it a reliable tool for studying energy use.
After testing in cells, the researchers moved to live animal studies. They injected the tracer into healthy mice and tracked its movement using PET scans.
The tracer spread quickly through the bloodstream. It accumulated in the liver and kidneys, which play key roles in metabolism and filtration. The heart also showed strong uptake, reflecting its high demand for energy from fats.
Interestingly, skeletal muscle showed little signal. Scientists believe this is because muscle absorbs carnitine more slowly, even though it stores large amounts.
The team then tested what happens when carnitine levels are artificially increased. When mice received high doses of natural carnitine, the kidneys became overwhelmed. The tracer passed quickly into the bladder and was excreted.
This experiment helped confirm how the body regulates carnitine under different conditions.
The most striking findings came from tumor studies. Researchers implanted lung cancer cells into mice and used the tracer to monitor energy use in growing tumors.
The tumors absorbed the tracer steadily over time. After two hours, uptake levels were high enough to clearly distinguish tumors from surrounding tissue. This shows that cancer cells actively use carnitine to support their growth.
![Imaging aberrant carnitine utilisation in NSCLC with [18F]FMC PET.](https://www.thebrighterside.news/uploads/2026/03/fat-5-e1774797448465.jpg)
When the team blocked the carnitine transporter, tumor uptake dropped significantly. This confirmed that the process depends on specific biological pathways.
These results suggest that some cancers rely on fat metabolism more than previously thought. This insight could change how scientists approach treatment.
Instead of focusing only on glucose use, future therapies may target fat-based energy systems as well.
The study also revealed how different organs use carnitine in unique ways. Blood samples showed high levels of acetyl-carnitine, a common metabolic product.
In the heart, more complex forms of carnitine appeared. This reflects the heart’s heavy reliance on fat for energy. In contrast, other tissues showed simpler patterns.
These differences highlight how each organ adapts its metabolism to meet its needs. By mapping these patterns, scientists can better understand both health and disease.
The tracer also showed how drugs like meldonium affect the heart. This could help doctors monitor treatment response in patients with heart disease.
This research opens a new window into how the body uses energy. For the first time, scientists can track carnitine metabolism in living systems with precision.
The findings suggest that metabolic changes in cancer and heart disease may be more complex than once believed. By studying these changes, researchers can develop more targeted therapies.
The work also highlights the importance of looking beyond glucose. While sugar metabolism has dominated cancer research, fat metabolism may play an equally important role.
The team is now working to scale up production of the tracer. This will allow broader use in research and, eventually, clinical settings.
This study could reshape how doctors understand and treat diseases that involve metabolism. By tracking how cells use fat for energy, clinicians may detect cancer earlier or identify aggressive tumors more accurately. It could also lead to treatments that target fat metabolism, offering new options for patients who do not respond to current therapies.
In heart disease, the tracer may help identify metabolic changes before symptoms appear. Earlier detection could improve treatment outcomes and reduce long-term damage. The ability to monitor how drugs affect carnitine transport also provides a powerful tool for evaluating therapies in real time.
Beyond disease, the research may influence how scientists study human performance. Understanding how the body uses fat during exercise could guide better training strategies and supplement use. It may also help explain why some metabolic treatments show mixed results.
Overall, this tool brings scientists closer to understanding how the body fuels itself under stress, illness, and normal conditions. That knowledge could lead to more precise, personalized approaches to medicine and health.
Research findings are available online in the journal Advanced Science.
The original story “New imaging tool reveals how cancer reshapes the body’s energy use” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post New imaging tool reveals how cancer reshapes the body’s energy use appeared first on The Brighter Side of News.