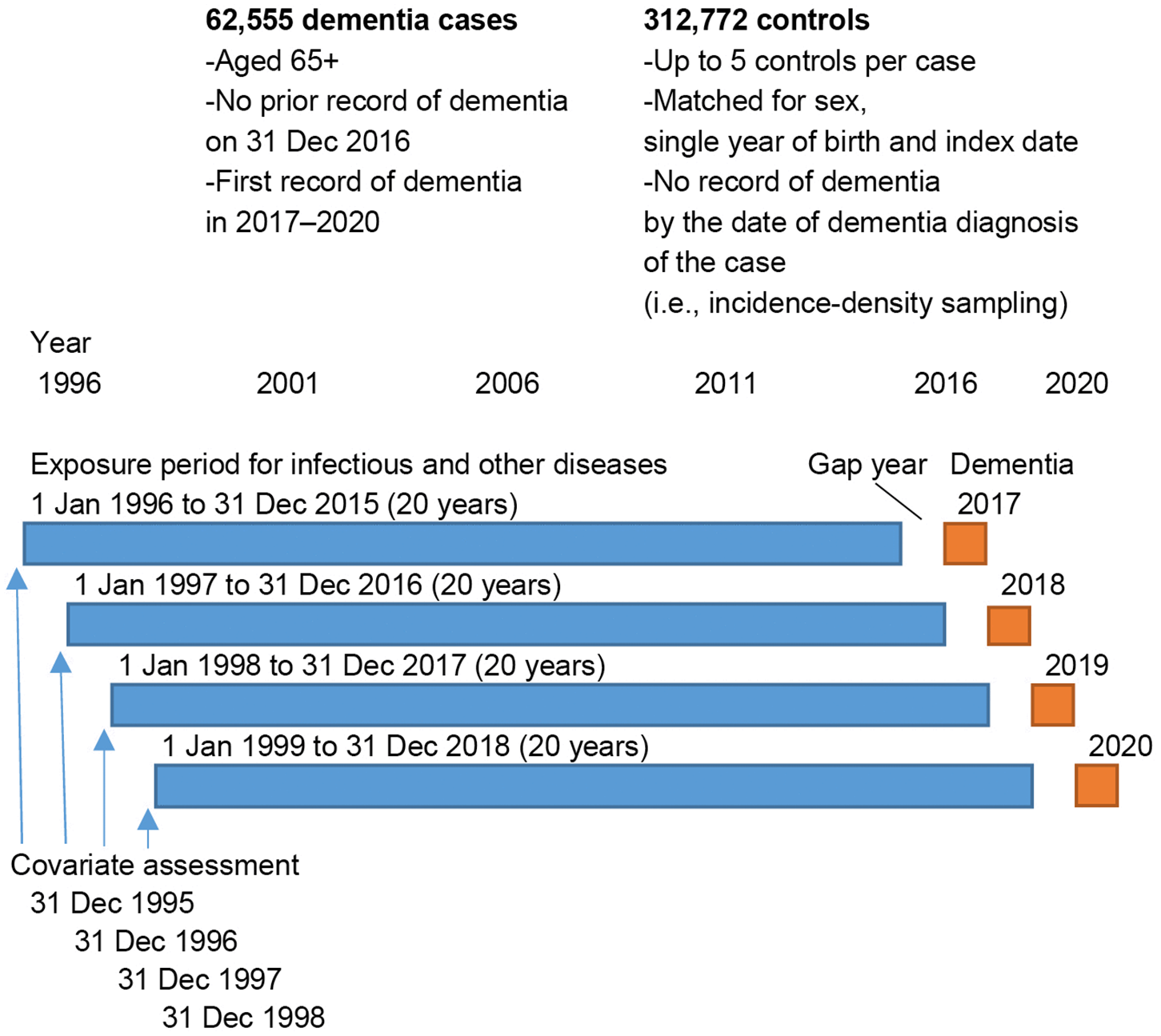
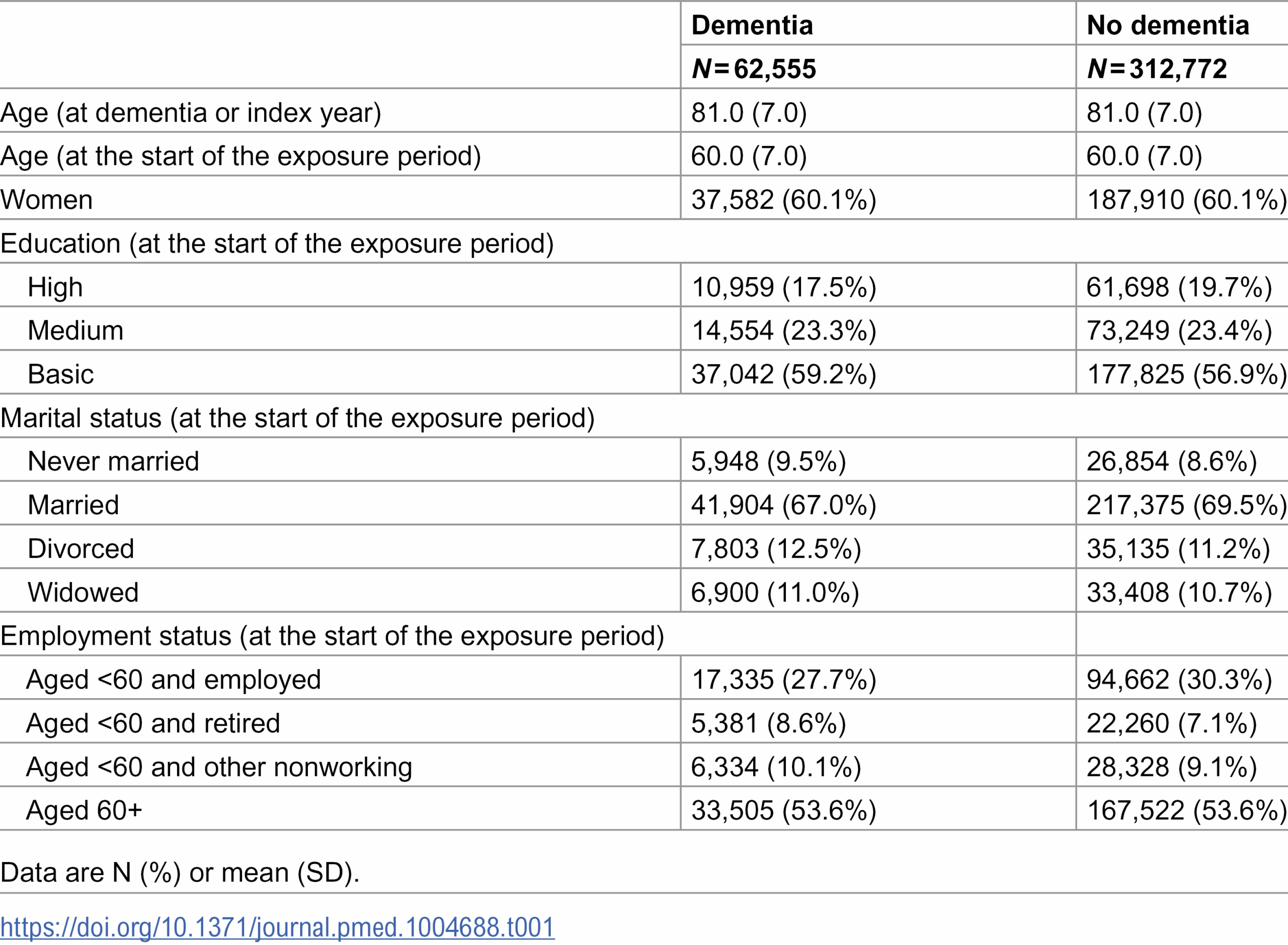
The human gut is alive with activity. Millions of microorganisms compete, cooperate, and coexist in ways that can profoundly affect your health. Yet, for all the research on which bacteria live in our intestines, scientists have only begun to understand the invisible rules that govern their communities. A new study by Pyry Sipilä of the University of Helsinki, Finland, and colleagues sheds light on how these microbial neighborhoods organize themselves and hints at ways they might one day be guided to improve health.
The research examined microbial communities not just as collections of species but as dynamic systems. Instead of cataloging who is present, the scientists focused on how these species interact. Their goal was to understand why certain community “types” emerge in some people and not others, even when diet and environment are similar.
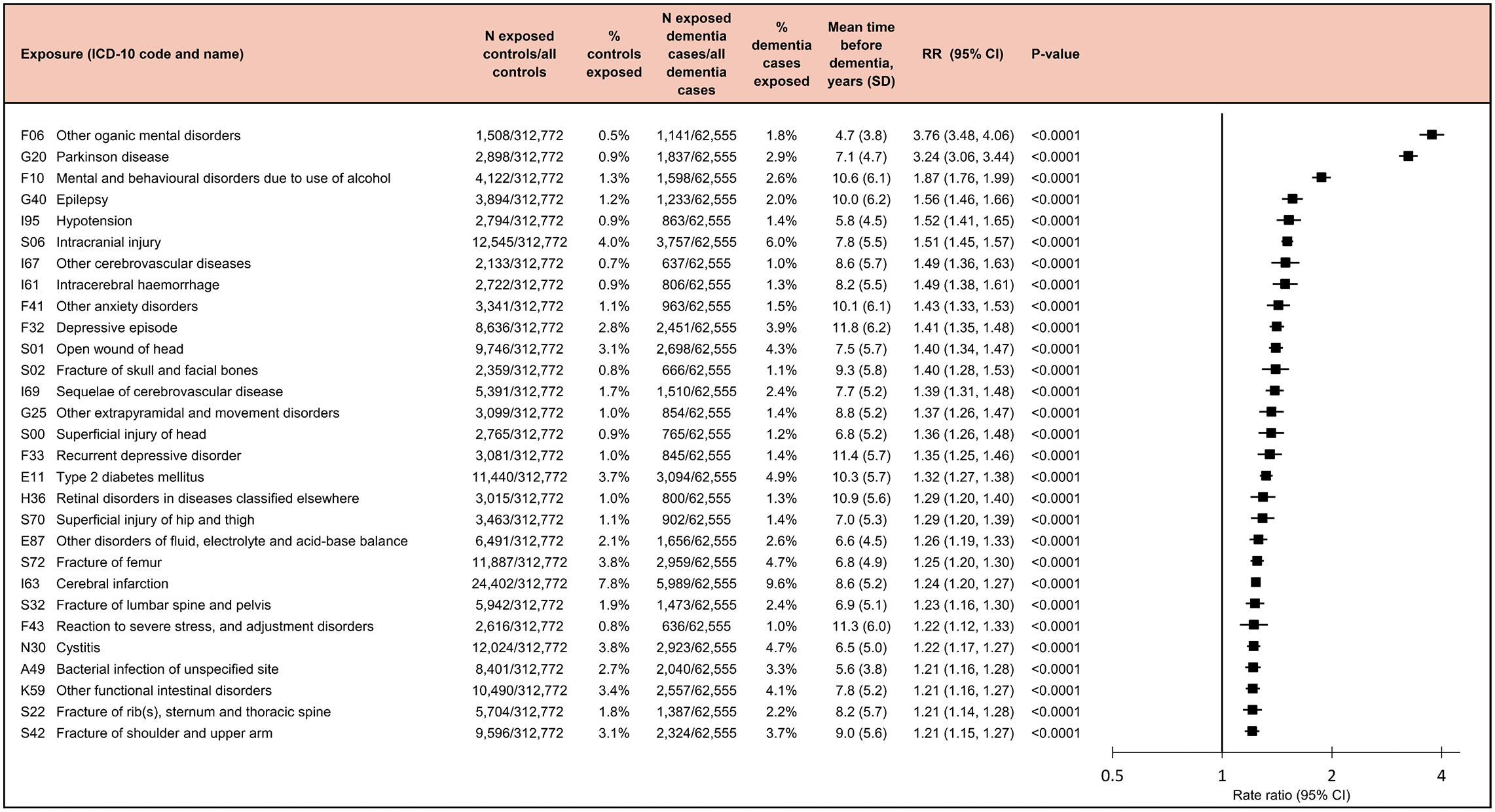
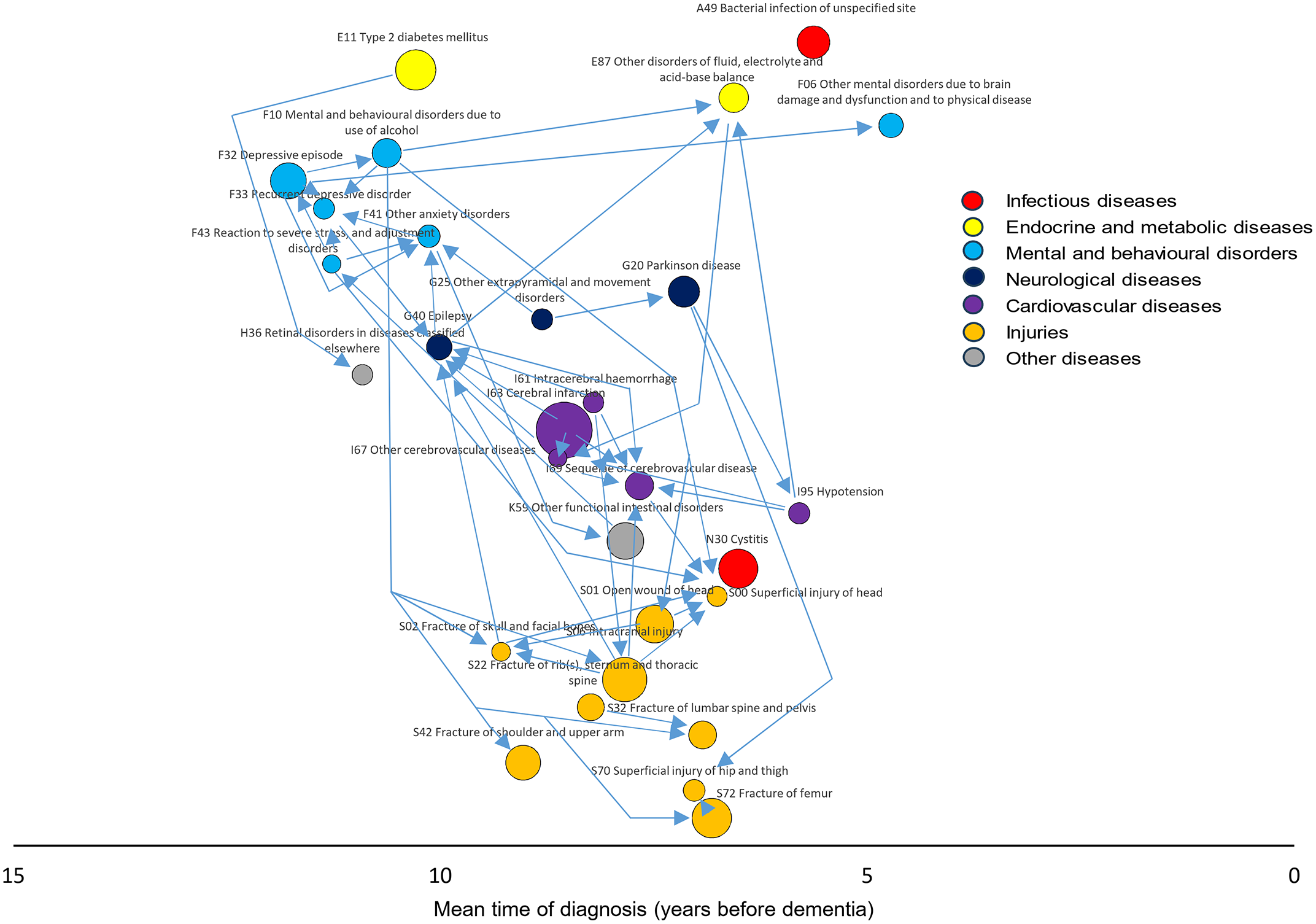
“Our findings suggest that the overall network structure is less important than the strength of interactions among key species,” they write. In other words, it’s not just which microbes exist in the gut, but which ones strongly influence the rest of the ecosystem.
Among the trillions of microbes in the body, a few species hold disproportionate power. The study describes these as “strongly interacting species,” microbes whose connections shape the behavior of entire communities. Even if they are not the most abundant, their presence can determine which community type forms.
The researchers found that when these influential species are present, a particular community configuration emerges. Remove them, and the community can shift into a different stable arrangement. This insight helps explain why microbiomes vary so dramatically between individuals, even among people living in the same household or consuming similar diets.
These strongly interacting species act as control points within the microbial ecosystem. By targeting these points, it may be possible to steer the community toward a desired state. This control could be exercised through diet, probiotics, or other interventions designed to promote beneficial species or suppress harmful ones.
To explore these patterns, the team used a dynamic systems model. Unlike traditional approaches that provide a single snapshot of the microbiome, this method simulates how communities evolve over time. By adjusting the strength of species interactions, the model predicted shifts in community types, illustrating how small changes could ripple through the system.
The model also revealed multiple stable states for microbial communities. Once a community settles into one configuration, it tends to remain there unless a major disruption occurs. This stability explains why microbiomes often resist change, even when people drastically alter their diets or lifestyle.
Transitions between community types can happen when certain thresholds are crossed. For example, if a key species’ influence drops or rises enough, the system may flip into a new stable state. This suggests that targeted interventions could potentially move a microbiome from one type to another, rather than relying on broad, generalized strategies.
Understanding these dynamics has significant implications for human health. The microbiome affects digestion, immunity, and even behavior. If scientists can identify which community types correlate with better health outcomes, they could develop therapies aimed at promoting these configurations.
Personalized medicine stands to gain from this approach. Each individual’s microbiome is unique, shaped by genetics, environment, and lifestyle. Treatments that focus on the key species influencing community structure could be tailored to a person’s specific microbial ecosystem, rather than applying a one-size-fits-all solution.
Beyond treatment, the findings may improve disease diagnosis. Certain illnesses are associated with specific microbial community types. By mapping these patterns, doctors could detect risks earlier and design interventions to prevent or mitigate disease progression.
Despite the promise, challenges remain. The human microbiome is incredibly complex, and researchers caution that controlling it in practice will require more study. Diet, genetics, and environmental exposures all influence microbial interactions, and the effects of interventions may vary widely between individuals.
Future research will likely focus on identifying strongly interacting species in different body regions and mapping how they influence health. Scientists may also explore how these interactions shift over time or in response to treatment, aiming to predict and guide the behavior of microbial communities.
Dynamic modeling will continue to play a central role. By simulating various scenarios, researchers can test hypotheses about community behavior and potential interventions before applying them in humans. This approach could accelerate the development of therapies that precisely reshape the microbiome to promote wellness.
This study reframes the microbiome as a dynamic, interconnected system, rather than a static collection of microbes. The behavior of a few influential species drives the structure of entire communities, suggesting a new avenue for interventions. By focusing on these key players, scientists hope to shift microbiomes toward states that support better digestion, immunity, and overall health.
The findings underscore that microbial communities are resilient but not unchangeable. They are shaped by strong interactions, and understanding these interactions may unlock opportunities to guide them. This perspective moves the field closer to treatments that are proactive, precise, and personalized.
The work opens doors to medical strategies that aim to control the microbiome, from improving gut health to potentially influencing broader physiological and cognitive outcomes. While practical applications remain on the horizon, this research highlights a path toward harnessing the human microbiome as a dynamic partner in health.
By identifying the species that control microbial community types, scientists could design interventions that reshape the microbiome deliberately.
Such approaches could lead to better digestive health, improved immune responses, and personalized therapies for metabolic or inflammatory disorders.
Understanding and leveraging these control points may make it possible to stabilize healthy microbiomes, reduce disease risk, and tailor treatments to individual patients.
Research findings are available online in the journal PLOS Medicine.
The original story “Scientists reveal the hidden forces shaping the human microbiome” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Scientists reveal the hidden forces shaping the human microbiome appeared first on The Brighter Side of News.