A drug long used to fight cancer may soon take on a very different role. Researchers at the University of Illinois Chicago have found that doxorubicin, an FDA-approved chemotherapy medicine, can block drug-resistant herpes infections in early studies. The discovery offers hope for patients who face limited treatment options when standard antivirals stop working.
The research centers on herpes simplex virus type 1, known as HSV-1. This virus infects billions of people worldwide and stays in the body for life. For many, it causes cold sores. For others, especially those with weakened immune systems, it can lead to serious complications, including brain inflammation and organ failure.
“This opens up an unexpected, potentially fast-moving path toward treating drug-resistant herpes infections,” said Deepak Shukla, a virologist in the College of Medicine at UIC. “HSV-1 infections have serious, sometimes life-threatening consequences, and this drug may help save lives.”
HSV-1 has challenged doctors for decades. Standard antiviral drugs such as acyclovir can control outbreaks by stopping the virus from copying itself. These medicines help reduce symptoms, but they do not remove the virus from the body. HSV-1 can hide in nerve cells and return later.
Over time, some strains develop resistance to these treatments. This problem is especially severe in immunocompromised patients, including those undergoing cancer treatment. When resistance develops, options become limited and often more toxic.
Drug-resistant infections can be difficult to treat and may lead to severe outcomes. In vulnerable patients, HSV-1 can spread beyond the skin or eyes and cause life-threatening complications. These cases highlight the urgent need for new strategies that go beyond traditional antiviral approaches.
Researchers at UIC turned to a different idea. Instead of developing a brand-new drug, they searched for an existing one that could be repurposed. This approach can shorten the time needed to reach patients because safety profiles and dosing are already known.
In 2024, Shukla’s team developed a screening tool called HerpDock. This system scans large numbers of chemical compounds and flags those that may work against herpes infections. It searches for unexpected matches by analyzing how molecules might interact with viral processes.
The program identified doxorubicin as a strong candidate. The drug has been used for years to treat various cancers. It is well studied, widely available, and already approved by the U.S. Food and Drug Administration.
“We were excited when we realized that doxorubicin is already FDA-approved,” Shukla said. “That matters because its safety profile and dosing are already well-understood. This drug could reach clinicians and patients much faster than a brand-new discovery.”
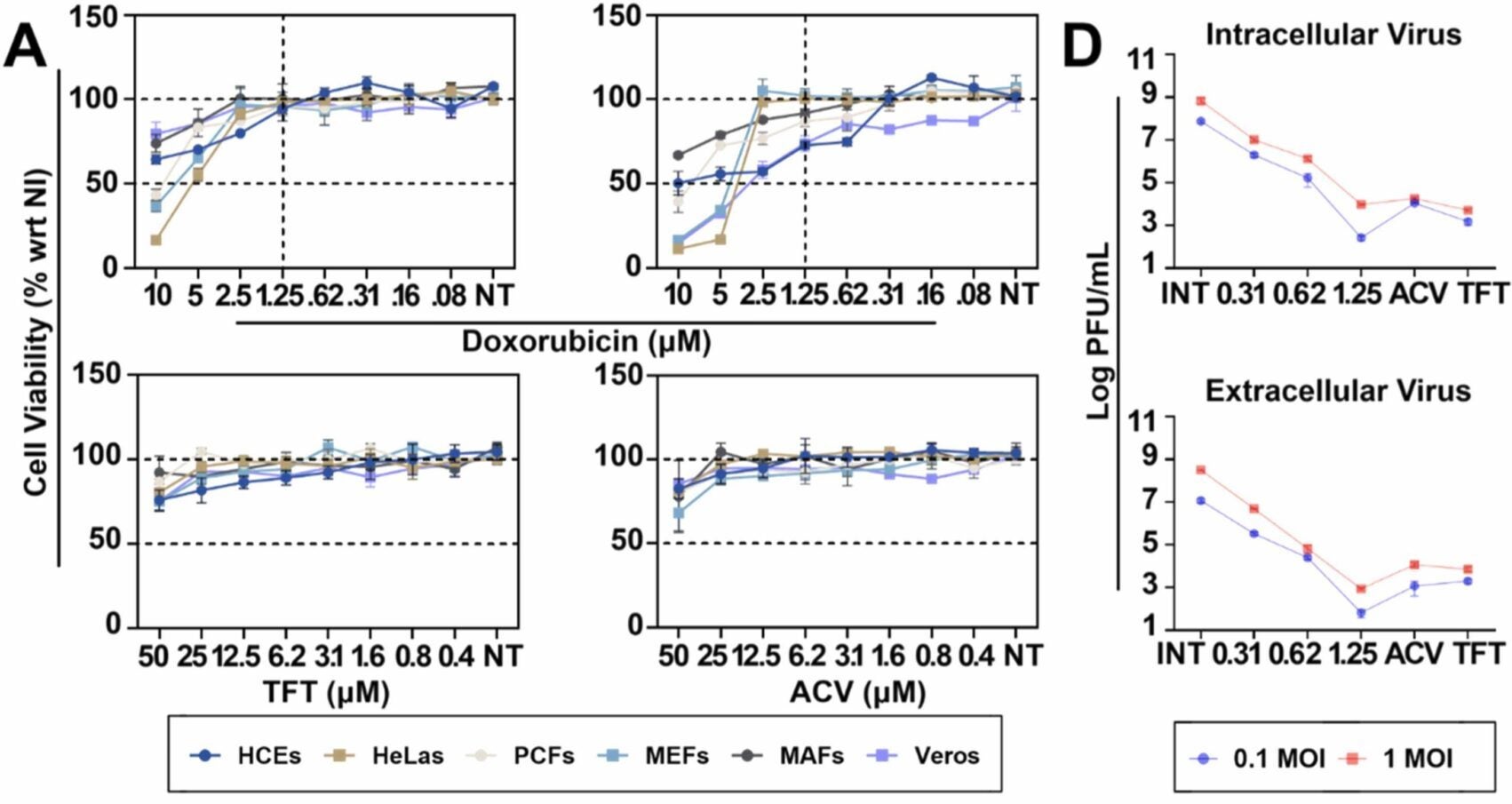
The team then moved from prediction to testing. They evaluated how doxorubicin performed against HSV-1 in several experimental settings, including human cells, tissue models, and mice.
Most antiviral drugs attack the virus directly. They target the machinery the virus uses to copy its genetic material. Doxorubicin works differently. It targets a pathway inside human cells that the virus depends on to infect and spread.
This pathway, known as PI3K–AKT–mTOR, helps regulate cell growth and survival. HSV-1 uses it to enter cells and take control of cellular processes. By disrupting this pathway, doxorubicin prevents the virus from gaining a foothold.
“We were excited to see how doxorubicin halts the virus at its source,” said Pankaj Sharma, a postdoctoral researcher and first author of the study.
In experiments, the drug consistently blocked infection across several HSV-1 strains. This included strains that no longer respond to acyclovir. The results remained strong across different models, suggesting a broad effect.
By targeting the host cell instead of the virus, the drug may also reduce the chance of resistance developing. Viral mutations often allow escape from direct antiviral drugs. A host-based approach makes that escape more difficult.
The research team tested doxorubicin in multiple systems to confirm its effectiveness. In human cell cultures, the drug reduced infection rates across different viral strains. In tissue models, it limited the virus’s ability to spread.
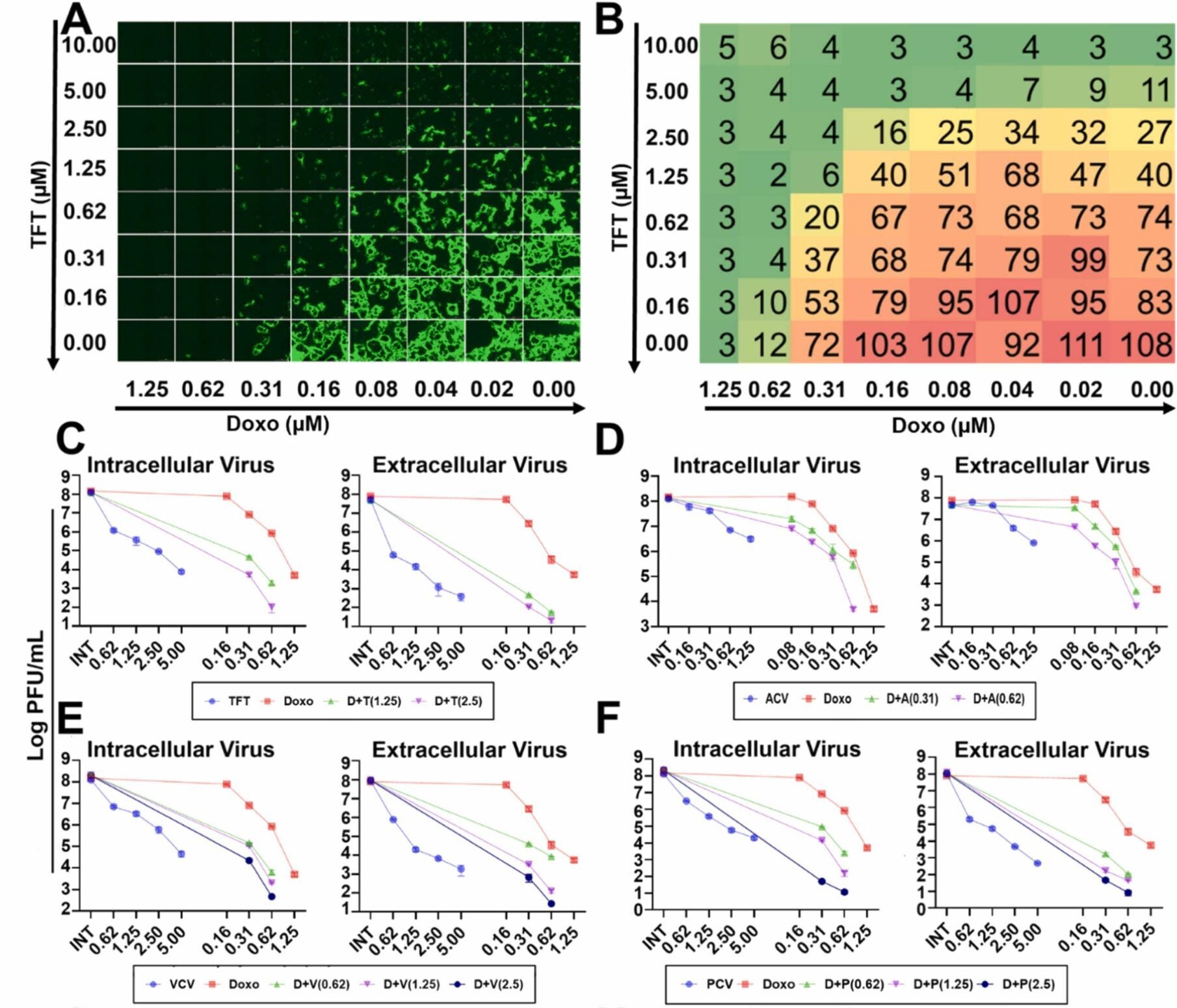
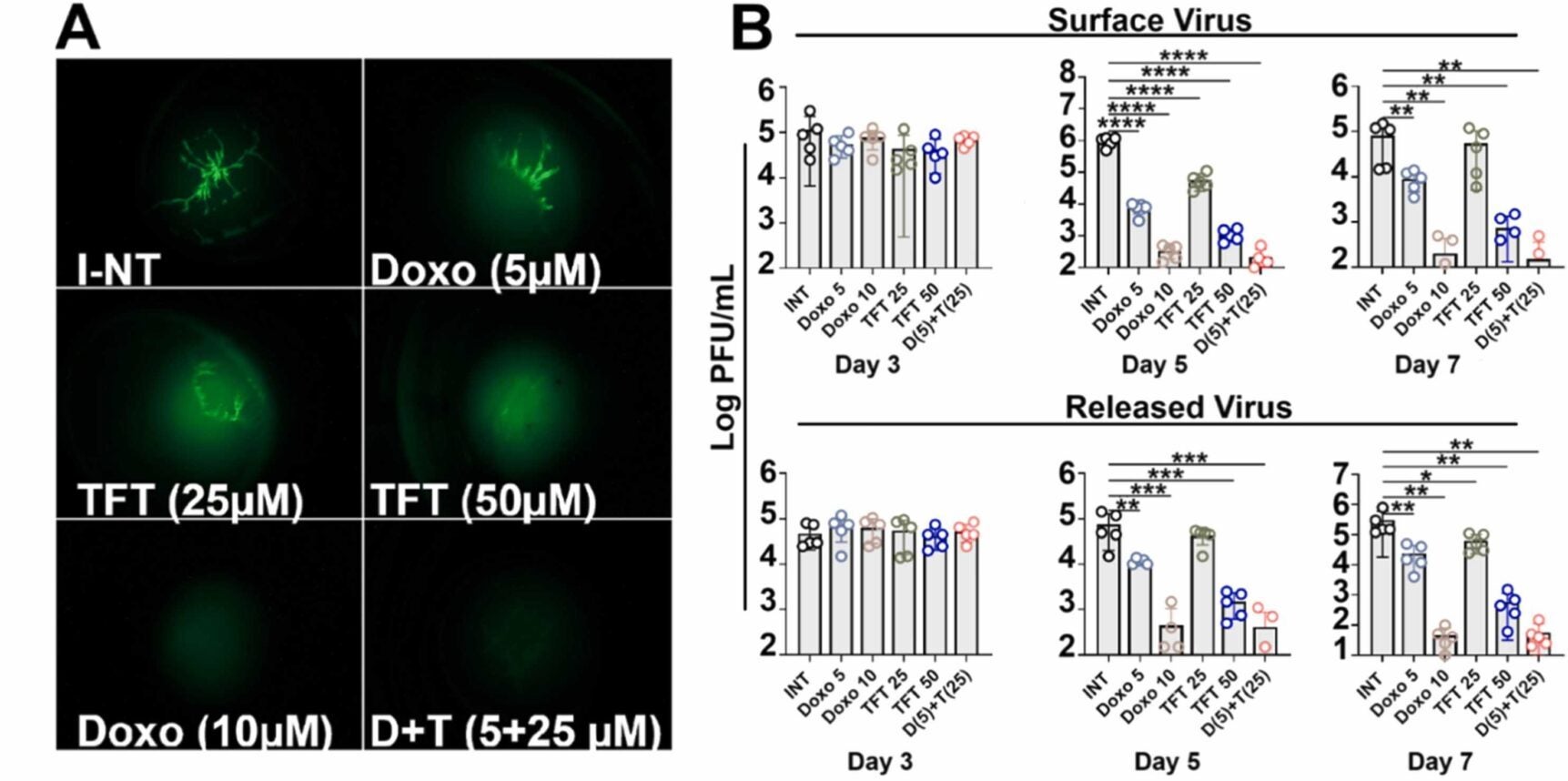
The team also used mouse models to see how the drug performs in a living system. These experiments showed that doxorubicin could suppress infection and reduce disease severity. The findings suggest the drug works not only in isolated cells but also in more complex biological environments.
Importantly, the drug blocked strains resistant to acyclovir. This is a key result because resistant infections represent one of the most urgent clinical challenges.
The researchers also found that doxorubicin worked well alongside existing antivirals. When combined with acyclovir, the drug enhanced the overall effect. This combination allowed for lower doses of acyclovir, which can cause kidney damage when used in high amounts.
A lower dose could reduce side effects and make treatment easier for patients. It could also extend the usefulness of current antiviral drugs by pairing them with a complementary therapy.
The findings may be especially important for immunocompromised individuals. Cancer patients, transplant recipients, and others with weakened immune systems face higher risks from HSV-1. In these groups, infections can become severe and difficult to control.
Doxorubicin may offer a dual benefit in some cases. It is already used in cancer treatment, and it may also suppress viral activity. This overlap could help protect patients who are at risk of both cancer progression and viral complications.
Divya Kapoor, a coauthor and graduate student researcher at UIC, reflected on the broader impact of the work. “I enjoyed being a part of something that can really help people,” she said. “This discovery has the potential to prevent herpes-related deaths and improve patient outcomes around the world, including for cancer patients who use doxorubicin.”
The research also highlights a shift in how scientists think about antiviral therapy. Instead of focusing only on the virus, they are looking at how the virus interacts with the body. This perspective may open new paths for treating other infections as well.
If future clinical trials confirm these findings, the impact could be significant. Repurposing an FDA-approved drug means the path to patient use could be faster than developing a new medication from scratch. Doctors may soon have another option for treating infections that no longer respond to standard therapies.
For patients, this could mean more effective treatment with fewer side effects. Combining doxorubicin with existing antivirals may allow lower doses of each drug, reducing toxicity while maintaining strong antiviral activity. This is especially important for people who require long-term treatment.
For researchers, the study supports a broader shift toward host-targeted therapies. By focusing on pathways within human cells, scientists may develop treatments that are harder for viruses to evade. This approach could help address resistance not only in HSV-1 but also in other persistent infections.
The work also underscores the value of computational tools like HerpDock. These systems can uncover hidden uses for existing drugs, accelerating discovery and reducing costs. As these tools improve, they may help identify new therapies for a wide range of diseases.
Research findings are available online in the journal Drug Resistance Updates.
The original story “FDA-approved cancer drug may treat drug-resistant herpes” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post FDA-approved cancer drug may treat drug-resistant herpes appeared first on The Brighter Side of News.