For something so small, the thymus has had an oddly quiet life in medicine.
It sits behind the sternum and helps train T cells, the immune system’s frontline recognizers. In childhood, that job is essential. Later, as the thymus shrinks and gives way to fatty tissue, it has often been treated as yesterday’s organ, useful early on, then mostly spent.
Two new studies suggest that view may have missed something important.
Researchers at Mass General Brigham report that adults with healthier-looking thymuses on CT scans were more likely to live longer and less likely to die from heart disease or develop lung cancer. In a separate study, cancer patients with better thymic health also tended to do better on immunotherapy, one of modern oncology’s most important treatments. Both papers were published in Nature, and together they make a case that the adult thymus may still be doing more work than many doctors assumed.
“The thymus has been overlooked for decades and may be a missing piece in explaining why people age differently, and why cancer treatments fail in some patients,” said Hugo Aerts, PhD, corresponding author on the papers and director of the Artificial Intelligence in Medicine Program at Mass General Brigham.
That line lands because the thymus has long been easy to dismiss. It gets smaller with age. It is not part of routine adult checkups. And compared with organs that fail loudly, the thymus has mostly faded into the background.
These studies bring it back into the frame.
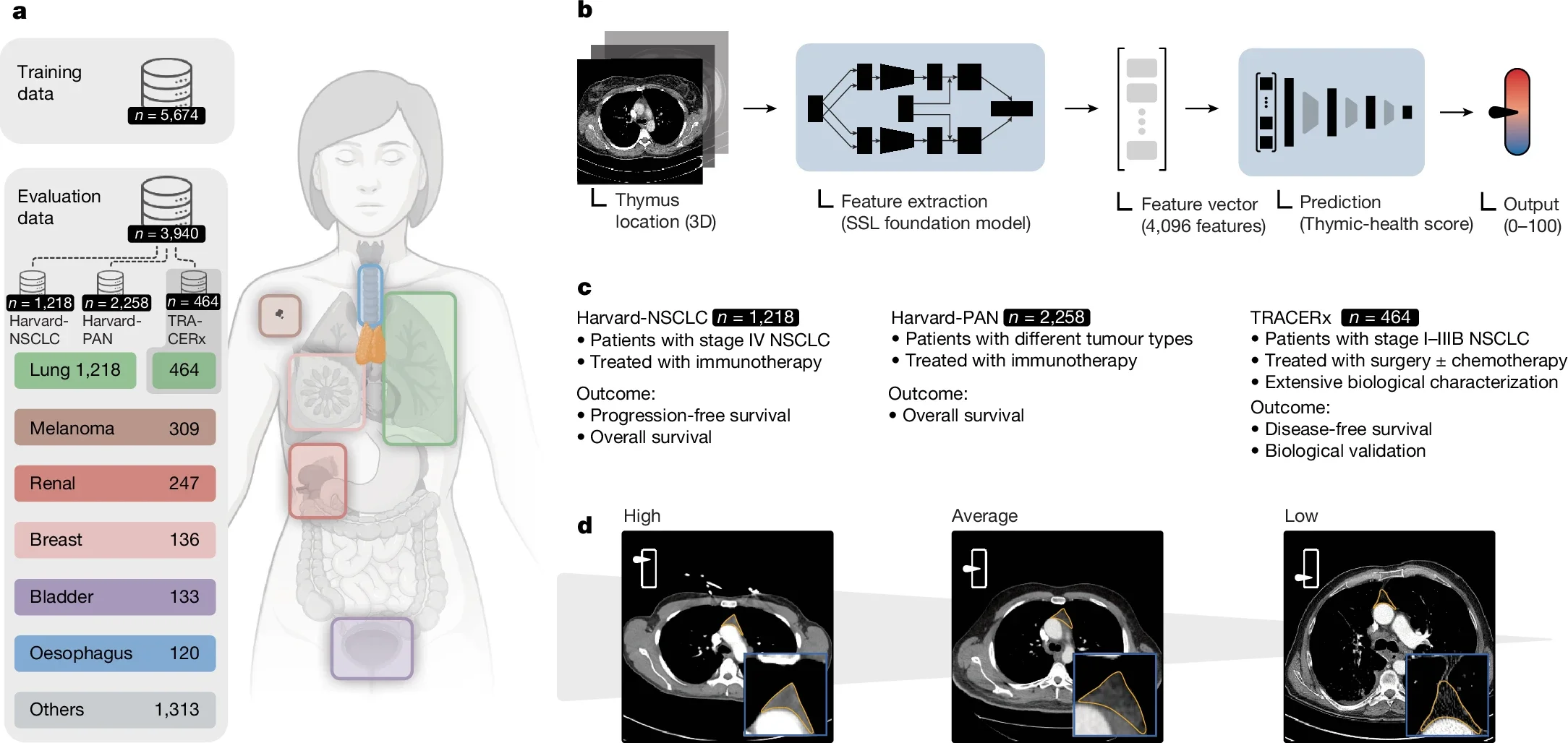
In the first study, the researchers analyzed CT scans from 27,612 adults drawn from two large groups: 25,031 people in the National Lung Screening Trial and 2,581 in the Framingham Heart Study. They used a deep learning system, developed on a separate dataset of 5,674 scans, to estimate “thymic health” based on the organ’s radiographic appearance.
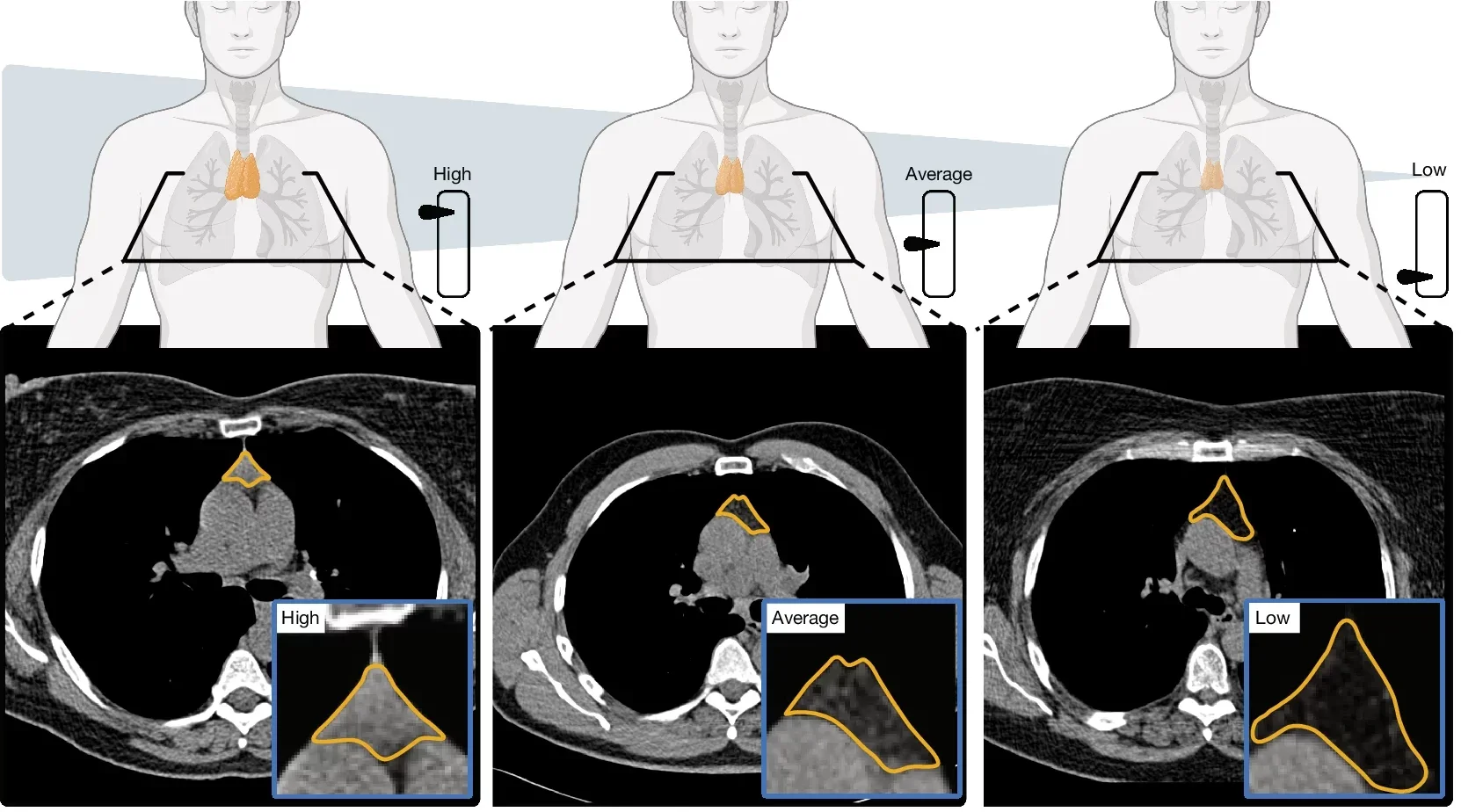
That score was then sorted into three broad groups: low, average, and high.
The gap between those groups was not subtle. In the lung screening trial, people with high thymic health had a 13.4% Kaplan-Meier mortality estimate at 12 years, compared with 25.5% for people with low thymic health. The hazard ratio was 0.49, which amounts to about a 50% lower risk of death.
Heart disease stood out even more sharply. Participants with high thymic health had a 2.9% cardiovascular-specific mortality estimate at 12 years, versus 7.5% for those with low thymic health. The hazard ratio there was 0.37. Lung cancer risk also moved in the same direction. People with high thymic health were less likely to develop lung cancer, with a hazard ratio of 0.64, and less likely to die from it, with a hazard ratio of 0.52.
The pattern held up after adjustments for age, smoking, and other health factors. In the Framingham cohort, the direction of the findings looked similar, though the link with overall survival did not remain statistically significant after thorough multivariable adjustment in that smaller group.
That matters. It keeps the story grounded.
The point is not that the thymus suddenly explains everything. It is that an organ many adults never think about kept showing up beside major health outcomes.
The second study asked a different question: not who stays healthier over time, but who responds better when cancer treatment depends on the immune system.
Here the researchers looked at 3,476 real-world patients treated with immune checkpoint inhibitors. That included 1,218 patients with non-small cell lung cancer in the Harvard-NSCLC cohort and 2,258 more in the Harvard-PAN group, which included melanoma, renal, breast, bladder, oesophageal, and other cancers.
Among patients with non-small cell lung cancer, those with high thymic health had a lower risk of progression or death after immunotherapy than those with low thymic health, with a hazard ratio of 0.65. They also had a lower risk of death overall, with a hazard ratio of 0.56. The effect appeared stronger in first-line treatment settings.
The researchers also compared thymic health with two familiar cancer biomarkers, PD-L1 and tumor mutation burden. In their analysis, thymic health showed effect sizes similar to those markers for progression-free and overall survival in lung cancer.
That is notable because thymic health is measuring something different.
PD-L1 and tumor mutation burden are tumor-centered. Thymic health, at least in this work, is a host-centered readout. It reflects the state of the patient’s immune system, not just the biology of the tumor.
That distinction gives the paper some of its force. Cancer care has spent years chasing better biomarkers inside tumors. These findings suggest part of the answer may also sit in the patient.
The authors are careful here. They describe thymic health as an imaging-based proxy for thymic functionality, not a direct measure of it.
Still, they do offer biological clues.
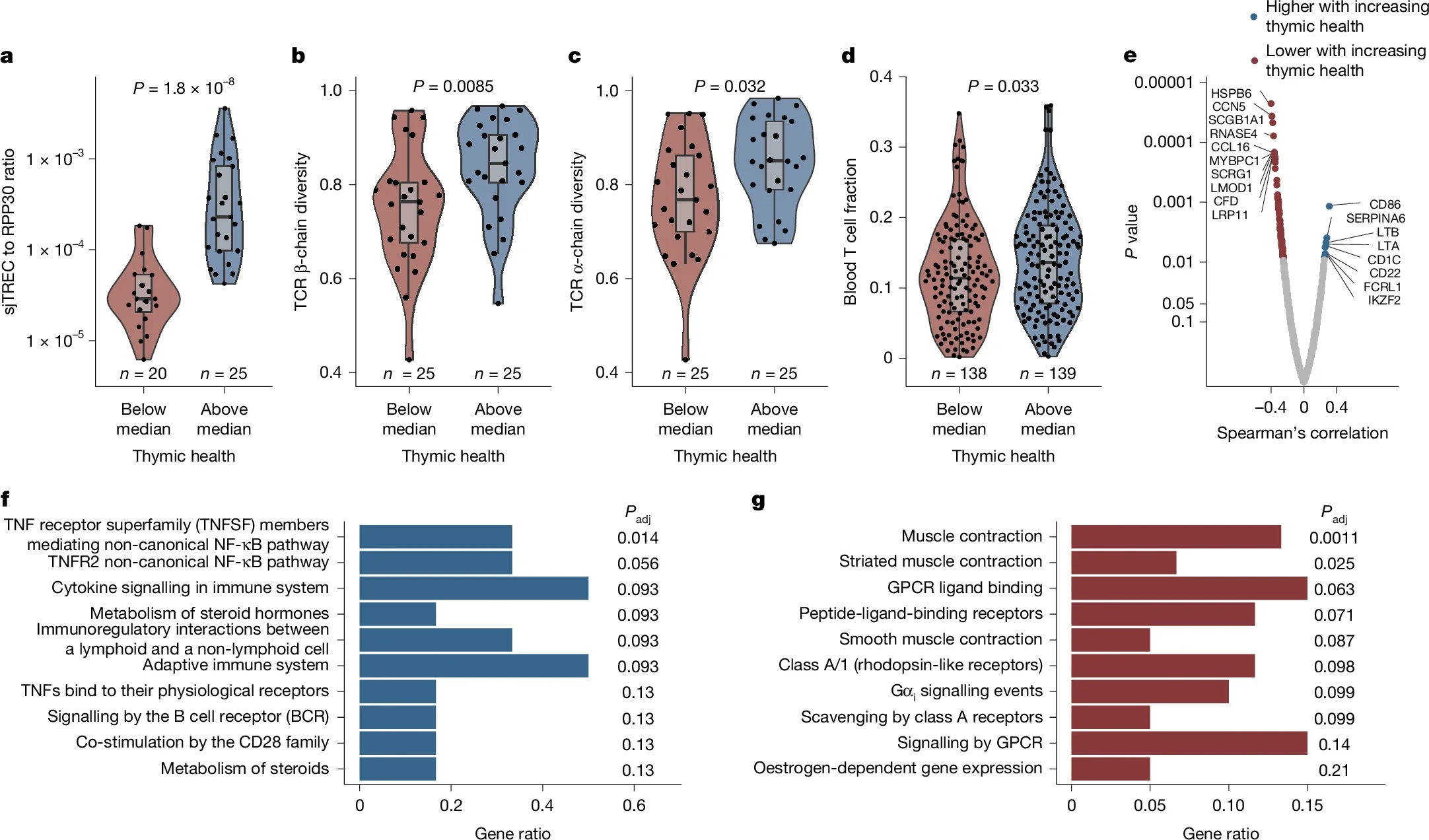
In an independent cohort from the TRACERx lung cancer study, involving 464 treatment-naive patients, higher thymic health was linked to higher levels of signal joint T cell receptor excision circles, or sjTRECs, a marker of thymic output. Patients with higher thymic health also had greater T cell receptor diversity in blood and signs of stronger tumor-specific immune activity.
That helps connect the scans to something more than appearance. The thymus is where T cells mature. If healthier thymic tissue means more sustained T cell production or diversity in adulthood, it becomes easier to see why the organ might matter in aging and cancer.
The studies also tied poorer thymic health to smoking, higher body mass index, and chronic inflammation. In the Framingham data, people with persistently elevated C-reactive protein over 5 to 10 years had substantially lower thymic health. Several inflammatory proteins, including IL-6 and IL-18, showed negative associations too.
So the thymus does not sit apart from the rest of the body’s wear and tear. It seems to move with it.
The findings are strong enough to shift the conversation, but not strong enough to settle it.
These were observational studies. They cannot prove that poor thymic health causes shorter life, more cancer, or weaker treatment response. It may be that thymic decline directly weakens immune resilience. It may also be that disease processes, inflammation, or other forms of physiological stress damage the thymus along the way.
The researchers also note several limits. The study populations were predominantly white. The thymic health model used population-specific thresholds, so there is no universal cutoff yet. The imaging approach is not ready for routine clinical use. In the immunotherapy paper, there was no matched non-immunotherapy comparison group, which limits how directly thymic health can be framed as a predictor of treatment benefit.
Those cautions are not footnotes. They are central to reading the work honestly.
Even with those limits, the studies open a useful door.
If later research confirms these results, thymic health could become a new way to estimate disease risk, understand immune aging, and help guide cancer treatment decisions. Because the thymus is visible on routine chest CT scans, the information might one day come without extra scans, extra procedures, or extra burden on patients.
The work also points to something broader. The thymus was once treated as an organ that mattered mainly in childhood. These papers suggest it may still help shape adult health, and that its decline may track with smoking, metabolic strain, and chronic inflammation. That does not prove lifestyle changes can restore thymic function. The authors did not test that. But it does hint that the organ may be part of the long, messy story of why some people age harder than others.
For now, the thymus remains mostly absent from adult medicine.
Both research findings are available online the journal Nature and Nature.
The original story “Previously irrelevant organ could be key to longer life and better cancer outcomes” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Previously irrelevant organ could be key to longer life and better cancer outcomes appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.