A bone marrow transplant can buy time for people with aggressive B-cell cancers. Too often, it does not buy enough.
For patients whose disease returns after an allogeneic stem cell transplant, the options are limited and the odds can be brutal. Standard donor lymphocyte infusions have long struggled to deliver strong responses without raising the risk of graft-versus-host disease. CAR T-cell therapy offered a more precise way to redirect immune cells against cancer, but even that has had a stubborn weak point: the engineered cells do not always take hold, multiply, and last.
A new first-in-human trial, conducted by researchers at the Leibniz Institute for Immunotherapy, suggests one way around that problem may be to start with a much narrower, more carefully built cell product.
Instead of using the mixed population found in conventional CAR T-cell manufacturing, researchers tested a donor-derived CD19 CAR T-cell therapy made from a highly enriched group of stem cell memory T cells, known as TSCM cells. These cells are prized for traits cancer immunologists want most, including long-term survival, strong proliferative capacity, and the ability to generate fresh waves of tumor-fighting descendants over time.
That difference in cell makeup appeared to matter. In the phase 1 trial, patients treated with the TSCM-enriched product showed stronger expansion and persistence of CAR T cells than those given standard donor-derived CAR T cells, even though the TSCM group received lower doses and no lymphodepleting chemotherapy before infusion.

“Today’s CAR T-cell products are heterogeneous, and that variability is reflected in the range of clinical responses and toxicity profiles we see in patients,” said Gabriele Inchingolo, a PhD student in Luca Gattinoni’s team and a shared first author of the study. “To address this, we developed a highly homogeneous CD8+ CAR T-cell product selectively enriched for TSCM cells and compared its performance to conventional CAR T cells.”
The trial, registered as NCT01087294 and led by James N. Kochenderfer and colleagues at the U.S. National Institutes of Health, enrolled patients with relapsed or refractory CD19-positive B-cell malignancies after allogeneic hematopoietic stem cell transplantation. This is a particularly difficult group to treat, with few good salvage options.
Patients were treated in two sequential cohorts. One received conventional donor-derived CD19 CAR T cells. The other received the TSCM-enriched product. Neither group received chemotherapy-based lymphodepletion beforehand, giving the investigators a cleaner look at what the infused cells could do on their own.
The contrast began in the manufacturing process. Standard CAR T cells came from peripheral blood mononuclear cells activated under conditions similar to those used for axicabtagene ciloleucel. The TSCM product, by comparison, was built from enriched naive CD8-positive T cells grown under conditions meant to preserve stem-like features and limit terminal maturation.
The result was a strikingly different infusion product. The TSCM therapy was almost entirely CD8-positive and highly enriched in stem cell memory cells, with a median TSCM fraction of 78.4%. The standard product was far more mixed, with a median TSCM fraction of 8.4% and larger proportions of effector memory and terminal effector cells.
That homogeneity became one of the central themes of the trial.
Despite receiving fewer cells overall, patients in the TSCM cohort had an overall response rate similar to the standard group, 55% versus 45%. Event-free survival did not differ significantly between cohorts, with a median of 4.9 months in the TSCM group and 3.3 months in the standard group. But the low-dose results drew the most attention.
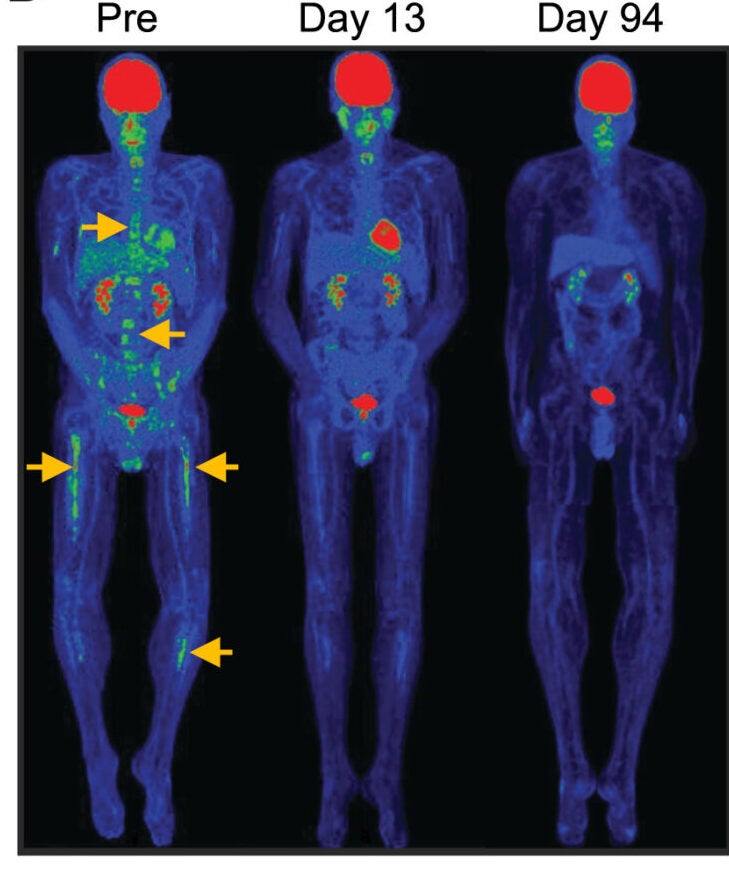
At doses below 3 × 10^8 CAR T cells, complete responses occurred in 5 of 11 TSCM-treated patients, or 45%, compared with 1 of 10 patients, or 10%, in the standard cohort.
“Seeing patients achieve complete responses at doses as low as 250,000 cells per kilogram, without chemotherapy preconditioning, validates years of preclinical work and opens a new chapter in CAR T-cell design,” said Gattinoni, head of the Functional Immune Cell Modulation division and first author of the study.
CAR T-cell therapy often lives or dies by what happens after infusion. If the cells do not expand in the bloodstream and persist, the cancer can return quickly. The new analysis found that the TSCM product behaved differently from the start.
Standard CAR T cells tended to peak within the first week. The TSCM cells expanded later, with peak levels in the second week. Yet on a per-cell basis, the TSCM therapy reached much higher blood levels. Median peak blood counts reached 30.4 CAR T cells per microliter in the TSCM cohort, compared with 6.7 in the standard cohort. The area under the curve through day 36 was also markedly higher, 185.8 versus 27.5.
Flow cytometry backed up the qPCR findings, showing a higher median expansion in the TSCM group, 7.3% versus 1%.
That stronger expansion did not seem to come from a more favorable baseline immune environment. The researchers checked baseline CD3-positive T-cell and natural killer cell counts and found no significant differences between cohorts. The cleaner explanation was that the infused TSCM cells themselves had better biological fitness.
James Kochenderfer, senior investigator at the National Cancer Institute and co-corresponding author, said that finding fits a broader pattern across the field. “The TSCM platform yielded higher CAR T-cell levels on a per cell basis, and across many CAR T-cell studies, high blood CAR T-cell levels have been one of the strongest predictors of clinical efficacy.”
Just as notable, the stronger expansion did not come with the same degree of toxicity.
Cytokine release syndrome, or CRS, is one of the best-known dangers of CAR T-cell therapy. In the standard cohort, 28.6% of patients experienced grade 3 or 4 CRS. In the TSCM cohort, no grade 4 CRS events occurred, and only one patient developed grade 3 CRS. No graft-versus-host disease was seen in the TSCM-treated group.
Kochenderfer, who has taken part in many CAR trials, noted that the inflammatory picture looked different from what he usually sees. “We also observed less cytokine-release syndrome on this study compared to most other CAR clinical trials that I have participated in.”
The timing of fever matched the altered expansion pattern. Fever appeared in the first week for the standard group and in the second week for the TSCM group. But despite similar temperatures, the standard cohort showed higher C-reactive protein levels and stronger IL-6 signals. The TSCM cohort instead showed a later interferon-gamma peak.
That matters because severe CRS is often linked to explosive inflammatory cascades. Here, the TSCM product appeared to preserve expansion while softening the toxic inflammatory profile.
The trial also gave researchers a rare view of what these cell products do over time inside patients.
Using multidimensional flow cytometry, bioinformatics, and longitudinal tracking of retroviral integration sites, the team followed the fate of infused cells after treatment. The picture that emerged was not simply that TSCM cells lasted longer. It was that they followed a different biological program.
Standard CAR T-cell clonotypes tended to expand rapidly, then contract or stabilize. The long-term persisting clones at 90 days mostly came from the same clones that dominated early expansion.
The TSCM product did something else. It repopulated patients in sequential waves.
Rather than emptying out a single pool of stem-like cells in one burst, the TSCM compartment kept generating new cohorts of active clones over time while preserving a reservoir of quiescent stem-like cells. By the memory phase, the TSCM group had rebuilt the TSCM compartment and replenished very early proliferating effector populations.
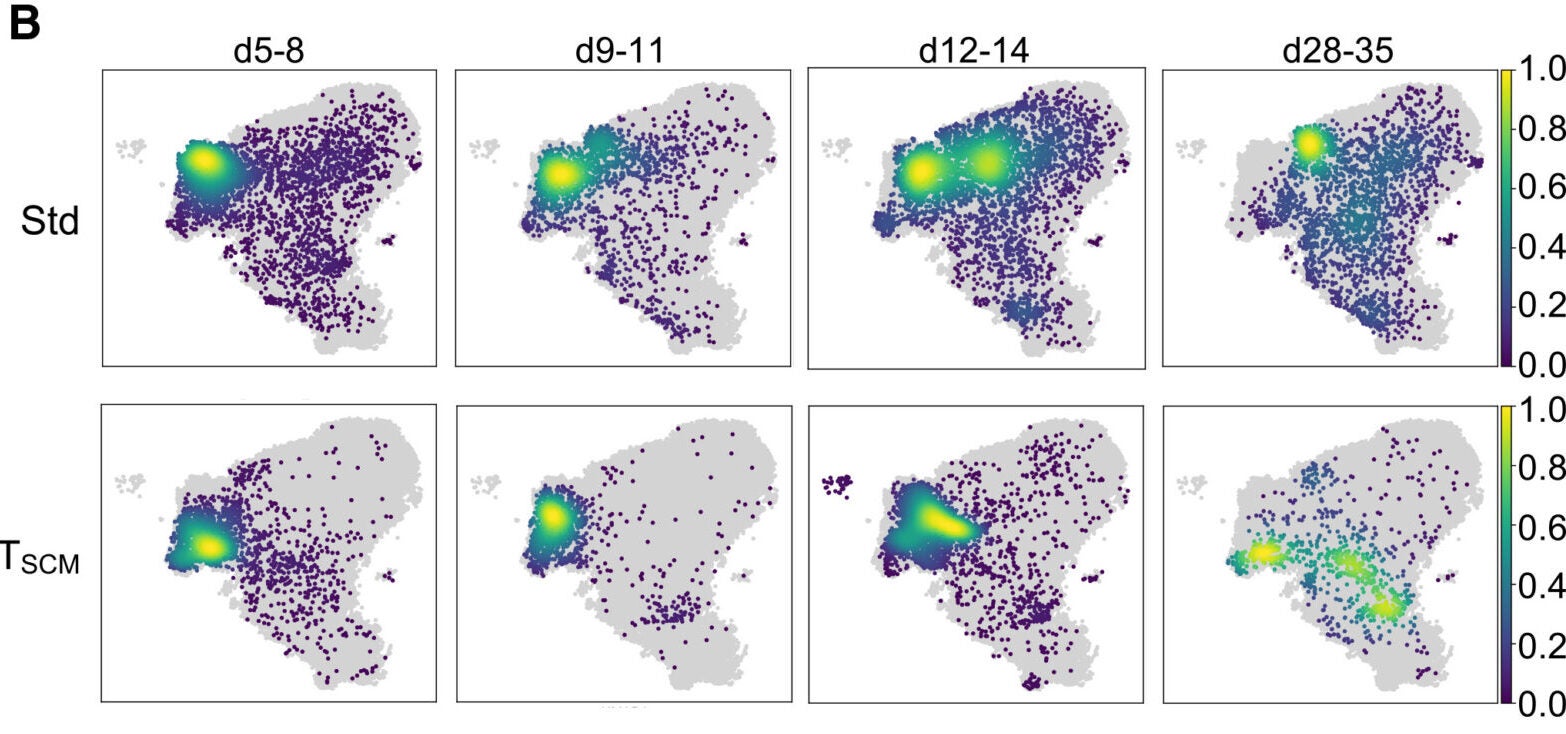
“For the first time, we are witnessing this fundamental biology play out directly in patients,” said Enrico Lugli, principal investigator in translational immunology at Humanitas Research Hospital and a coauthor of the study. He described the pattern as small, sequential waves of recruitment, with each active cohort succeeding the last while the long-term stem-like reservoir remains intact.
The clonal tracking data supported that interpretation. In TSCM-treated patients, the top persisting clones at day 90 often came from newly expanded clones that were not central to the first expansion wave. The authors compared that behavior to hematopoietic stem cell reconstitution, which preserves long-term function at the population level through clonal succession.
It is an important distinction. Much of the older thinking around stemness focused on whether individual cells self-renew through asymmetric division. This work points toward a population-level solution, where stemness survives because different clones take turns carrying the response.
Not every patient benefited, and those failures helped define the limits of the approach.
In one patient with progressive disease, CAR TSCM cells expanded strongly and remained functional enough to cause B-cell aplasia, yet the leukemia kept advancing. The key problem was low CD19 density on the tumor. Before treatment, leukemic blasts already expressed dim CD19, and after treatment, the CD19-negative fraction was enriched, pointing to antigen escape or inadequate recognition of weakly expressed targets.
Another patient failed to show CAR T-cell expansion despite receiving a product that was more than 80% TSCM. Serum analysis revealed unusually high levels of IL-10 in the first days after infusion. The authors say that, although this was a single case, it fits with IL-10’s established immunosuppressive role.
The team also found evidence that antibodies directed against the mouse-derived FMC63 component of the CAR construct may have blocked repeat therapy in some patients. Anti-CAR antibodies were detected in 35% of patients across the cohorts, and the levels tracked with the presence of B cells at infusion. In patients given a second CAR T-cell infusion, humoral responses rose further, suggesting a prime-boost effect. In some cases, a second infusion no longer produced the B-cell depletion seen after the first.
Together, those results point toward tumor and host barriers, not weak T cells, as the main explanation when the TSCM product fell short.
The authors are careful not to overstate the clinical results. This was a small, early-phase study with disease heterogeneity, no randomization, and exploratory comparisons between cohorts. The absence of lymphodepletion may also have limited performance by allowing anti-CAR immune responses or host rejection to develop more easily. And because the TSCM product was intentionally CD8-enriched, the lack of CD4-positive CAR T cells may have affected longer-term immune control, particularly against CD19-negative variants.
This study points to a practical shift in how CAR T-cell therapy might be built: less as a mixed cell collection, more as a defined biological product. A highly enriched stem cell memory population may give clinicians a better chance of getting reliable engraftment, stronger expansion, and longer persistence, even at lower doses.
That could matter well beyond this transplant setting. The authors argue that the same biological logic may apply to autologous CAR T-cell therapy and perhaps to solid tumors, where poor persistence and exhaustion have repeatedly limited success. Future studies will need to test the approach in larger, randomized trials, likely with lymphodepleting chemotherapy, humanized CAR constructs, and possibly added CD4-positive cells.
For now, the work offers something both modest and meaningful: evidence that the quality and identity of CAR T cells can change what happens in patients, not just in the lab.
Research findings are available online in the journal Cell.
The original story “Study finds low-dose CAR T therapy can drive complete cancer remission” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Study finds low-dose CAR T therapy can drive complete cancer remission appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.