The Iñupiat people of Alaska have long said that bowhead whales live two human lifetimes. That oral knowledge turned out to be conservative. Scientific measurements using multiple methods have confirmed that these animals can survive for more than 200 years, making them the longest-lived mammals on Earth. An 80-tonne animal, moving through Arctic water for two centuries, managing to avoid cancer across billions of cell divisions, presented biologists with one of the more compelling puzzles in aging science.
A study published in Nature by researchers at the University of Rochester has now identified a protein that may explain a significant part of how bowhead whales pull this off, and what it could mean for human health is already being investigated.
The protein is called CIRBP. Bowhead whales produce roughly 100 times more of it than humans do. And when researchers introduced the whale version of this protein into human cells and fruit flies, the results were striking enough to suggest that human DNA repair capacity, long assumed to be near its biological ceiling, may have substantial room to improve.
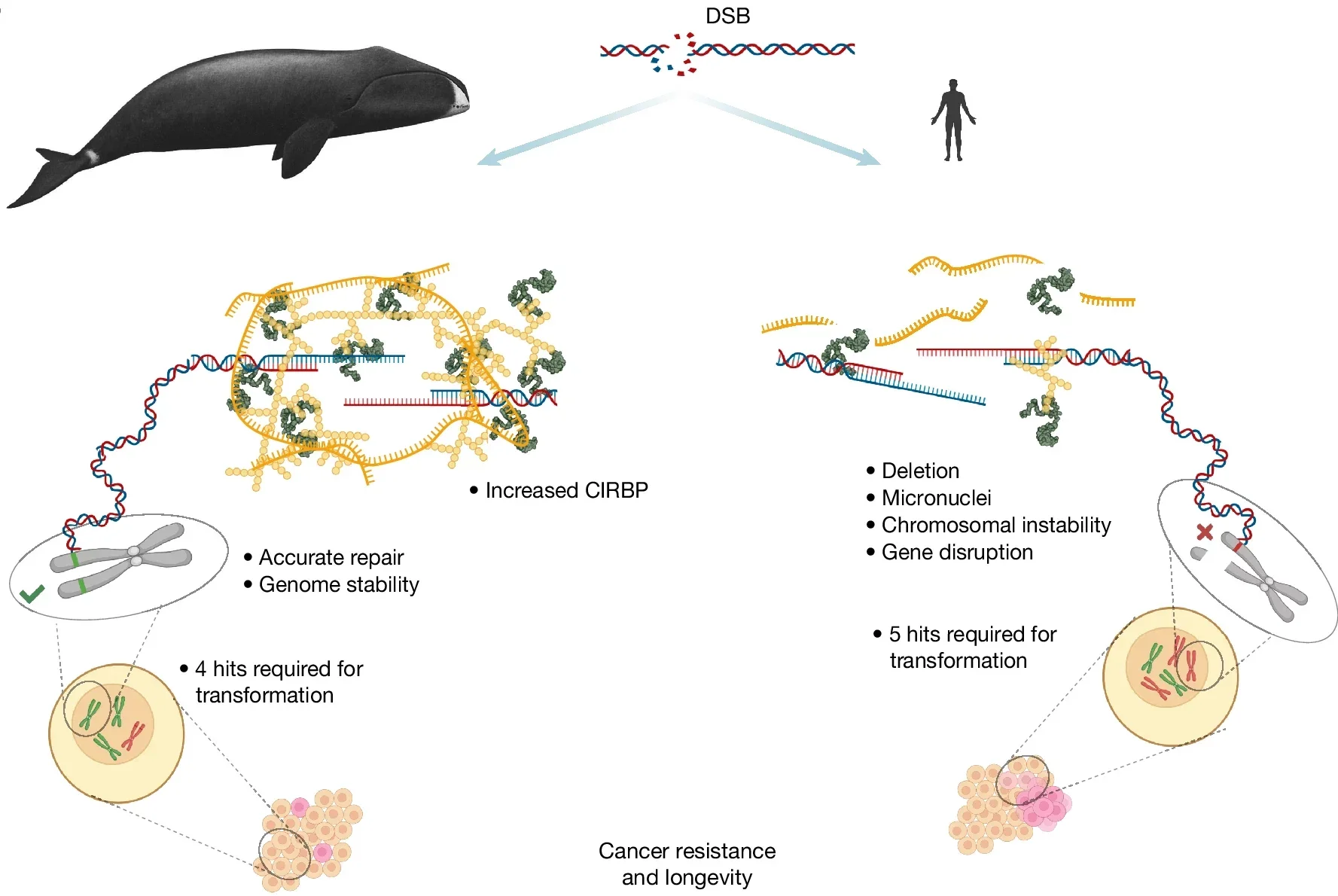
Before getting to the protein, it helps to understand why the bowhead whale’s survival record is scientifically puzzling in the first place.
Cancer arises when DNA mutations accumulate and eventually allow a cell to escape normal growth controls. Larger animals have more cells, and longer-lived animals have more time for those mutations to accumulate. By that logic, a creature the size and age of a bowhead whale should be riddled with cancer. Yet bowhead whales rarely develop it.
This apparent contradiction has a name in biology: Peto’s paradox, after the epidemiologist who first noted that cancer rates do not scale with body size across species. Elephants face the same puzzle. Researchers studying elephants found that they carry extra copies of the tumor suppressor gene TP53 and respond to DNA damage with a heightened cell-death response, eliminating damaged cells before they can become cancerous.
Bowhead whales use a different strategy entirely.
The Rochester team, led by biologists Vera Gorbunova and Andrei Seluanov, found that whale cells are not more likely to eliminate damaged cells through apoptosis than human cells. They do not show elevated p53 activity. They also, counterintuitively, require fewer genetic mutations to become cancerous in laboratory transformation assays, not more.
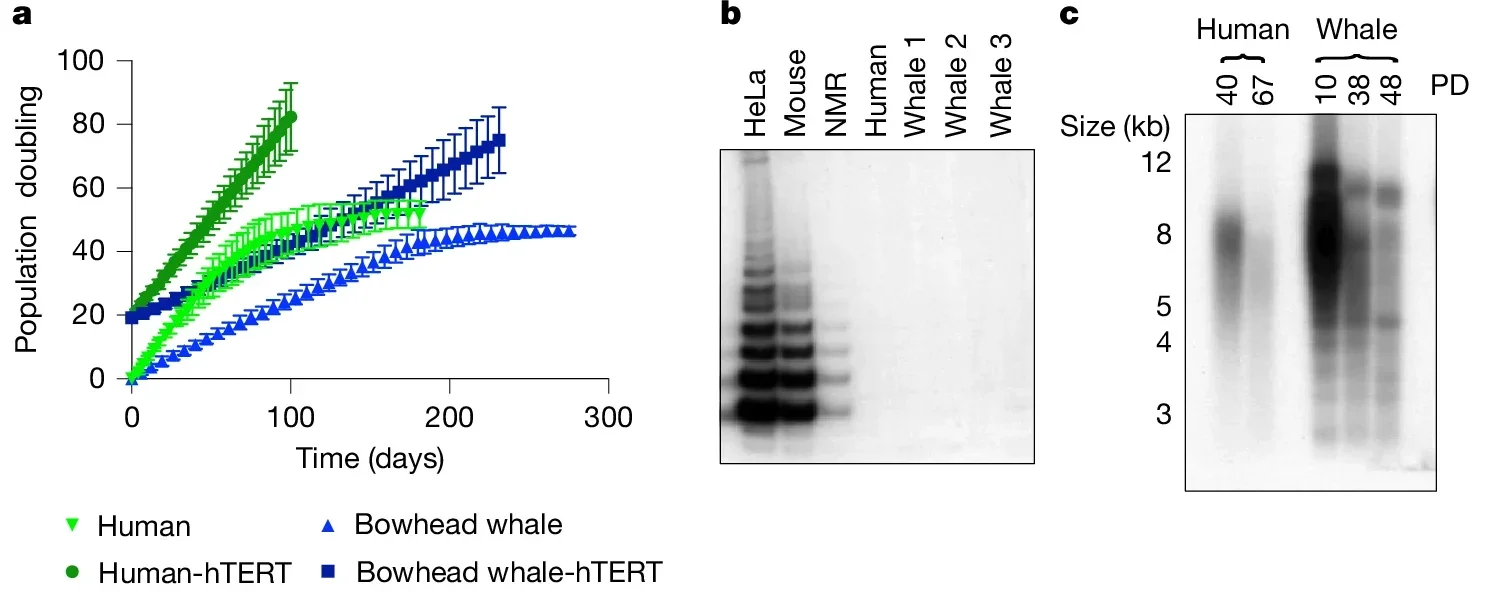
What they do instead is repair their DNA with unusual accuracy. Whale cells accumulate fewer mutations in the first place, suffer fewer structural rearrangements in their chromosomes, and fix the most dangerous type of DNA damage, double-strand breaks, more efficiently and with greater precision than cells from humans, mice, or cows tested alongside them.
“What we’re finding is that these kinds of repairs are very important for long life,” said Gorbunova.
When the team searched for what might be driving this enhanced repair capacity, they did not find an unusual suite of repair enzymes. Proteins commonly associated with DNA repair in humans were actually less abundant in whale cells. What stood out was CIRBP.
CIRBP stands for cold-inducible RNA-binding protein. It is expressed in many mammals, including humans, but at low levels. In bowhead whales, its abundance was far beyond anything observed in other species tested, present in cells and tissues at concentrations roughly 100 times those found in humans. The protein was also detectable, though at much lower levels, in humpback whales and dolphins, suggesting a pattern across cetaceans rather than a quirk of a single species.
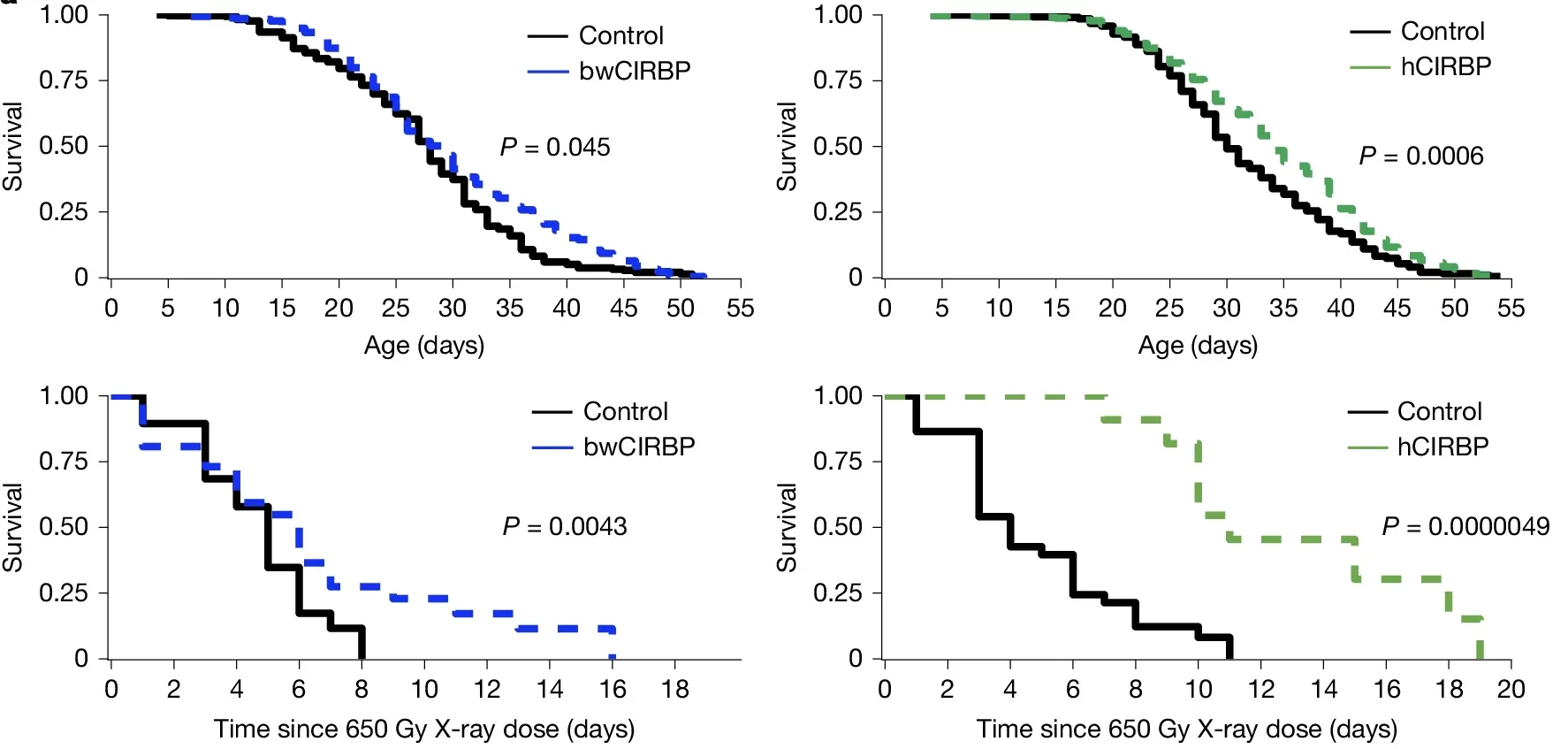
The protein’s name offers a clue to one of its known triggers. CIRBP levels rise when cells experience cold. Bowhead whales spend their entire lives in Arctic and sub-Arctic waters, which may explain why evolution pushed their CIRBP expression so high. The researchers confirmed that cooling human cells to 33 degrees Celsius, rather than the normal 37, increased both CIRBP protein levels and the efficiency of DNA repair in those cells.
That finding opens a practical question: could brief cold exposure in humans, such as cold-water swimming or cold showers, raise CIRBP levels enough to matter? Gorbunova’s team is now investigating this, though they acknowledge that pharmacological approaches are also on the table for people less enthusiastic about cold immersion.
The mechanistic experiments were direct. When the researchers introduced the bowhead whale version of CIRBP into human cells, the proportion of double-strand DNA breaks that were successfully repaired roughly doubled. The repair was also cleaner: cells made fewer deletion errors at the break sites, a form of damage that can contribute to cancer-driving mutations.
When CIRBP was silenced in whale cells using RNA interference, the efficiency of both major repair pathways dropped sharply, and error rates increased. The protein was not a passive bystander; it was actively contributing to the whale’s DNA maintenance capacity.
Laboratory experiments with purified proteins showed how CIRBP works at the molecular level. It binds to broken DNA ends and shields them from enzymes that would otherwise chew away the exposed strands, degradation that makes accurate repair much harder. It also helps recruit the molecular machinery responsible for joining broken ends back together.
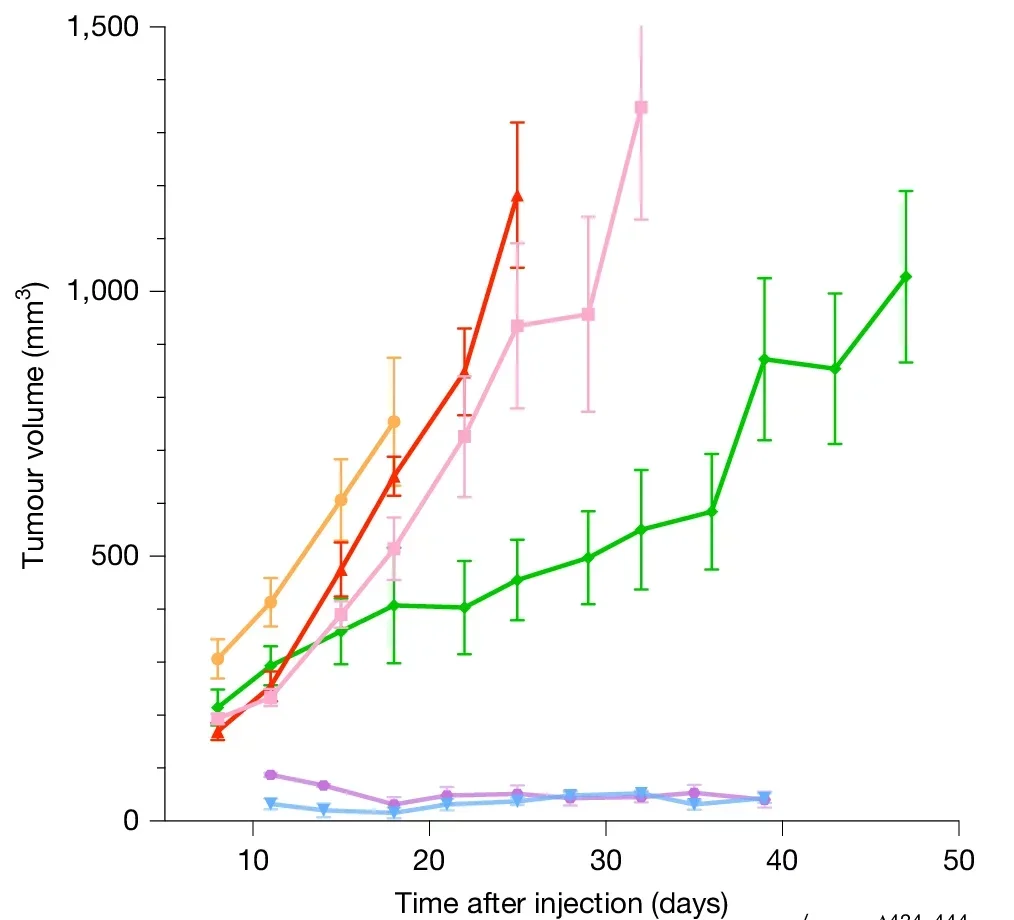
Overexpressing CIRBP in human cells that had been engineered to undergo cancerous transformation slowed that transformation and reduced chromosomal instability. In mouse experiments, tumors grew more slowly in cells with elevated CIRBP.
Among the most compelling results in the study were the experiments in fruit flies. When the researchers expressed either the human or the bowhead whale version of CIRBP in flies, the insects lived significantly longer than controls. They also survived exposure to ionizing radiation at higher rates, consistent with greater resilience against DNA damage in living organisms rather than just in cell cultures.
The extension of lifespan in a whole animal, even one as different from a whale as a fruit fly, suggests that CIRBP’s effects on genome stability are not merely a laboratory artifact confined to isolated cells. They operate through biology that is conserved enough across species to produce measurable effects in vivo.
Experiments in mice with elevated CIRBP are now underway to determine whether the lifespan effects observed in flies extend to a mammalian system closer to humans.
The study is candid about what remains unknown. Most of the cellular experiments were conducted in fibroblasts, a type of connective tissue cell, while the majority of human cancers arise in epithelial cells lining organs. Whether the same CIRBP dynamics operate in those cell types has not yet been established.
The whale’s longevity almost certainly involves multiple mechanisms beyond CIRBP. Gorbunova’s team identified enhanced mismatch repair and higher PARP activity in whale cells as additional contributors to genome stability, and the full picture of how these systems interact over a two-century lifespan remains to be worked out.
Prof. Gabriel Balmus, who studies DNA damage and repair at the UK Dementia Research Institute at the University of Cambridge, offered a cautiously optimistic assessment: “Enhancing our cells’ ability to repair DNA could, in principle, slow the ageing and associated disease processes. Yet translating this into humans will be far from straightforward, demanding a balance between resilience and the body’s natural limits on renewal.”
The central finding challenges a long-standing assumption in biology: that human DNA repair is already operating near its maximum capacity and cannot be meaningfully improved. The bowhead whale demonstrates that this is simply not true. A mammal exists that repairs its DNA far better than we do and lives roughly four times longer as a result.
If CIRBP levels in human cells can be safely elevated, whether through cold exposure, pharmacological agents, or eventual gene-based approaches, the implications extend well beyond longevity in any abstract sense. Reduced mutation accumulation would mean lower lifetime cancer risk. More precise DNA repair could protect the genome during chemotherapy or radiation treatment. Enhanced repair capacity in transplanted organs could improve outcomes during surgery.
“People used to think we can’t improve DNA repair, that it’s already optimal, but the whale does it better than we do,” Gorbunova said.
The bowhead whale has been accumulating answers to biological questions for two centuries before anyone thought to ask them. The work of translating those answers into human medicine is now seriously underway.
Research findings are available online in the journal Nature.
The original story “Bowhead whales may hold the key to humans living up to 200 years” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Bowhead whales may hold the key to humans living up to 200 years appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.