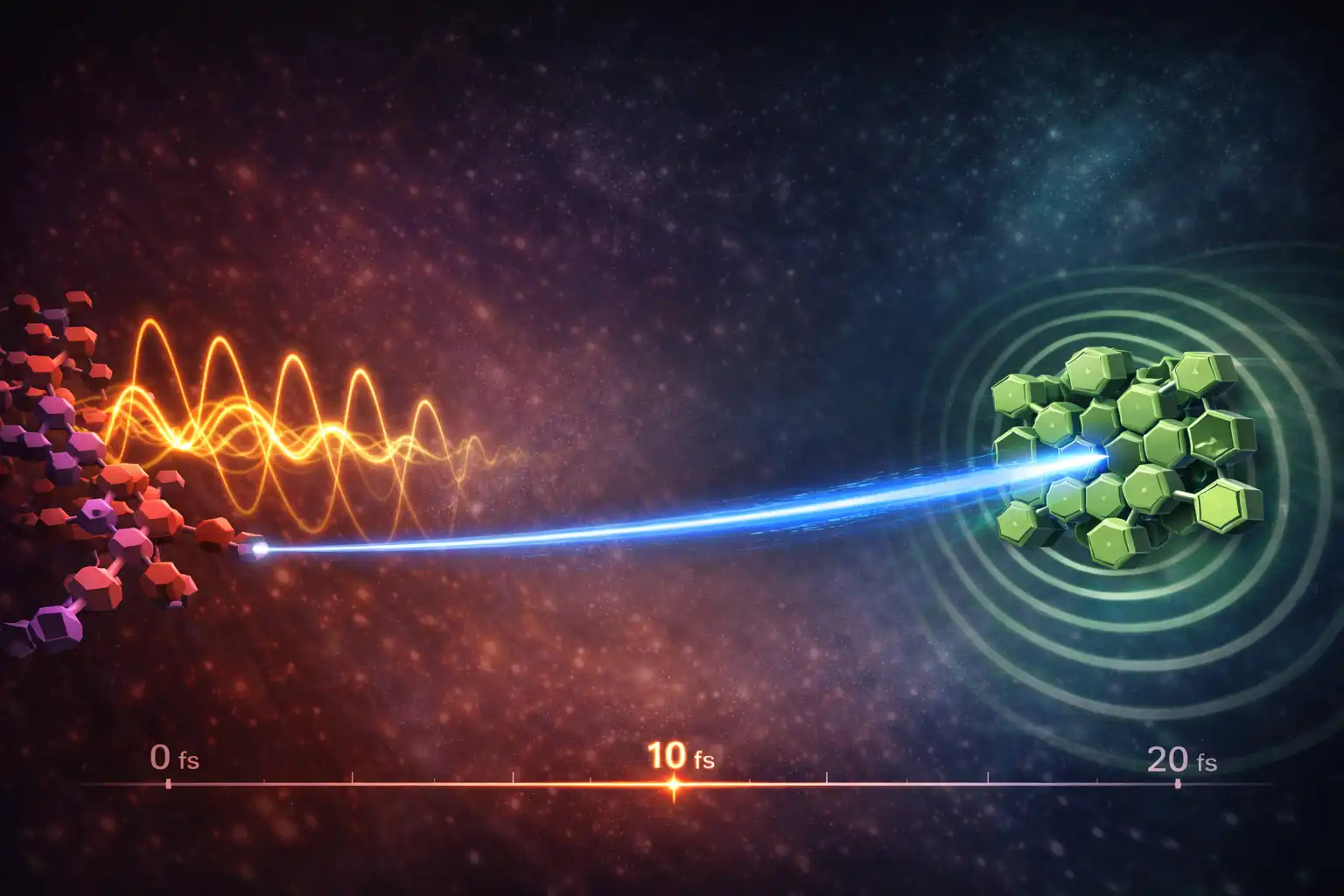
A tiny burst of motion inside a molecule may be enough to shove an electron across a solar material almost as fast as nature permits.
That is the striking result from a University of Cambridge-led study that tracked charge transfer in just 18 femtoseconds, a span so short that it unfolds within a single molecular vibration. The work challenges a long-standing assumption in solar energy research: that electrons move this quickly only when materials have a large energy gap between them or are very strongly linked.
“We deliberately designed a system that should not have transferred charge this fast,” said Dr Pratyush Ghosh, Research Fellow at St John’s College, Cambridge, and first author of the study. “By conventional design rules, this system should have been slow and that’s what makes the result so striking.”
The finding matters because charge transfer sits at the heart of devices that turn light into something useful. In organic solar cells, photocatalytic systems and photodetectors, light first creates an exciton, a tightly bound electron-hole pair. To make electricity or drive chemistry, that pair has to split.
The faster that happens, the less energy gets wasted.
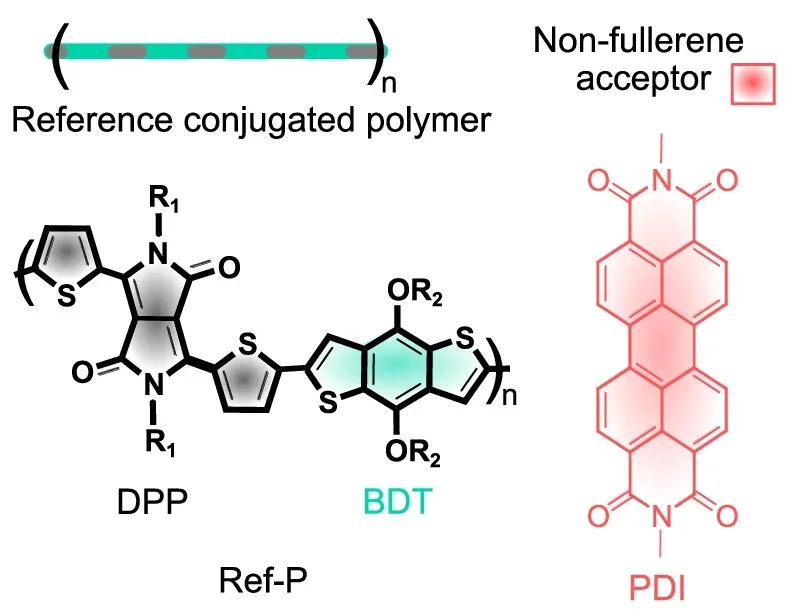
The team built a deliberately weak model junction from a polymer donor and a non-fullerene acceptor. The pair had almost no energy offset and only minimal interaction, conditions that standard thinking says should slow charge transfer dramatically.
Instead, one version of the system, called TS-P3, moved the electron across in 18.1 ± 3.1 femtoseconds, with the researchers placing a conservative upper bound of about 21.2 femtoseconds on the process. A related arrangement, TS-P2, was much slower at about 376 femtoseconds.
That contrast turned out to be central. Both systems used the same components, but their geometry differed. In TS-P3, a key vibration on the polymer backbone lined up with the donor-acceptor interface. In TS-P2, that motion was displaced from the acceptor, and the transfer slowed sharply.
“Instead of drifting randomly, the electron is launched in one coherent burst,” Ghosh said. “The vibration acts like a molecular catapult. The vibrations don’t just accompany the process, they actively drive it.”
The study, published in Nature Communications, argues that this motion is not a side effect. It is the mechanism.
Using ultrafast laser spectroscopy, the researchers found that after the polymer absorbed light, it began vibrating in specific high-frequency modes. Those vibrations mixed the system’s excited states and kicked the electron across the boundary in a directional, ballistic way rather than through slower, random diffusion.
Once the electron landed on the acceptor molecule, it triggered a new coherent vibration there, a signal the team treated as a fingerprint of just how rapid and clean the transfer was.
“That coherent vibration is a clear fingerprint of how fast and how cleanly the transfer occurs,” Ghosh said.
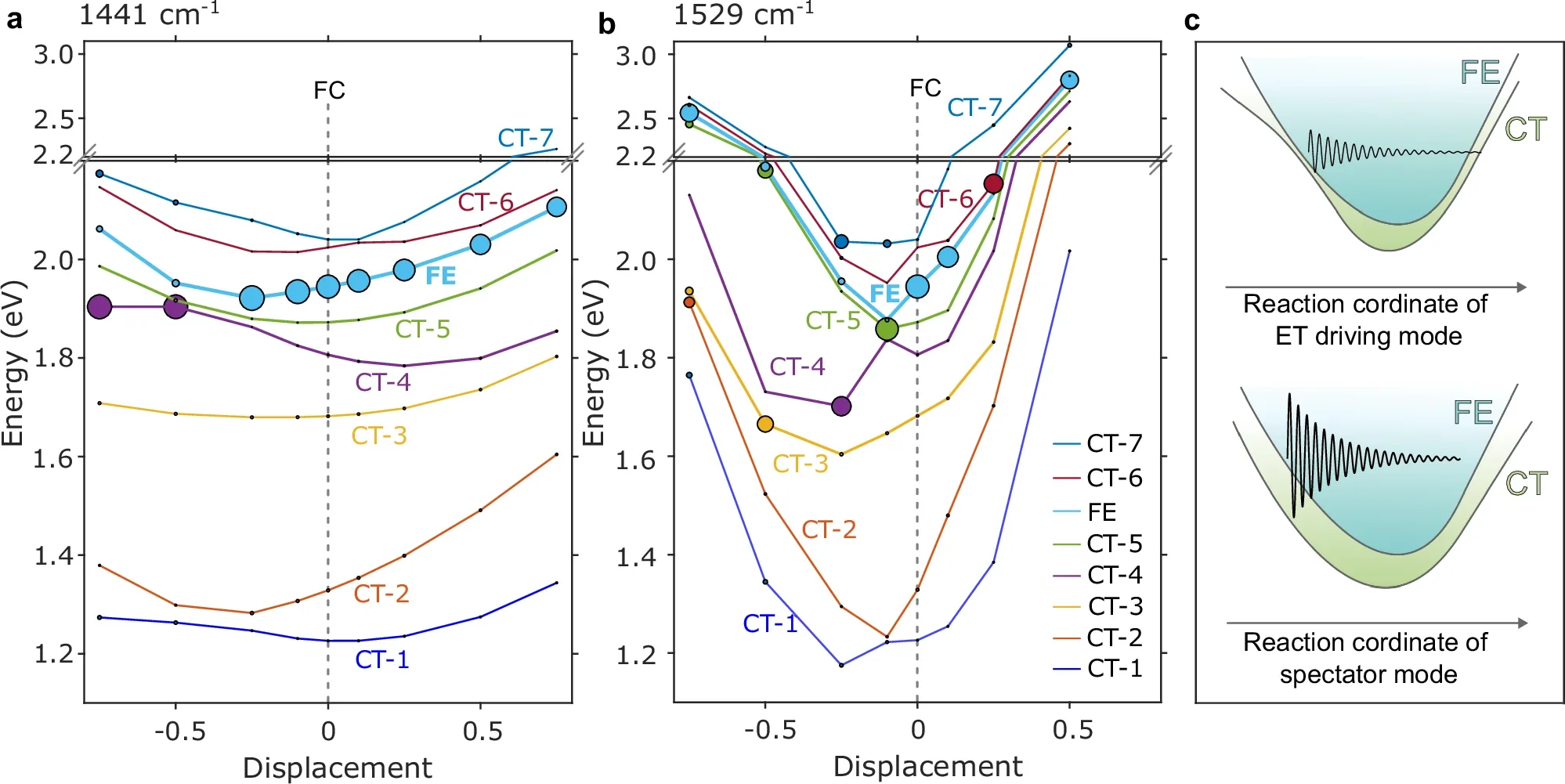
The calculations backed up the measurements. Density functional theory and quantum dynamics simulations pointed to one polymer-centered vibration, around 1529 cm⁻¹ in theory and 1512 to 1529 cm⁻¹ in experiment, as a driving mode. A different vibration, around 1441 cm⁻¹, behaved more like a spectator. It moved, but it did not strongly mix the exciton and charge-transfer states.
That distinction helped the team propose a new design rule: put the right vibration in the right place.
When a high-frequency mode is co-localized with the interface where the electron must cross, ultrafast transfer can happen even in a weakly coupled system.
For years, researchers have often assumed that fast charge transfer comes at a price. Large energy offsets can help split charges quickly, but they also limit voltage in photovoltaics or raise energy losses in photocatalysis.
This work suggests that tradeoff may not be as rigid as once thought.
“Our results show that the ultimate speed of charge separation isn’t determined only by static electronic structure,” said Dr Ghosh. “It depends on how molecules vibrate. That gives us a new design principle. In a way, this gives us a new rulebook. Instead of fighting molecular vibrations, we can learn how to use the right ones.”
Professor Akshay Rao, Professor of Physics at the Cavendish Laboratory and former St John’s College Research Associate, who was a co-author of the study, put it more simply: “Instead of trying to suppress molecular motion, we can now design materials that use it – turning vibrations from a limitation into a tool.”
The study involved researchers at the Cavendish Laboratory and the Yusuf Hamied Department of Chemistry at the University of Cambridge, including Dr Rakesh Arul, alongside collaborators in Italy, Sweden, the United States, Poland and Belgium.
The authors also drew a clear boundary around what they had done. Their system was a model heterojunction studied in solution, built to isolate the first moments of charge separation. It was not designed to optimize power-conversion efficiency in a real solar device. That means the work does not yet show a ready-made better solar cell, but it does identify a mechanism that could guide future materials design.
The immediate value of the work is conceptual. It gives researchers a new way to think about organic solar cells, photodetectors and photocatalytic materials, including systems for clean hydrogen production.
Instead of relying mainly on strong coupling or large energy offsets, designers may be able to place donor and acceptor units so that the right molecular vibrations help launch charges across an interface quickly while avoiding some of the usual energy penalties.
Research findings are available online in the journal Nature Communications.
The original story “Cambridge scientists capture electrons leaping across solar materials in 18 femtoseconds” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Cambridge scientists capture electrons leaping across solar materials in 18 femtoseconds appeared first on The Brighter Side of News.