The clinical trials told one story. When patients stopped semaglutide in controlled research settings, they regained roughly two-thirds of their lost weight within a year. When tirzepatide was discontinued in another major trial, only 17 percent of participants held on to at least 80 percent of what they had lost. Those numbers traveled fast, shaping how doctors and patients talked about what happens when the medication stops.
A large real-world analysis from Cleveland Clinic is now adding a different chapter. Among nearly 8,000 patients who discontinued injectable semaglutide or tirzepatide, average weight one year later had barely shifted. Those treated for obesity regained a mean of just 0.5 percent of body weight. Those treated for type 2 diabetes actually lost an additional 1.3 percent on average.
The gap between trial results and clinical reality, researchers say, comes down to what happens next.
The study, published in the journal Diabetes Obesity and Metabolism, was led by Hamlet Gasoyan, a researcher with Cleveland Clinic’s Center for Value-Based Care Research. His team tracked 7,938 adult patients in Ohio and Florida who had started injectable semaglutide or tirzepatide and then stopped the medication between three and twelve months after beginning treatment.
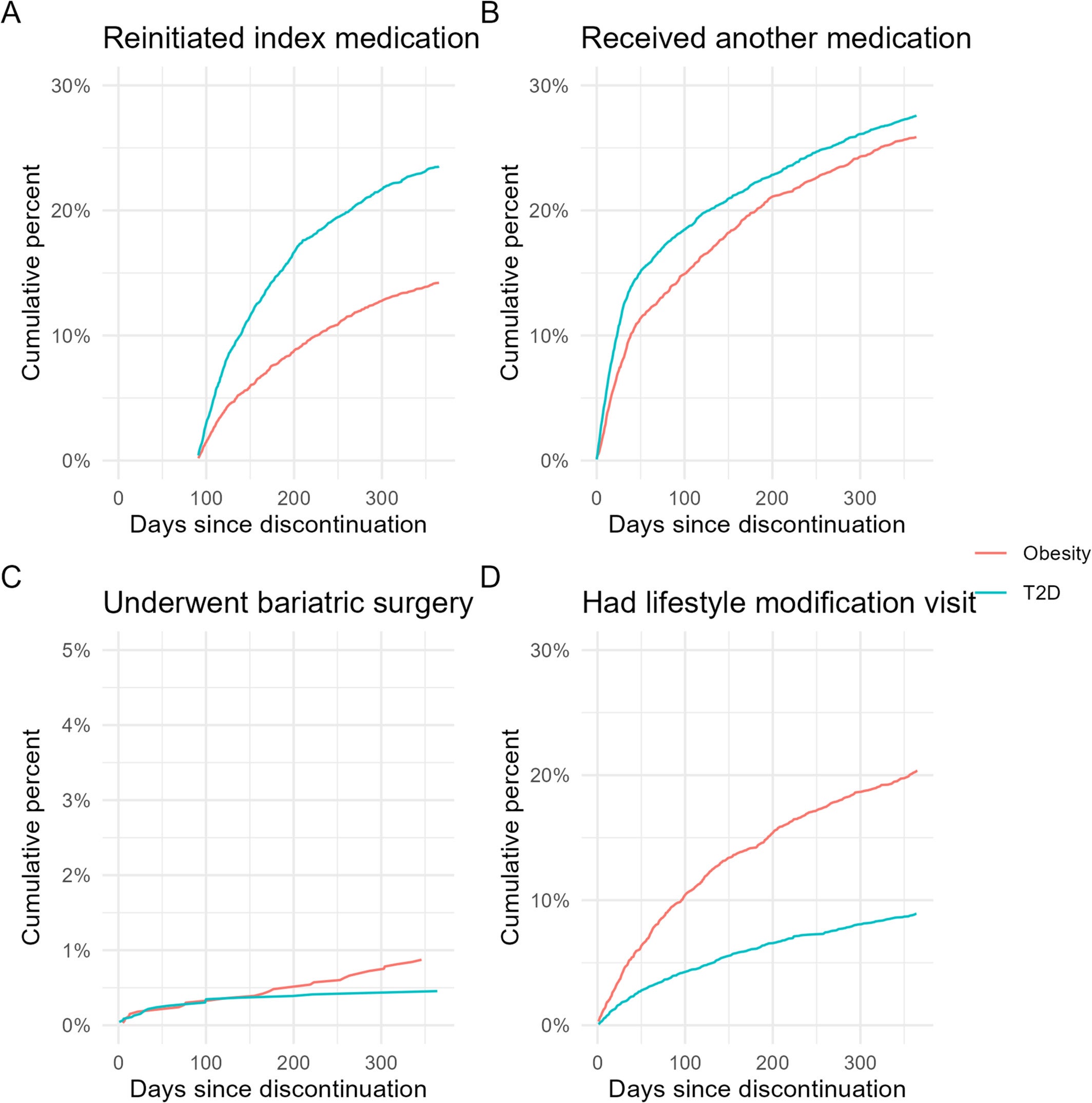
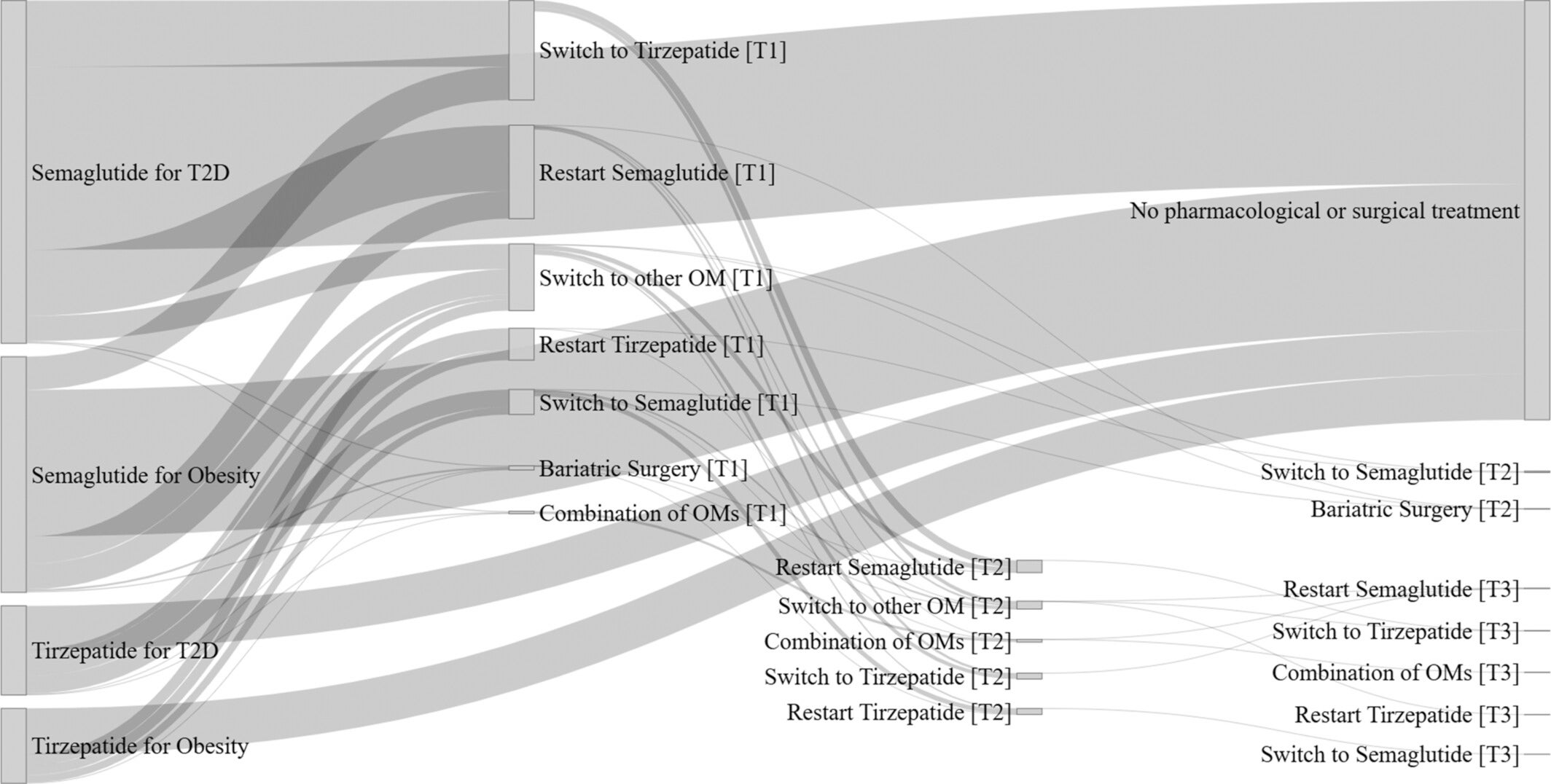
What Gasoyan’s group found was that discontinuation rarely meant the end of the treatment journey. Within a year of stopping, 19.6 percent of patients had restarted the same medication. Another 35.2 percent pursued some form of alternative treatment. Combined, that means more than half the cohort was doing something deliberate about their weight within twelve months of their initial prescription lapsing.
“Many patients do not give up on their obesity treatment journey, even if they need to stop their initial medication,” Gasoyan said.
The breakdown of what patients tried next reveals just how varied those paths were. About 27 percent switched to a different medication, which included older obesity drugs, or crossed between semaglutide and tirzepatide. Twenty percent restarted their original prescription. Another 14 percent attended structured lifestyle modification visits with dietitians, exercise specialists, or other health professionals. Less than 1 percent moved forward with bariatric surgery within the first year, though that figure rose to 1.3 percent at two years, consistent with general population rates given how long surgical pre-authorization typically takes.
The contrast with earlier randomized controlled trials is not a contradiction so much as a difference in context. Clinical trials measure what happens when patients stop a medication and receive no alternative. That is a useful scientific question, but it does not describe most patients in the real world.
“Our real-world data show that many patients who stop semaglutide or tirzepatide restart the medication or transition to another obesity treatment, which may explain why they regain less weight than patients in randomized trials,” Gasoyan said.
Insurance coverage played a significant role in shaping who could restart and how quickly. Patients using the drugs for type 2 diabetes restarted their original medication at nearly double the rate of those using them for obesity, 23.5 percent versus 14.2 percent.
Coverage for these drugs is considerably broader when diabetes is the documented indication. For obesity alone, insurance authorization remains inconsistent and frequently denied, pushing some patients toward self-pay options or abandonment of treatment altogether. Earlier work from the same research group identified cost and coverage limitations as the dominant reason patients stopped in the first place.
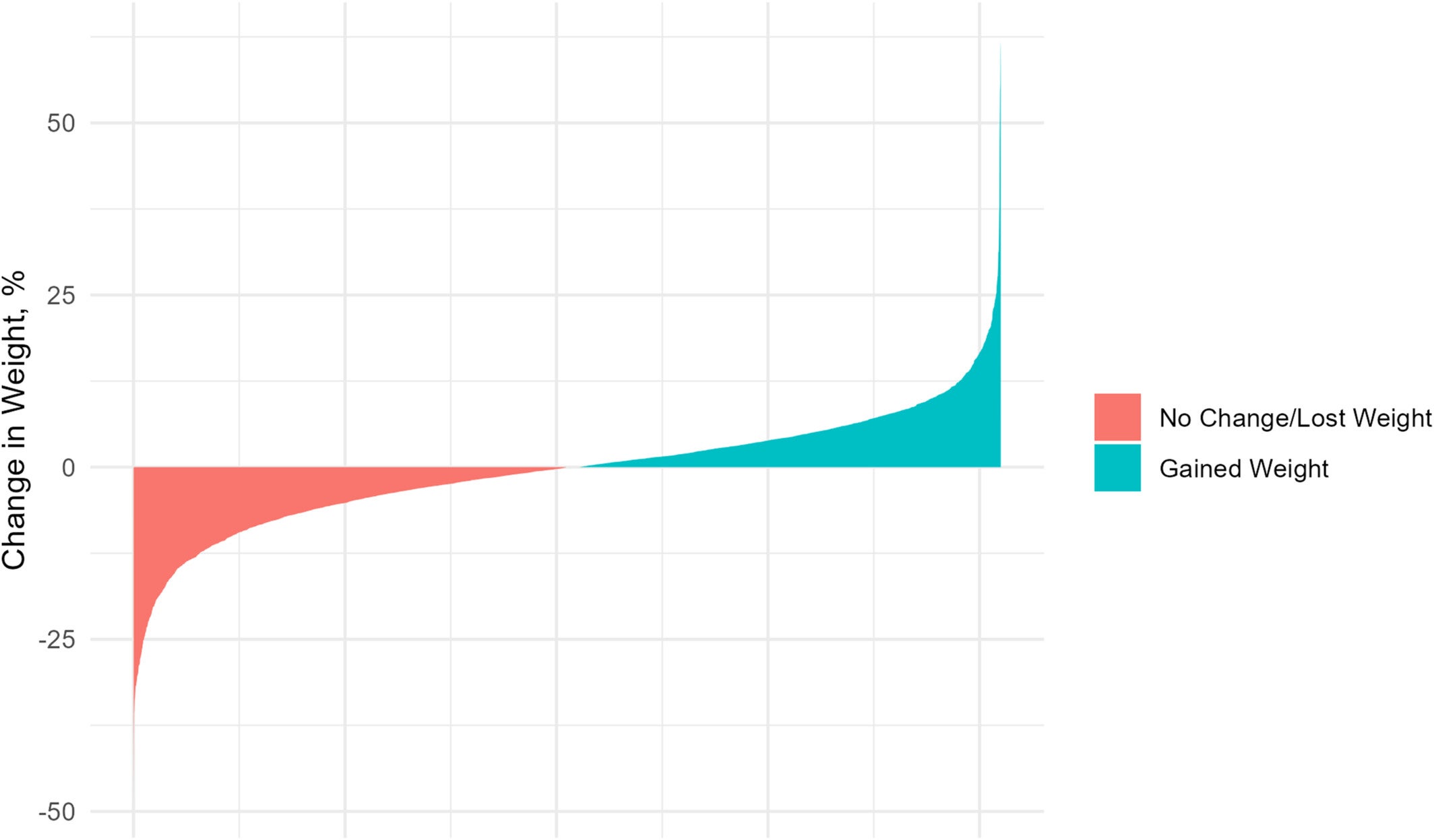
Average figures can obscure a great deal. While the mean weight change after discontinuation was small in both groups, individual outcomes varied considerably.
Among patients who had started treatment for obesity, 55 percent gained weight during the year after stopping. Forty-five percent either held steady or continued losing. Among those who had used the drugs for diabetes, the pattern was more favorable: 56 percent kept losing or stayed the same, while 44 percent gained. Those numbers suggest that for a meaningful share of patients, the year after stopping is not a period of runaway regain. It can be a period of active management, with some success.
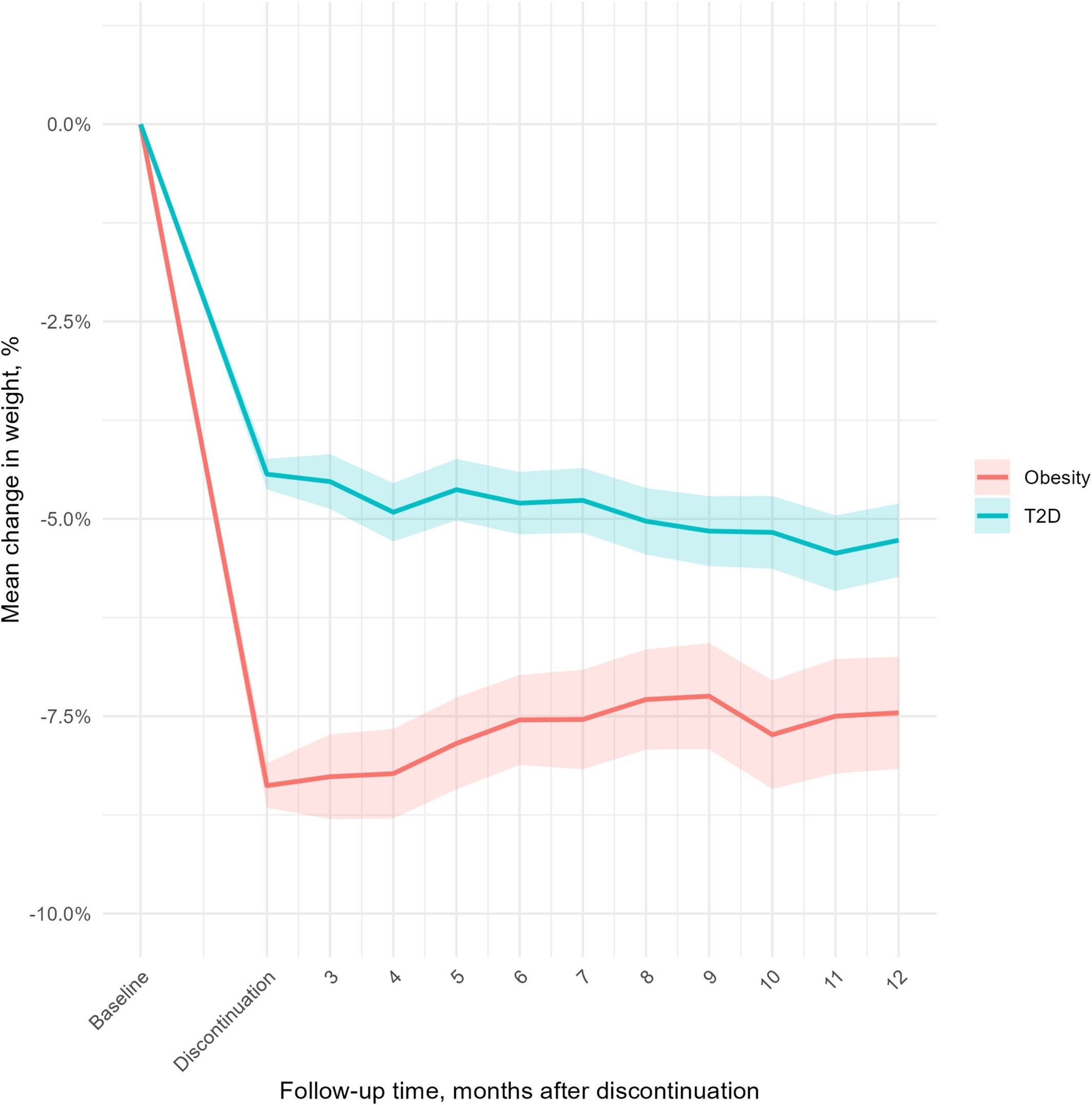
The average patient in the study was 55.7 years old, weighed about 249 pounds at baseline, and had a body mass index of 39.4. More than 63 percent were women. Before stopping their medication, those treated for obesity had lost an average of 8.4 percent of body weight. Those treated for diabetes had lost 4.4 percent.
One detail from the data is worth pausing on: many patients stopped before reaching the higher therapeutic doses associated with the greatest weight loss. At the time of discontinuation, only 27 percent of those taking semaglutide for obesity had reached a dose of 1.7 mg or above. The drugs follow a slow escalation schedule, and patients who stop early, whether due to side effects or cost, are likely leaving before the medication has reached its full effect.
The Cleveland Clinic team is explicit about where their analysis stops. The study comes from a single large health system in Ohio and Florida, which limits how broadly the findings can be applied across different demographics and care settings.
Patients who do not return to the same health system after stopping their medication are not captured, potentially skewing weight outcomes toward those who remain more engaged with care. Drug shortages during the study period may have caused some temporary discontinuations that looked like intentional stops.
The study also could not track compounded versions of these drugs, though a separate Cleveland Clinic review found that only about 2.4 percent of patients who stopped the brand-name versions had done so to switch to a compounded alternative. Unstructured lifestyle changes, meaning diet shifts or exercise increases that patients made on their own outside clinical settings, were not visible in the data either.
The researchers note that no evidence-based guidelines currently exist for managing obesity in patients who discontinue semaglutide or tirzepatide. Given how common that situation is becoming, with one-year discontinuation rates ranging from 47 to 65 percent in published literature, the absence of formal clinical guidance represents a genuine gap.
For the millions of Americans now using GLP-1 drugs, and the many who have already stopped, these findings offer a more nuanced picture than the trial data alone. Weight regain after stopping is real, but it is not inevitable, and the degree of regain appears tied to what happens after the prescription ends.
Patients who maintain some form of treatment continuity, whether through medication switches, restarts, or regular contact with dietitians and weight management specialists, appear to fare better than trial conditions would predict. That distinction matters for clinicians designing care plans and for patients navigating an insurance landscape that still treats obesity and diabetes very differently.
Gasoyan’s team plans to follow up by examining which alternative treatments work best for patients after discontinuation, aiming to give both patients and their doctors clearer guidance for a situation that is already common and becoming more so as GLP-1 use continues to grow.
Research findings are available online in the journal Diabetes Obesity and Metabolism.
The original story “Cleveland Clinic study reveals what happens when patients stop taking GLP-1 drugs” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Cleveland Clinic study reveals what happens when patients stop taking GLP-1 drugs appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.