Researchers at the University of California, Davis, have traced a deadly cat coronavirus in a way that changes what you think you know about immune infections. Their work shows that feline infectious peritonitis, or FIP, spreads through far more immune cells than scientists long assumed. The findings may also help guide research into long COVID and other lasting inflammatory illnesses in people.
FIP starts with a common feline coronavirus. Most cats never get seriously ill from that virus. In some cats, though, the virus changes inside the body and turns dangerous. The result is FIP, a disease that is almost always fatal if left untreated. It can trigger intense inflammation, damage multiple organs, and bring symptoms that linger or come back.
The UC Davis team focused on where this virus hides and grows. They examined lymph nodes from cats with naturally occurring FIP. Lymph nodes act like immune command centers. White blood cells gather there, share signals, and plan attacks on infections. If a virus spreads through these hubs, it can throw the whole defense system off balance.
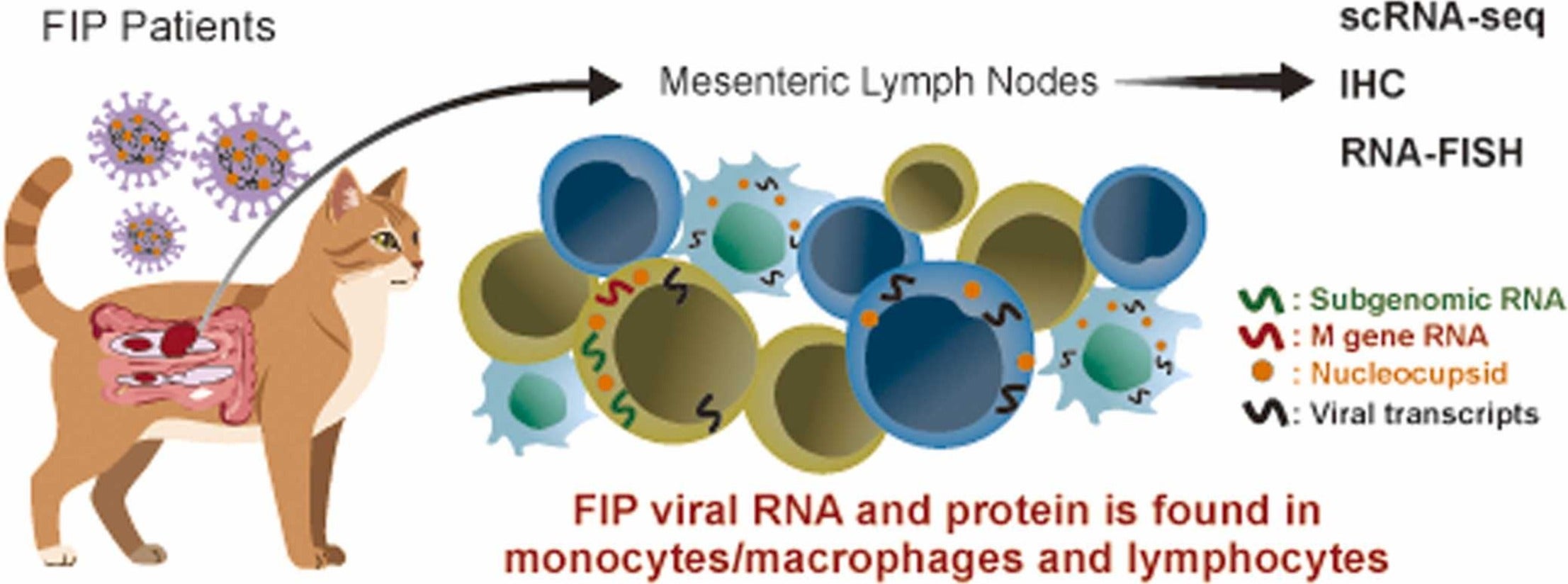
The study challenges a long-standing belief. For years, many researchers thought the virus behind FIP mainly infected one type of immune cell. The new work suggests that view was too narrow.
“What we found is that it actually infects a much broader range of immune cells, including those that are critical for fighting infection,” said lead author Amir Kol, an associate professor at the UC Davis School of Veterinary Medicine.
The team found viral material inside several kinds of immune cells in the lymph nodes. That list included B lymphocytes, which make antibodies, and T lymphocytes, which help the immune system recognize and destroy infected cells. Those two cell types form a core part of immune protection. Seeing signs of infection in them raises urgent questions about how the disease overwhelms the body.
The researchers also found evidence that the virus was not just leaving behind harmless debris. The results pointed to active viral replication inside these immune cells. That detail matters. A cell containing viral fragments may simply be cleaning up after an infection. A cell hosting replication can act as a factory, helping the virus spread and persist.
In practical terms, you can picture a virus gaining access to the very rooms where the immune system organizes its response. If those rooms are compromised, the system may misfire, overreact, or fail to clear the threat.
Lymph nodes are not just storage sites for immune cells. They are places where immune memory forms and where responses become coordinated. When a coronavirus reaches this tissue, it has the potential to affect both short-term defense and long-term stability.
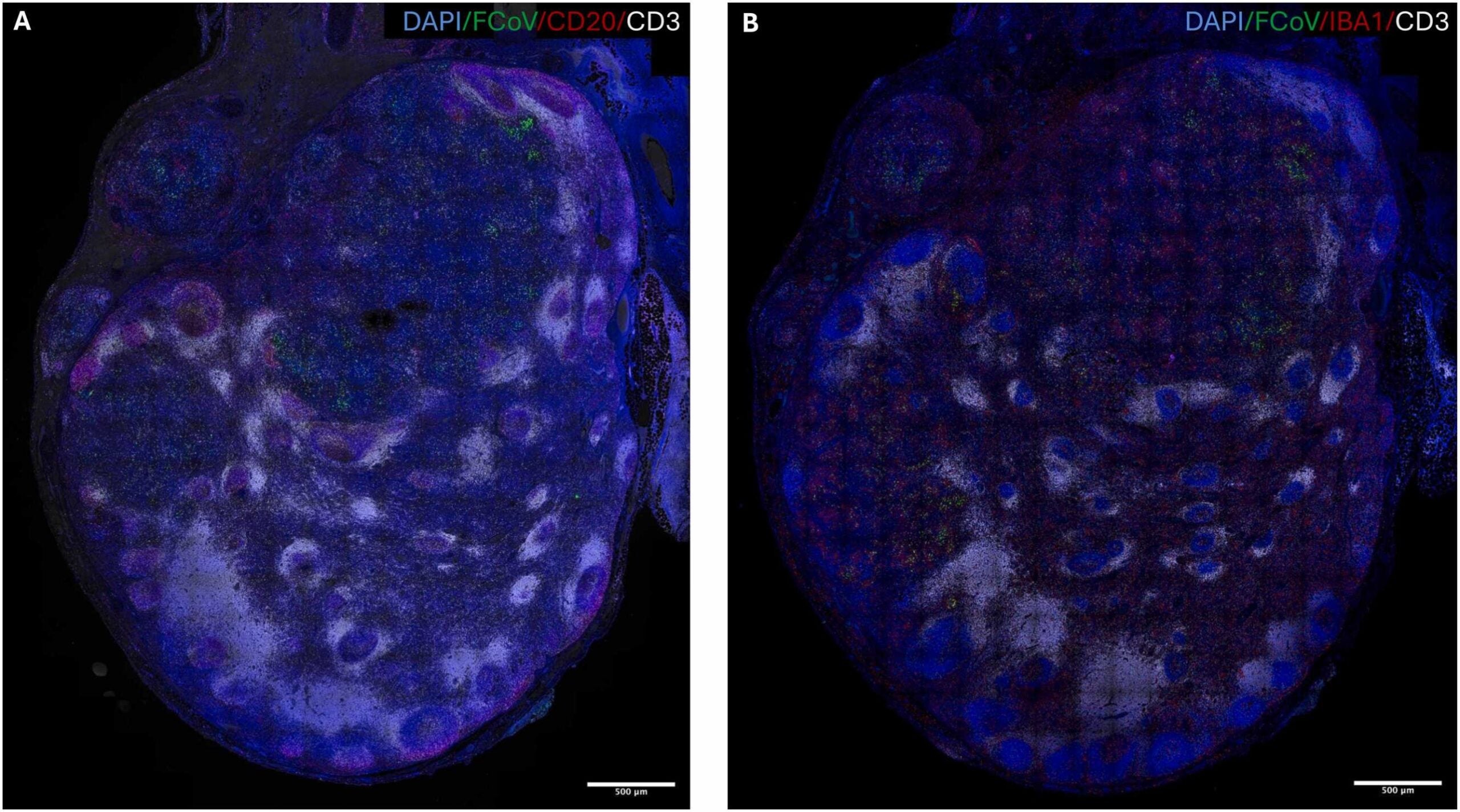
That is one reason these samples are so valuable. It is rare to get a close look at infected immune tissues during a naturally occurring coronavirus disease. In human medicine, doctors do not often remove lymph nodes from living patients just to study viral behavior. That limits what researchers can directly observe in people dealing with lingering symptoms after infection.
“This is where cats give us a unique opportunity,” Kol said. “We can directly study infected immune tissues in a naturally occurring coronavirus disease; something that’s very difficult to do in people.”
The cat disease offers a working model. It is not the same as human illness. But it shares patterns that matter, including severe inflammation and effects that can persist or return.
One of the most striking findings involves what happens after treatment. The researchers observed traces of the virus inside immune cells even after antiviral therapy ended and cats appeared healthy.
That does not automatically mean those cats were actively sick again. But it suggests the virus, or its genetic material, can linger in cells that may survive for a long time. Some immune cells can live for years. If viral traces remain inside them, they could help explain why an illness can relapse or why the immune system stays disturbed.
This idea echoes a major question in human coronavirus research. In people with severe or long-lasting coronavirus illnesses, scientists have suspected that viral remnants might persist in parts of the body or continue to disrupt immune signals. Direct proof can be hard to gather. Immune tissues are not easy to sample, and many lingering symptoms are invisible on routine tests.
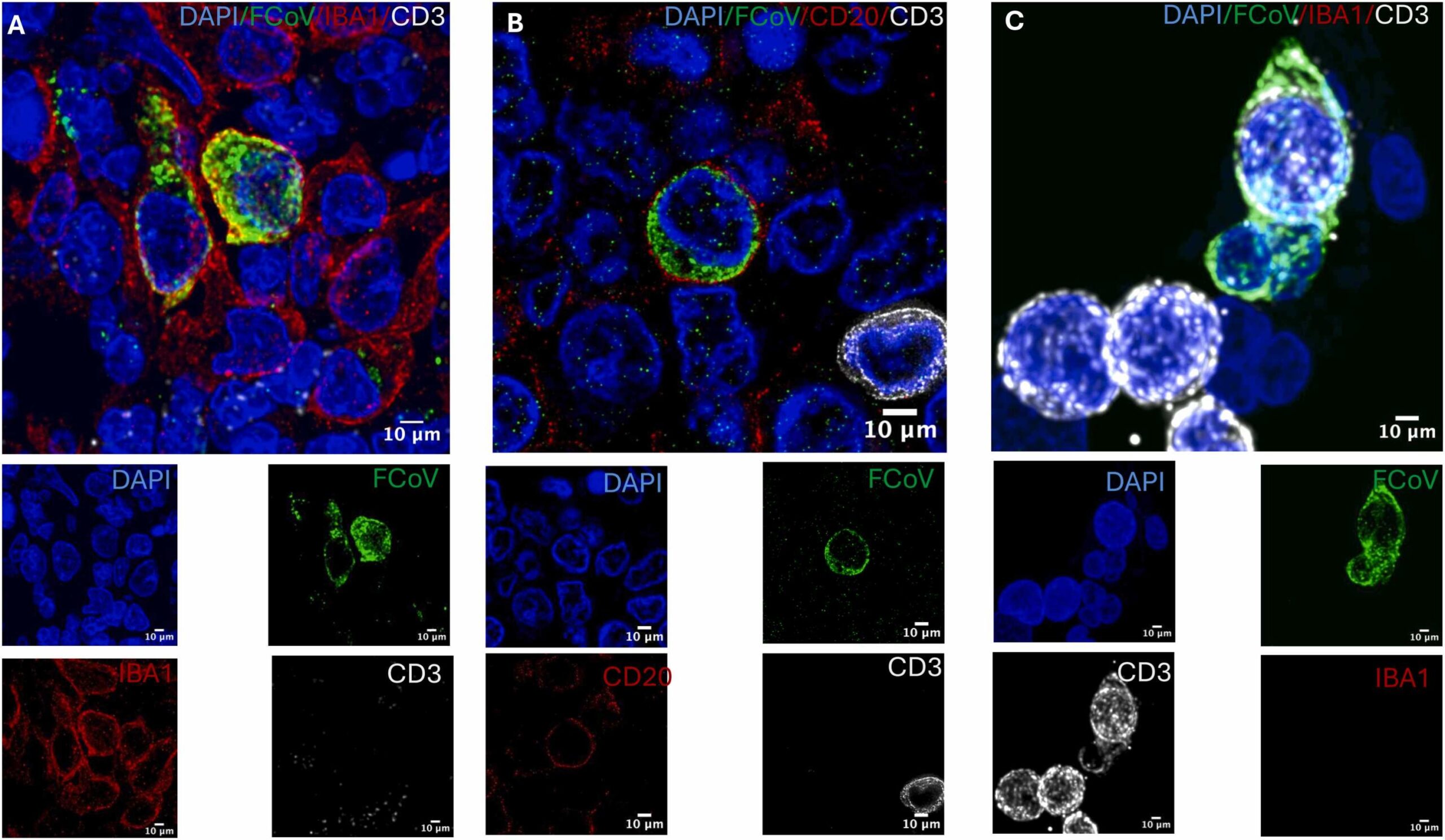
By showing persistence in immune cells in cats, the UC Davis study adds weight to the broader concept that coronaviruses may leave longer trails than symptoms suggest.
FIP does not infect people. It is a cat disease. Still, it can teach lessons about how coronaviruses behave when they trigger strong inflammation and interact with immune cells over time.
The study points to FIP as a real-world model for long-term coronavirus disease. It shows how a coronavirus can infiltrate immune hubs, reach cell types central to defense, and leave traces that remain even after a patient looks well again.
That model could help scientists ask sharper questions about long COVID and other chronic inflammatory conditions. Instead of focusing only on organs where symptoms show up, researchers can consider what happens in immune tissues that coordinate the response.
The work also highlights a broader theme in modern science. Diseases that occur naturally in animals can reveal mechanisms that are difficult to capture in human studies. When that happens, veterinary research becomes a quiet partner in human health progress.
This study gives researchers a clearer picture of how a coronavirus can spread within an immune system, especially inside lymph nodes where immune responses are organized.
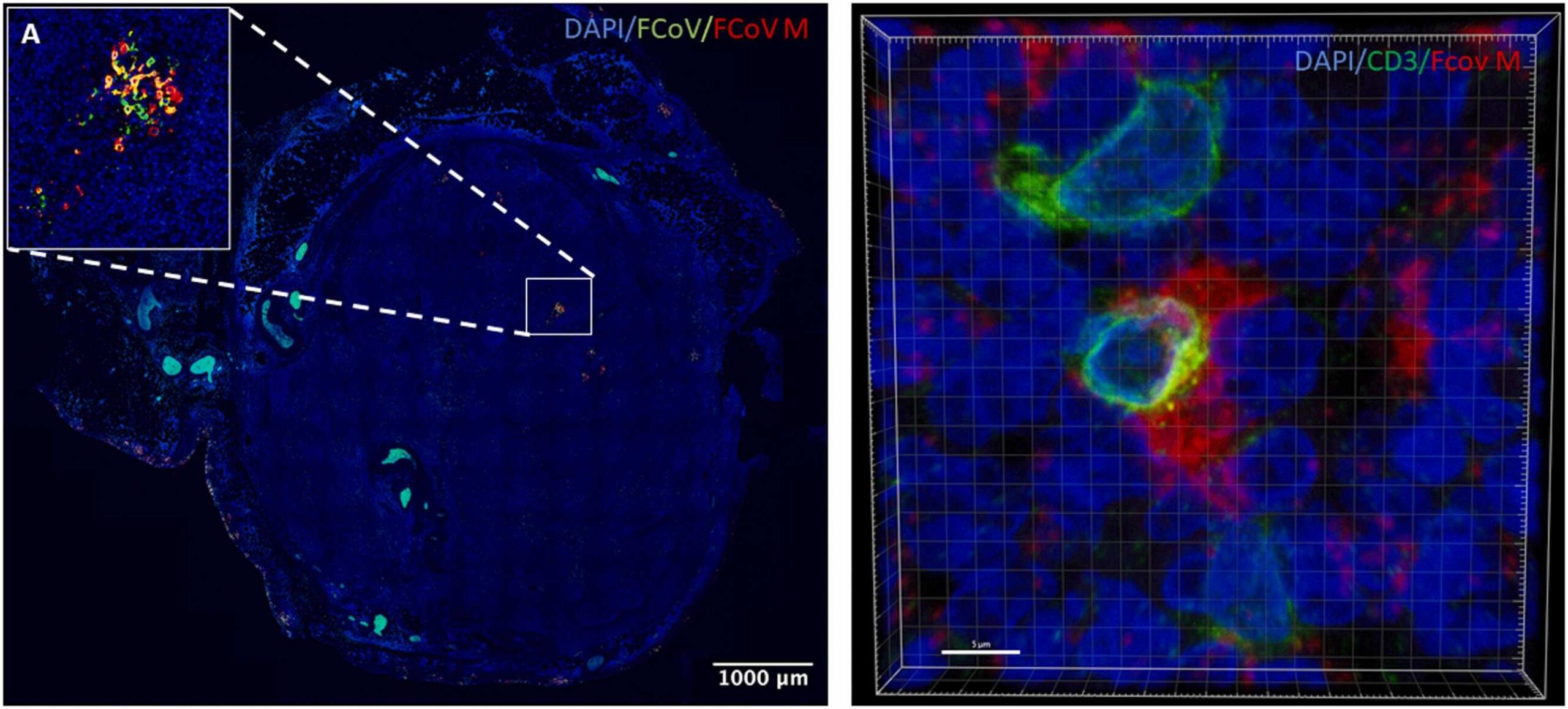
By showing that the FIP virus reaches B and T lymphocytes and may replicate inside them, the work shifts attention toward immune cell diversity, not just a single target cell type. That shift can shape future studies on persistent inflammation, relapse, and immune dysfunction in both veterinary and human medicine.
For human health research, the findings support a line of investigation that is difficult to test directly in people: whether viral material can linger in long-lived immune cells after treatment or apparent recovery. If similar patterns occur in human post-viral conditions, it could help explain why some symptoms last, return, or resist simple explanations. The cat model may also help guide what tissues to study, what cell types to watch, and what signs might indicate lingering immune disruption.
For veterinary care, understanding the virus’s broader reach could influence how scientists think about treatment timing, monitoring after therapy, and the biology of relapse. Over time, this approach could help refine strategies that protect cats and deepen your understanding of how immune systems react to coronaviruses in general.
Research findings are available online in the journal Veterinary Microbiology.
The original story “Deadly cat coronavirus helps guide research into long COVID and other illnesses in humans” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Deadly cat coronavirus helps guide research into long COVID and other illnesses in humans appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.