A few drops of blood taken within days of birth are now adding to one of the most troubling questions in environmental health: what happens when exposure to “forever chemicals” begins before a child even leaves the hospital?
Researchers at the University of California, Irvine found that newborns with higher levels of certain PFAS compounds in dried blood spots had higher odds of later developing acute lymphoblastic leukemia, or ALL, the most common childhood cancer. The link was strongest for two of the best-known PFAS chemicals, PFOA and PFOS, and it appeared to grow when both were elevated together.
The work, published in the Journal of Exposure Science & Environmental Epidemiology, does not prove that PFAS cause leukemia. Still, it pushes the evidence a step closer to the earliest window of life by measuring chemicals present at birth rather than estimating exposure from drinking water or other outside sources.
PFAS, short for per- and polyfluoroalkyl substances, have been used since the 1950s in products built to resist heat, grease, stains, and water. They have turned up in nonstick cookware, food packaging, stain-resistant furniture treatments, water-resistant clothing, firefighting foam, and personal care products. Many of them do not break down easily, and some linger in the human body for years.

That persistence is part of the concern.
Earlier work by the same research group followed more than 40,000 California children and linked PFAS in drinking water to higher risks of several childhood cancers, including acute myeloid leukemia and Wilms tumor. This time, the team tried to get even closer to the start of life.
The analysis included 125 children diagnosed with ALL and 219 children without cancer, all born in Los Angeles County between 2000 and 2015. The children were identified through the California Linkage Study of Early-onset Cancers, and the researchers used dried blood spots collected roughly 36 hours after birth and archived by the California Biobank Program.
That gave the team a way to look directly at chemical exposure in newborn blood.
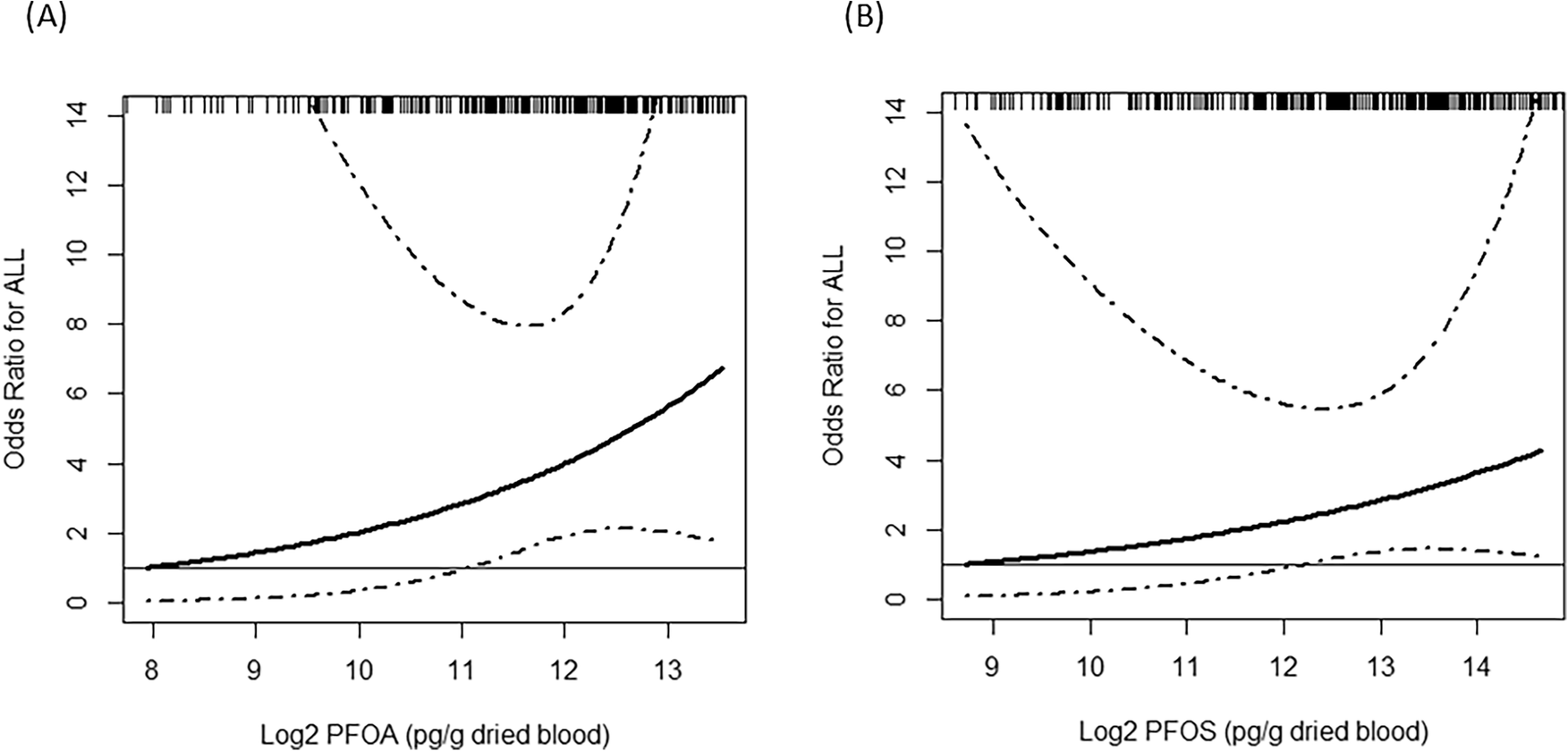
Among 23 PFAS measured with a targeted approach, 17 were detected in the blood spot samples. But only a handful appeared often enough to analyze in depth. PFOS showed the highest mean levels, followed by PFOA. Both were more concentrated, on average, in children who later developed ALL than in controls.
The association was suggestive rather than definitive. In the main statistical models, a doubling of PFOA exposure was linked to a 15% increase in the odds of childhood ALL, while a doubling of PFOS was linked to a 12% increase. Those estimates did not reach statistical significance, which means the data were too imprecise to rule out chance.
Even so, the pattern did not look random. Children in the highest exposure groups for PFOS and PFOA tended to have higher odds of leukemia than those in the lowest group, and the risk looked highest when both chemicals were elevated together.
“This research moves us closer to understanding what babies are exposed to from the very start by directly measuring PFAS present at birth, rather than estimating exposure from drinking water,” said corresponding author Veronica Vieira, chair and professor of environmental and occupational health at UC Irvine’s Joe C. Wen School of Population & Public Health. “By capturing exposures during a critical window of development, we are gaining a clearer picture of how environmental contaminants may contribute to childhood cancer risk.”
The study did not stop with the usual suspects.
Using non-targeted analysis, the team also identified 26 additional PFAS compounds in the newborn blood spots. Some belonged to newer classes of PFAS, including chemicals used as replacements for older compounds such as PFOA in fluoropolymer manufacturing. Others could not be fully identified.
That matters because PFAS are not a single chemical. They are a large family with thousands of structures, and most health studies have focused on a relatively small number of legacy compounds.
Nine of the PFAS found through the non-targeted work appeared in more than half of the study population. Several showed patterns that tracked with higher leukemia risk. One compound labeled C10HF19O5 showed a statistically significant upward trend across increasing exposure quartiles. Another, C4HF7O3, stood out in mutually adjusted models with a notably elevated odds ratio, though that estimate came from a much smaller subset of participants.
The authors were careful not to oversell those findings. Many of the non-targeted signals came from a limited number of detectable samples, and some compounds could not be fully characterized. Even so, the results hint that the PFAS story may reach beyond the compounds that usually get measured in public health monitoring.
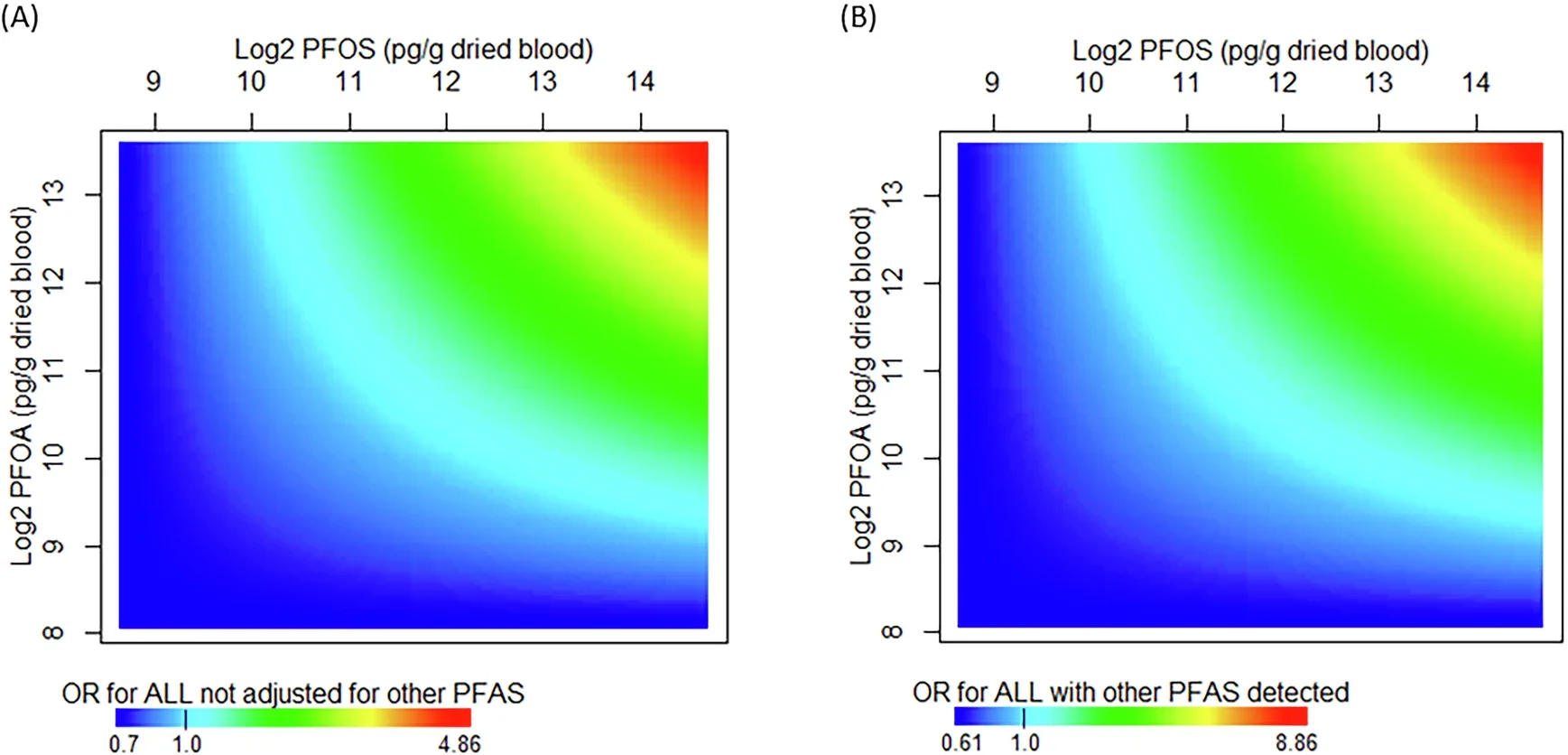
That is one of the most striking parts of the work. It suggests that focusing only on PFOA and PFOS may miss other chemicals moving through the same exposure pathways.
The strongest associations in the study generally appeared among non-Hispanic children.
Researchers treated those results as preliminary. The subgroup analyses were based on smaller numbers, which can make estimates unstable. But the pattern matched broader differences in exposure seen in the study population.
Legacy PFAS such as PFOS and PFOA tended to be higher among children born earlier in the study period, those from higher-value neighborhoods, and those whose parents were older, more educated, privately insured, or born in the United States. Non-Hispanic white children also tended to have higher levels of PFOS and PFOA, although those differences were not statistically significant.
Some of the newer or less-studied PFAS showed different patterns. Certain ether-linked compounds were more common among Asian children and among children of mothers born outside the United States and Mexico. Some longer-chain compounds identified in the non-targeted analysis were higher among Hispanic children and those born in later years.
Those contrasts do not explain the leukemia findings on their own, but they do show that PFAS exposure is not evenly distributed.
Diet, consumer product use, geography, housing, and contaminated water systems may all shape who gets exposed to what, and when.
One of the clearest strengths of the research is timing. The dried blood spots were collected before cancer diagnosis, so the measurements could not have been altered by treatment or by parents trying to recall past exposures. The team also used a survey design that intentionally oversampled children born in public water districts with known PFAS contamination, then adjusted statistically for that sampling design.
The blood spots also offered something earlier California PFAS work could not: a direct biological measure at birth.
Still, the evidence has limits.
The study was relatively small, with 125 cases and 219 controls. Many PFAS in the targeted panel were not detected in most participants, which reduced statistical power. The newborn blood spots provided only a single measure of exposure, and dried blood spot testing has constraints because of the small amount of blood available and the challenge of measuring very low concentrations. In some samples, contamination from the card blank led researchers to exclude measurements altogether.
The analysis also did not establish cause and effect. Most of the observed links, including those for PFOA and PFOS, were not statistically significant. The authors describe the results as suggestive, not conclusive.
That caution runs throughout the research.
The new findings also sit inside a mixed body of evidence. The UC Irvine team notes that another recent California study found elevated but non-significant risks between PFOS in newborn blood spots and childhood ALL. Other studies, including one in Finland and one California study based on maternal serum or modeled water exposure, reported null results for some PFAS-ALL links, though the Finnish work did find positive associations in certain time periods when PFOS levels were higher.
So the story is not settled. But it is getting harder to ignore.
The study adds to growing concern that PFAS exposure may begin affecting health during the earliest stages of life, including a period when organs and immune systems are still developing. For public health officials, that raises questions not just about contaminated water, but also about food packaging, household products, and the many lesser-known PFAS that remain poorly tracked.
It also points to the value of newborn blood spots as a research tool. By showing that these archived samples can capture both well-known and less familiar PFAS, the work opens a path for studying how early-life exposure may connect to disease years later.
For families, the findings do not offer a simple checklist or a direct warning that a child exposed to PFAS will develop leukemia. They do suggest that reducing PFAS exposure at the population level, especially in drinking water and common consumer products, remains an important public health goal.
More studies will be needed, particularly larger ones that can better measure rare exposures and test newer PFAS compounds now entering the marketplace. But this much is clear: the chemicals turning up in newborn blood are not just markers of a contaminated world. They may also be clues to how some childhood cancers begin.
Research findings are available online in the Journal of Exposure Science & Environmental Epidemiology.
The original story “‘Forever chemicals’ in newborn blood linked to childhood leukemia” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post ‘Forever chemicals’ in newborn blood linked to childhood leukemia appeared first on The Brighter Side of News.