A person can seem healthy and still carry subtle biological signs of trouble long before the first tremor or slowed movement appears. In Parkinson’s disease, one of those early signals may be living in the gut.
A new study led by researchers at University College London found that people with Parkinson’s have a distinct pattern of gut microbes, and that similar patterns also appear in some people who do not yet have the disease, including those with a known genetic risk. That raises a striking possibility: changes in the microbiome could help flag elevated Parkinson’s risk before symptoms begin.
Parkinson’s is already one of the world’s fastest-growing neurological disorders. By the time doctors can diagnose it through motor symptoms, more than half of the brain’s dopamine-producing neurons have typically already been lost. That makes early detection one of the field’s biggest priorities.
Professor Anthony Schapira of the UCL Queen Square Institute of Neurology said the need is urgent. “Parkinson’s disease is a major cause of disability worldwide, and the fastest growing neurodegenerative disease in terms of prevalence and mortality. There is an urgent need to develop treatments that can stop or slow the disease’s progression.”
He added that such treatments will be most useful only if doctors can identify people much earlier, before the disease is firmly established.
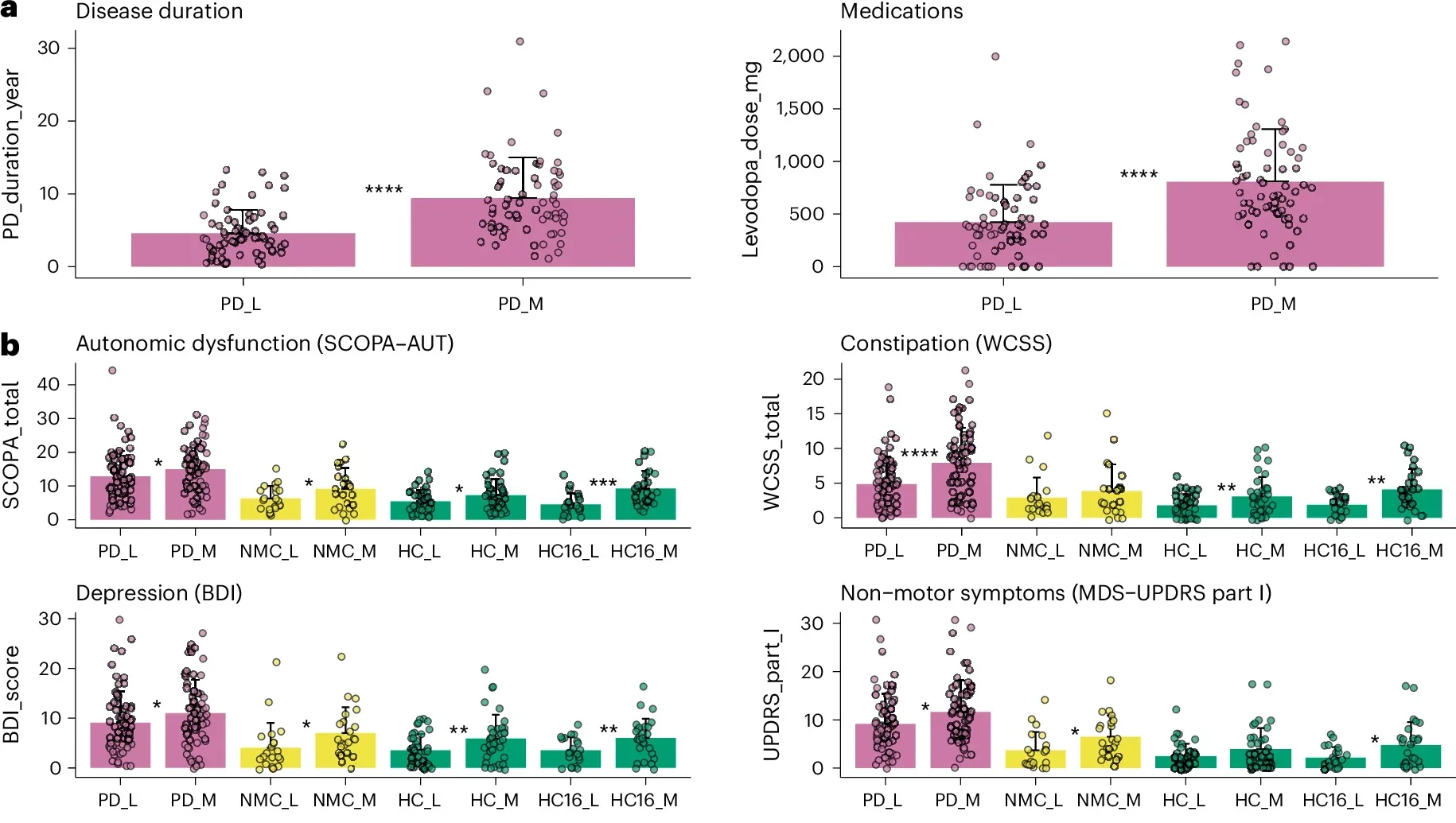
The research team analyzed clinical and stool data from participants in the United Kingdom and Italy. The main microbiome analysis included 271 people with Parkinson’s disease, 43 people who carried a GBA1 gene variant linked to much higher Parkinson’s risk but had no clinical symptoms, and 150 healthy controls.
The GBA1 variant is the most common known genetic risk factor for Parkinson’s. Even so, not everyone who carries it will develop the disease. Lifetime risk estimates cited in the study range from 10% at age 60 to 19% at age 80. That uncertainty has made it difficult to identify which carriers are actually moving toward Parkinson’s.
The gut findings offered a possible clue.
Researchers found that 176 microbial species differed in abundance when they compared people with Parkinson’s to healthy controls. Some species were more common in Parkinson’s, while others were depleted. The altered microbes accounted for more than a quarter of the microbiome.
What stood out even more was what happened in the symptom-free GBA1 carriers. Of those 176 Parkinson’s-linked species, 142 shifted in the same general direction in the at-risk carriers. Their microbiomes looked like an intermediate state, not fully healthy, but not as altered as those in people with diagnosed Parkinson’s.
That pattern suggests the microbiome may begin changing before the disease becomes obvious.
“For the first time we identify bacteria in the gut of people with Parkinson’s that can also be found in those with a genetic risk for the disease, but before they develop symptoms,” Schapira said.
The study also found that some microbial changes may emerge earlier than others.
Certain species moved steadily across the three groups, from healthy controls to at-risk carriers to people with Parkinson’s. Others seemed to shift more abruptly once the disease was clinically present. Among the early and coherent changes were increases in some oral and inflammatory-associated bacteria, along with drops in bacteria tied to butyrate production, a process often linked to gut health and anti-inflammatory effects.
The work adds weight to a growing view of Parkinson’s as more than a brain disorder alone. Researchers have increasingly focused on the gut-brain axis, especially because non-motor symptoms such as constipation, autonomic dysfunction, sleep changes, and loss of smell can appear years before the classic motor signs.
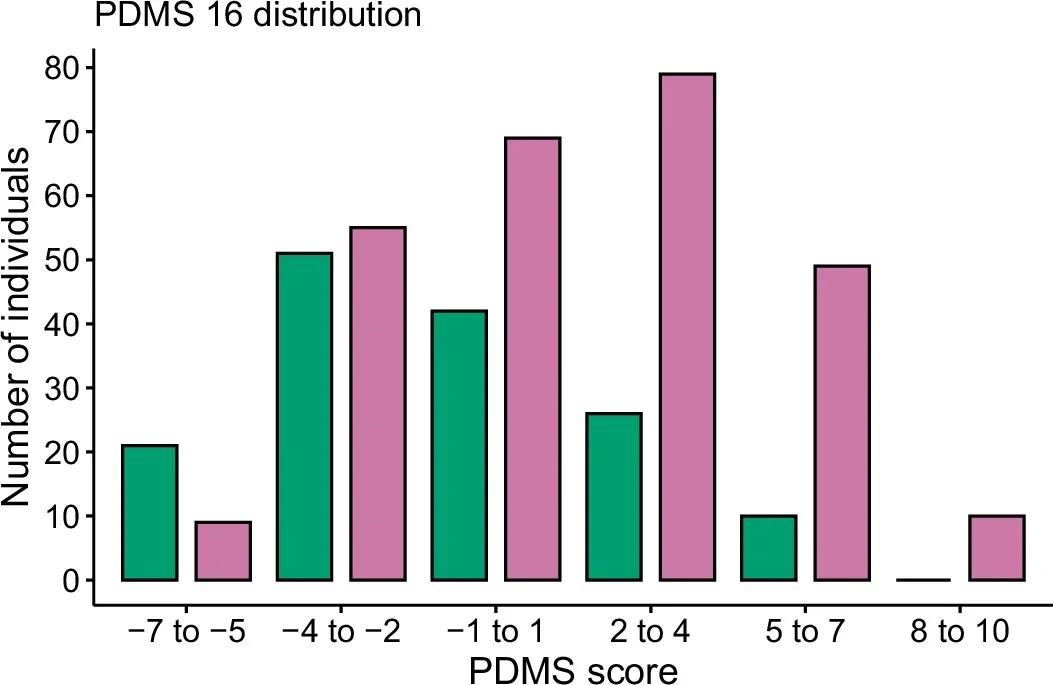
In this study, people with more altered microbiomes often had worse non-motor symptoms. That was true not only in Parkinson’s patients, but also in symptom-free GBA1 carriers, where stronger Parkinson’s-like microbiome patterns lined up with more prodromal features. Among the GBA1 carriers, 10 individuals met the threshold for estimated prodromal Parkinson’s based on available criteria.
The study also found something unexpected in the healthy control group.
A small share of those participants, about 20%, had microbiome changes that resembled those seen in Parkinson’s. They did not carry the known genetic risk factor studied here, but some also showed traits associated with poorer health profiles, including more depression, anxiety, autonomic dysfunction, constipation, and less healthy eating habits.
That does not mean they are destined to develop Parkinson’s. The researchers were careful not to make that claim. But it does suggest the microbiome may be detecting risk that genetic screening alone would miss.
Participants also reported on their diet, and the results hinted at another possible thread. People with more balanced, varied diets were less likely to have microbiomes that looked Parkinson’s-like. Lower dietary quality, including lower fruit and vegetable intake, appeared more often in those with the more altered gut profiles.
Co-lead author Professor Stanislav Dusko Ehrlich said the findings raise the possibility of practical prevention strategies. “Gut microbiome analysis can enable us to identify individuals who are at risk of developing Parkinson’s disease, so that we can suggest ways for them to reduce their own risk, such as through dietary adjustments.”
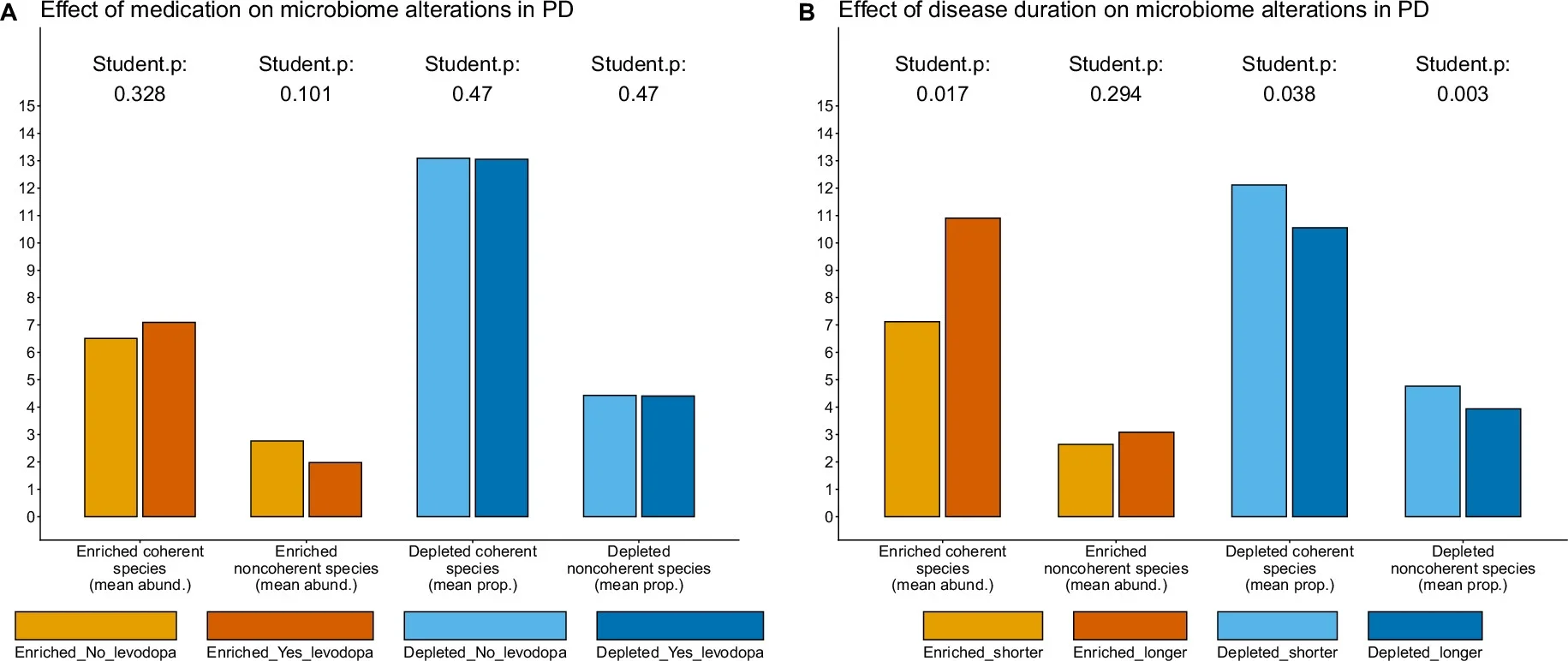
The team also found signs that microbiome disruption tracked more closely with disease duration than with medication use, suggesting the microbial changes may be more closely tied to disease progression than to treatment.
To test whether the broader pattern held up, researchers compared their findings with independent Parkinson’s cohorts from the United States, Turkey, and Korea. Across those groups, many of the same microbiome shifts appeared again, despite geographic differences.
Still, the study has important limits. It was cross-sectional, meaning it captured one moment in time rather than following people over years. The group of symptom-free GBA1 carriers was relatively small. And while outside cohorts supported the Parkinson’s signal, there were not enough well-characterized disease-free groups to fully validate the risk findings in healthy people.
That means the central question remains open: do these microbiome patterns predict who will actually go on to develop Parkinson’s?
The answer will require long-term follow-up.
This study points toward a future in which a stool sample could help identify people moving closer to Parkinson’s before the disease becomes clinically obvious.
That would not replace neurological evaluation, but it could become part of a broader early-risk screen alongside genetic and clinical data.
The work also suggests that diet and the gut microbiome may become targets for prevention research, especially for people already known to be at elevated risk.
Research findings are available online in the journal Nature Medicine.
The original story “Gut microbiome changes can signal Parkinson’s disease risk years before symptoms” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Gut microbiome changes can signal Parkinson’s disease risk years before symptoms appeared first on The Brighter Side of News.