A material built to tell left-handed light from right-handed light has long had a frustrating weakness. It mostly ignored visible light.
That limitation may now be easing. A University at Buffalo-led team reports that it paired a chiral semiconductor with a non-chiral molecule that absorbs visible light more readily, producing a hybrid system that not only responds to visible wavelengths but also keeps the optical handedness that makes chiral materials unusual in the first place.
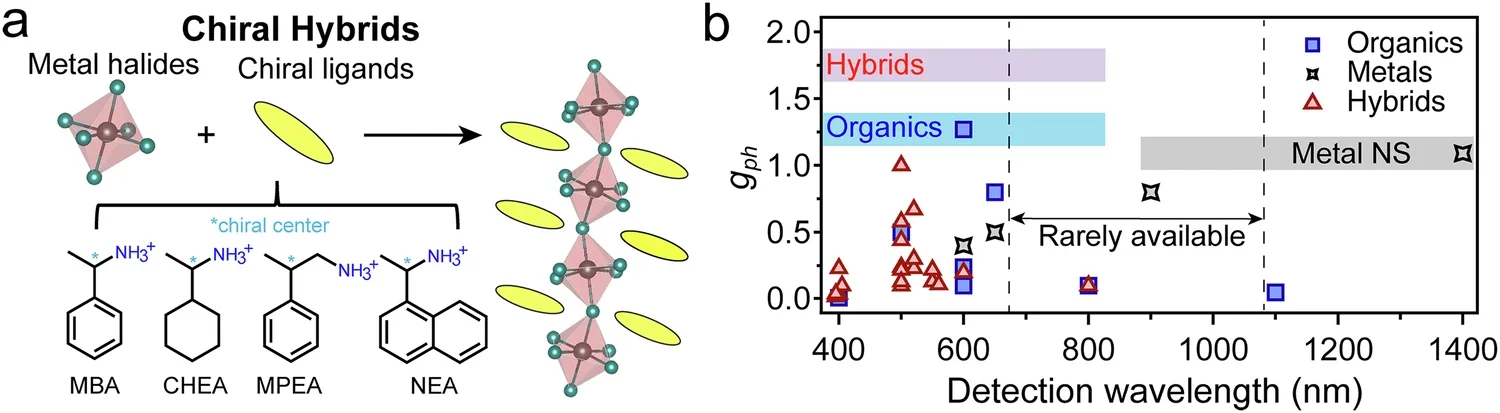
The work, published in Nature Communications, centers on chiral perovskites, a class of semiconductors whose structures come in left- and right-handed forms. Those materials can respond differently to left- and right-circularly polarized light, making them attractive for polarized-light detection, optical communication systems and other optoelectronic uses. The problem is that many chiral semiconductors work best under higher-energy ultraviolet light, not the visible wavelengths used in many practical devices.
“We were able to transfer the properties of chirality to a non-chiral molecule,” said Wanyi Nie, associate professor in the University at Buffalo Department of Physics and the study’s corresponding author. “The resulting material retains the handedness that makes chiral semiconductors promising building blocks for next-generation electronics, while adding the ability to respond to visible light.”

That shift came from chemistry, not from rebuilding the whole material from scratch.
The researchers combined a chiral perovskite with F4TCNQ, an organic dopant molecule known for accepting electrons. Once the two were brought together, the system developed a new absorption band in the visible range, specifically from 550 to 750 nanometers. The team says this happened because electrons moved from the chiral host into higher-energy states associated with the dopant, forming what is known as a charge-transfer state.
Chirality matters in chemistry because a left-handed structure and a right-handed one can behave differently, even when they contain the same atoms. The source material points to DNA as a familiar example of a right-handed chiral structure. In semiconductors, that same kind of handedness can shape how a material interacts with circularly polarized light.
“This allows for more complex ways of detecting, processing and transmitting information using light, with potential applications in advanced polarized light sensors, optical communications systems and photocatalysis,” said co-author Dave Tsai, assistant professor in the University at Buffalo Department of Chemical and Biological Engineering.
Still, the visible-light gap has hung over the field. The paper notes that most organic and hybrid chiral semiconductors cut off between about 400 and 550 nanometers, with relatively few chiral semiconductor detectors developed in the 600-to-1000-nanometer range. Low-dimensional chiral perovskites also tend to have low conductivity, which limits detector performance.
The new study tackled both problems at once. Adding 1% F4TCNQ changed the material’s appearance, turning the solution and thin film green, a visual sign that the optical behavior had shifted. More importantly, circular dichroism measurements showed that the broad visible-light absorption band still carried a chiral signature. The non-chiral dopant on its own showed no such signal.
In other words, the guest molecule appeared to inherit handedness from the host.
“The core physics is associated to the electron transfer that carries chirality from the chiral perovskite host to the non-chiral dopant molecule,” Nie said.
Tsai put it more casually, comparing the two materials to basketball players. “The chiral molecule is the guard and the dopant molecule is the forward. Guards read the play and then pass the ball to the forward,” he said.
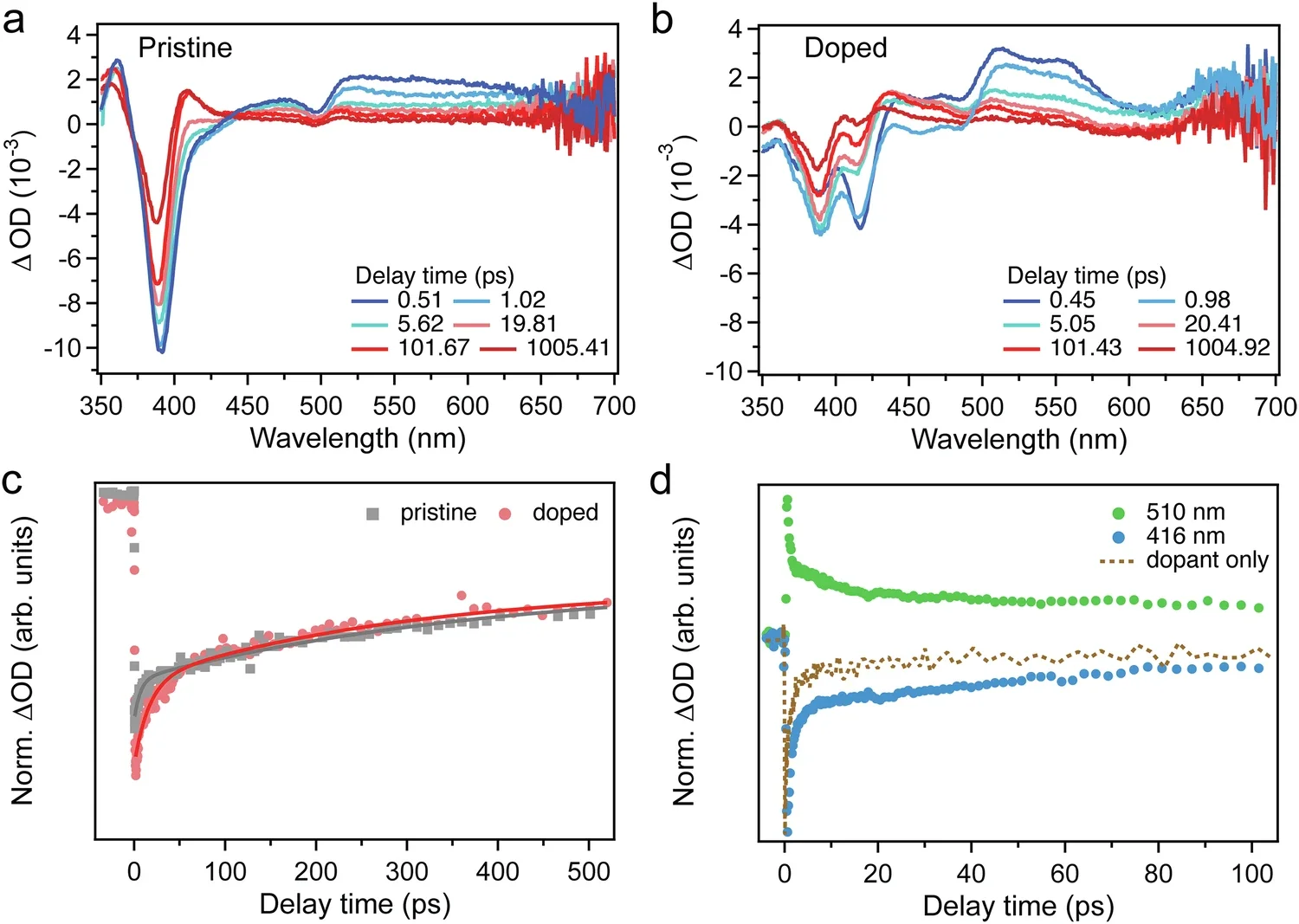
The team used several tools to probe what was happening inside the hybrid. In transient absorption spectroscopy, the doped films showed new features at 416 nanometers and around 510 nanometers that were absent in the same form in the pristine material. The researchers say those signals point to charge transfer and recombination at the interface between host and guest.
Their timing data suggested very fast transfer. The paper states that charge moved from the excited chiral perovskite matrix to the dopant at a rate faster than 1 picosecond. In the pristine sample, one decay component measured 9.01 picoseconds and another 416.67 picoseconds. In the doped sample, those values shifted to 16.13 picoseconds and 303.03 picoseconds.
The conductivity changed too. According to the study, the room-temperature dark conductivity of the doped film improved by more than two orders of magnitude compared with the undoped version. The activation energy also dropped, from 480 millielectronvolts in the undoped device to 350 millielectronvolts in the film with 1% F4TCNQ. The researchers interpret that as a sign that the dopant creates a more favorable path for charge transport.
Device tests gave the clearest practical result. Photodetectors made from the guest-host films responded differently to left- and right-circularly polarized light under both blue light at 405 nanometers and red light at 635 nanometers. The anisotropic factor, or gph, reached 0.18 under blue light and 0.12 under red light, showing that the detector could distinguish polarization states even in a region where the original chiral perovskite was far less active.
That matters because the red-light response came from the newly introduced charge-transfer absorption feature, not just from the material’s original band edge.
The paper does not present the system as finished technology. The authors say the devices had relatively low responsivity and external quantum efficiency, which they attribute to an unoptimized device structure and testing conditions. The detectors were also tested without bias, partly to avoid large injected currents that would swamp the polarization-dependent signal.
There are deeper scientific uncertainties as well. The researchers modeled two possible ways the dopant might sit within the host lattice. One configuration, called model-2, produced much stronger optical activity in the calculations, with a 20-fold increase in oscillator strength over the alternative model. But the paper is careful here: the atomistic interaction models were not directly derived from experimental X-ray diffraction data. Instead, they represent plausible dopant arrangements.
The calculations also came with other limits. The modeled absorption onset was blue-shifted relative to experiment because the simulations used a finite crystal fragment, and unresolved positions and orientations of the chiral molecules limited direct spectral comparison.
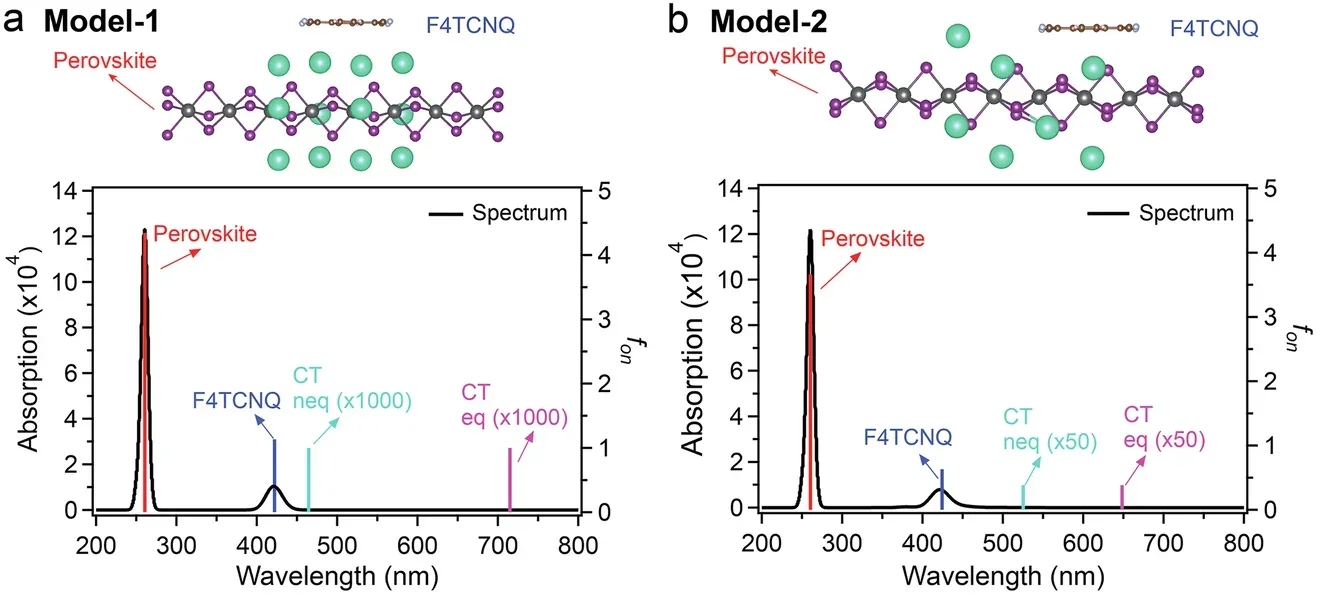
Even so, the broad picture held together across the measurements and simulations. The dopant did not simply sit nearby. The evidence suggests it diffused into the lattice, formed a closely packed structure with the chiral perovskite, and created optically active charge-transfer states that extended absorption into the visible range.
“We see that the ability to tell left- from right-handed light is being passed from one material to another, but we don’t yet fully understand how electrons carry that information across, and what governs this process,” Nie said.
The study points to a possible workaround for one of the main bottlenecks in chiral semiconductors: weak visible-light performance. By using molecular doping to create charge-transfer states, the researchers expanded light absorption into the 550-to-750-nanometer range while preserving the ability to distinguish circular polarization.
That combination could help in the design of polarized-light photodetectors and other optoelectronic devices that need both chiral selectivity and stronger response in the visible part of the spectrum. The conductivity gain is also important because it may make thicker, more light-absorbing films more useful without losing as much signal in transport.
The broader claim in the paper is that this host-to-guest chirality transfer could extend beyond this one material pair. If that proves true, it could offer a general strategy for improving other chiral semiconductors that currently struggle with narrow absorption ranges or weak electrical performance.
Research findings are available online in the journal Nature Communications.
The original story “Molecular add-on helps chiral perovskite semiconductors detect visible light” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Molecular add-on helps chiral perovskite semiconductors detect visible light appeared first on The Brighter Side of News.