A quiet signal inside a tumor may soon guide how surgeons treat early lung cancer. Researchers now report that hidden genetic patterns can reveal whether a tumor has already begun to spread through blood vessels, even before surgery begins.
That discovery could change how doctors approach lung adenocarcinoma, the most common form of lung cancer in the United States. It may also help answer a painful question that many patients face after surgery: will the cancer come back?
Lung cancer remains the leading cause of cancer-related death. It kills more people in the United States than breast, prostate, and colon cancers combined. Even when doctors detect it early, outcomes can vary widely.
Some tumors behave quietly and never return after removal. Others come back months or years later, often more aggressive than before. One of the strongest warning signs is vascular invasion.
Vascular invasion occurs when cancer cells break into nearby blood vessels. Once inside, they can travel through the bloodstream and seed new tumors elsewhere. Even a small tumor can carry this risk.
Doctors can usually confirm vascular invasion only after surgery. Pathologists examine removed tissue under a microscope. They look for cancer cells inside blood vessels. By then, however, the surgical decision has already been made.
This gap leaves surgeons working with incomplete information. A less aggressive surgery may leave behind risk. A more aggressive approach may expose patients to unnecessary harm.
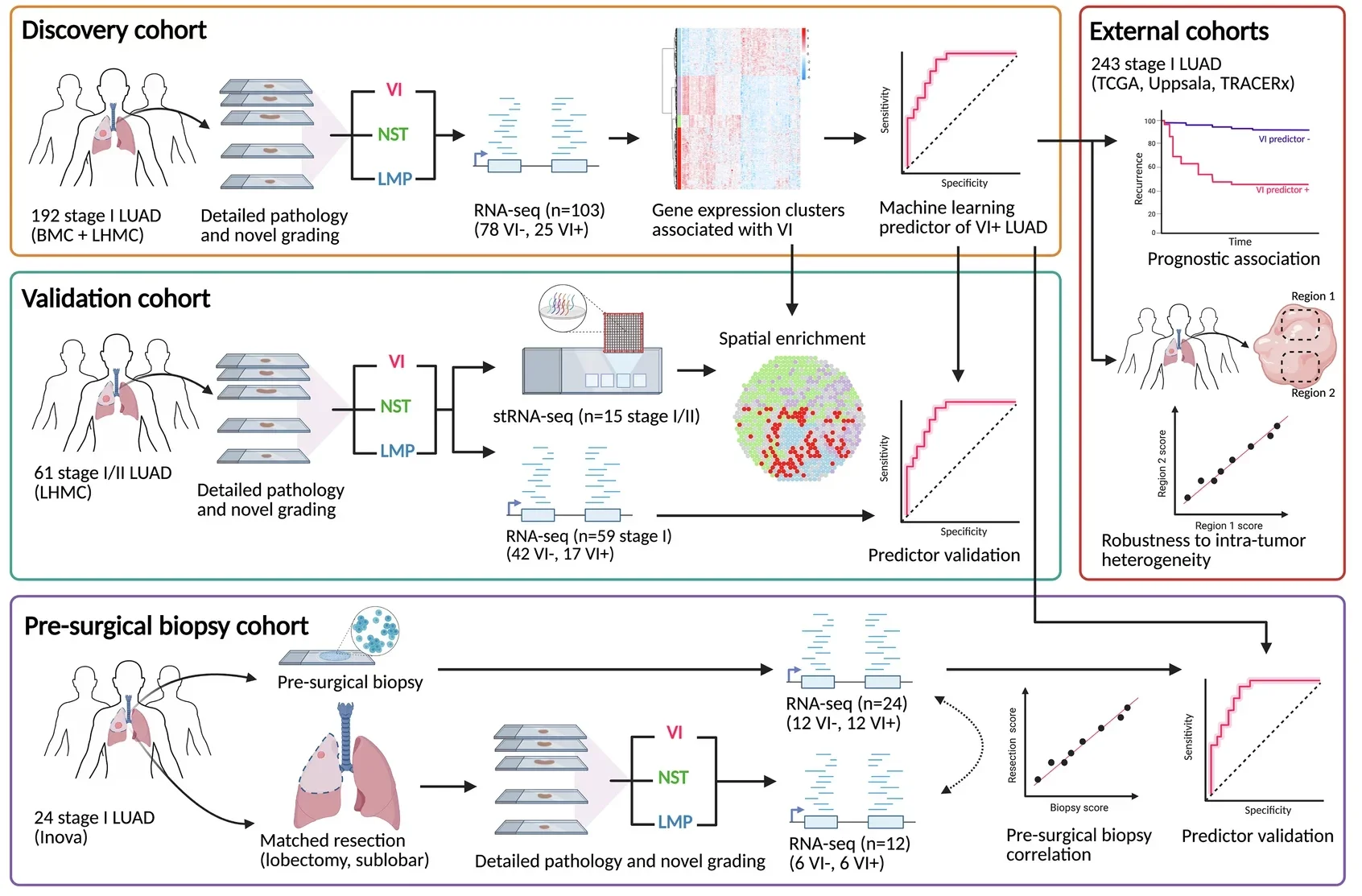
Researchers from Boston University Chobanian & Avedisian School of Medicine believe they have found a way to close that gap. Their study shows that tumors with vascular invasion carry a distinct pattern of gene activity.
More importantly, those patterns can be detected in small samples taken before surgery.
“We think this is a potential game changer for patients with early-stage lung cancer,” said Marc Lenburg, PhD, professor of medicine, bioinformatics and pathology at the school. “Our findings suggest a simple biopsy-based test could help doctors better identify patients at higher risk of recurrence and guide treatment decisions.”
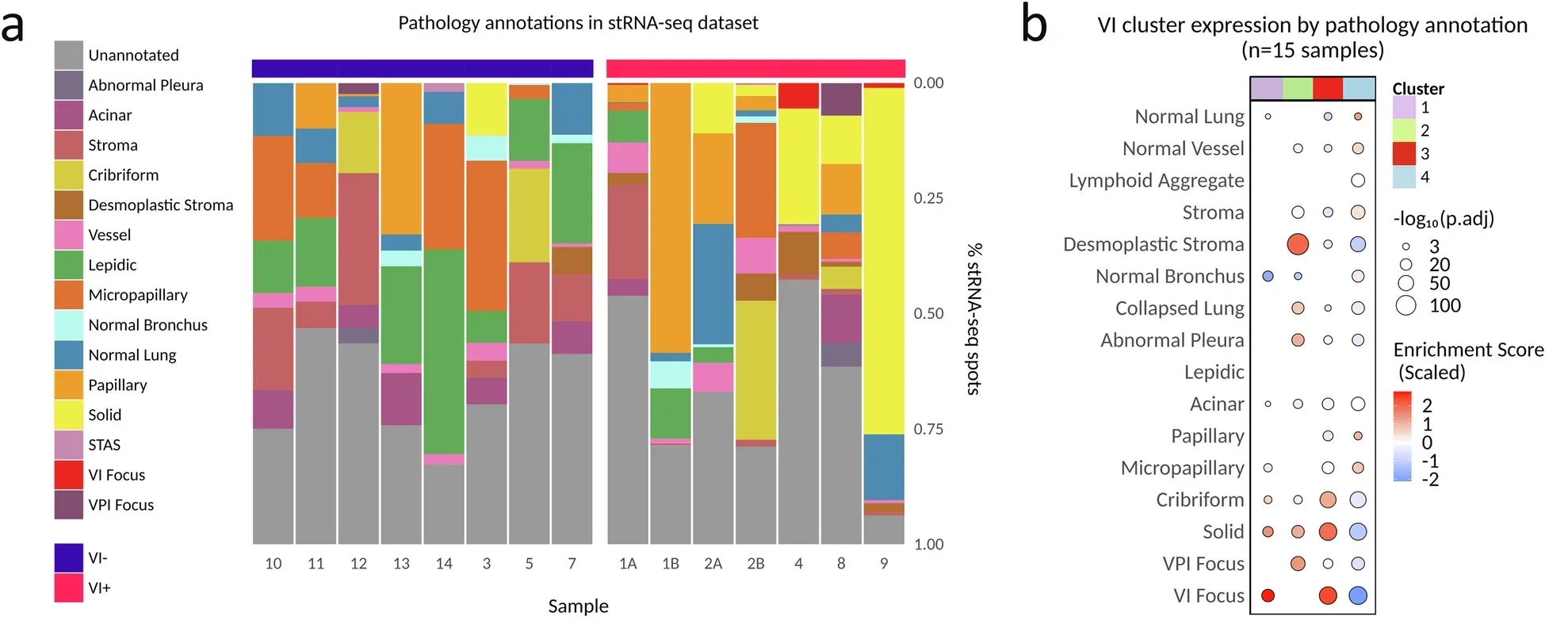
The team analyzed gene activity in tumors with and without vascular invasion. They identified more than 400 genes that behaved differently between the two groups. These genes formed a clear biological signature linked to aggressive tumor behavior.
The researchers then confirmed these patterns in a separate group of patients. That step strengthened confidence that the signal was real and not a statistical fluke.
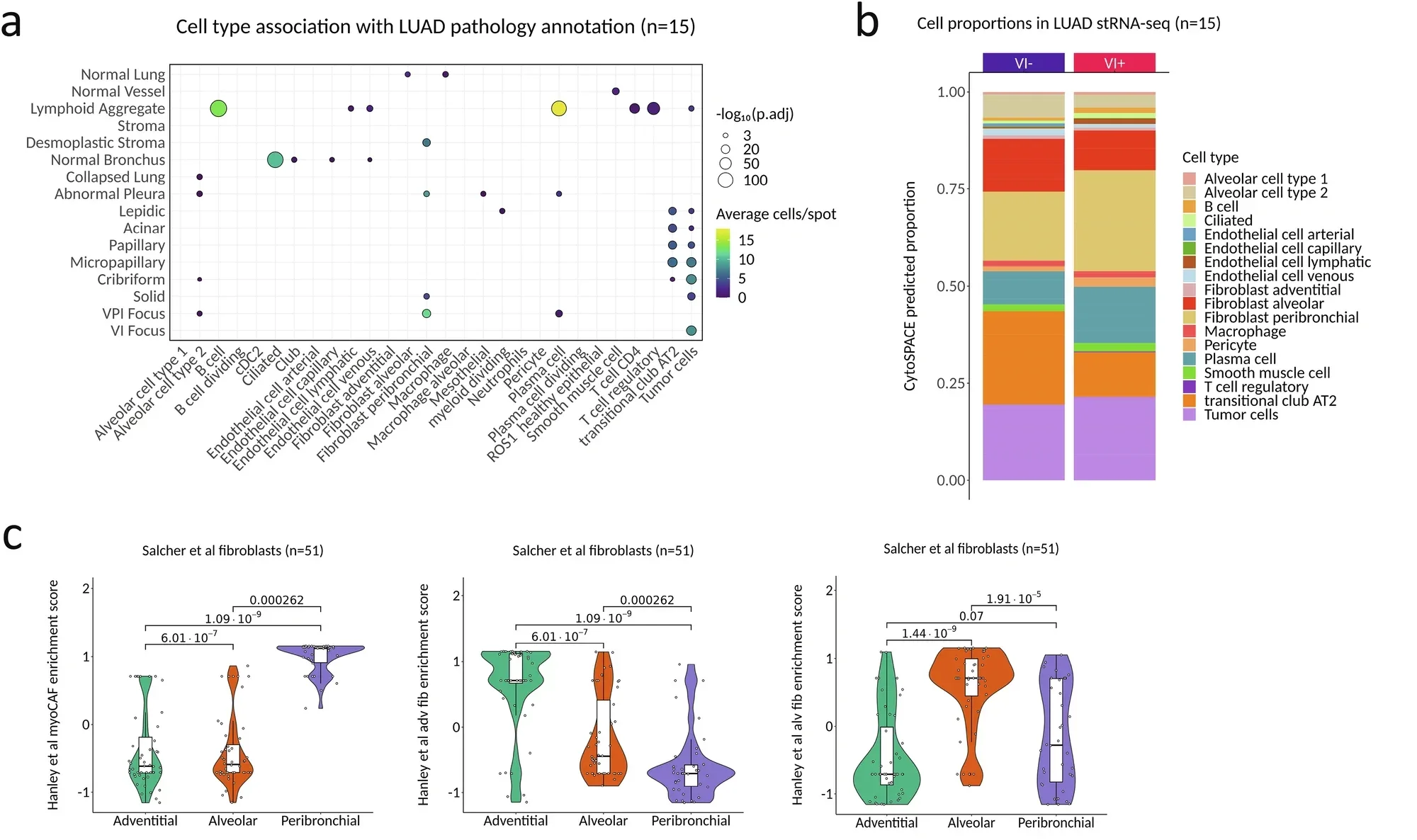
The genetic patterns revealed several key traits of aggressive tumors. Some genes showed increased activity related to rapid cell growth. Others pointed to changes in tissue structure and blood vessel formation.
Certain genes reflected how tumors adapt to low oxygen levels, a condition common in fast-growing cancers. At the same time, genes tied to protective pathways and immune response showed reduced activity.
Together, these changes paint a picture of a tumor that grows quickly, reshapes its surroundings, and weakens natural defenses. This combination may help explain why tumors with vascular invasion are more likely to spread and return.
The findings also suggest that vascular invasion is not a random event. Instead, it reflects a broader shift in tumor biology.
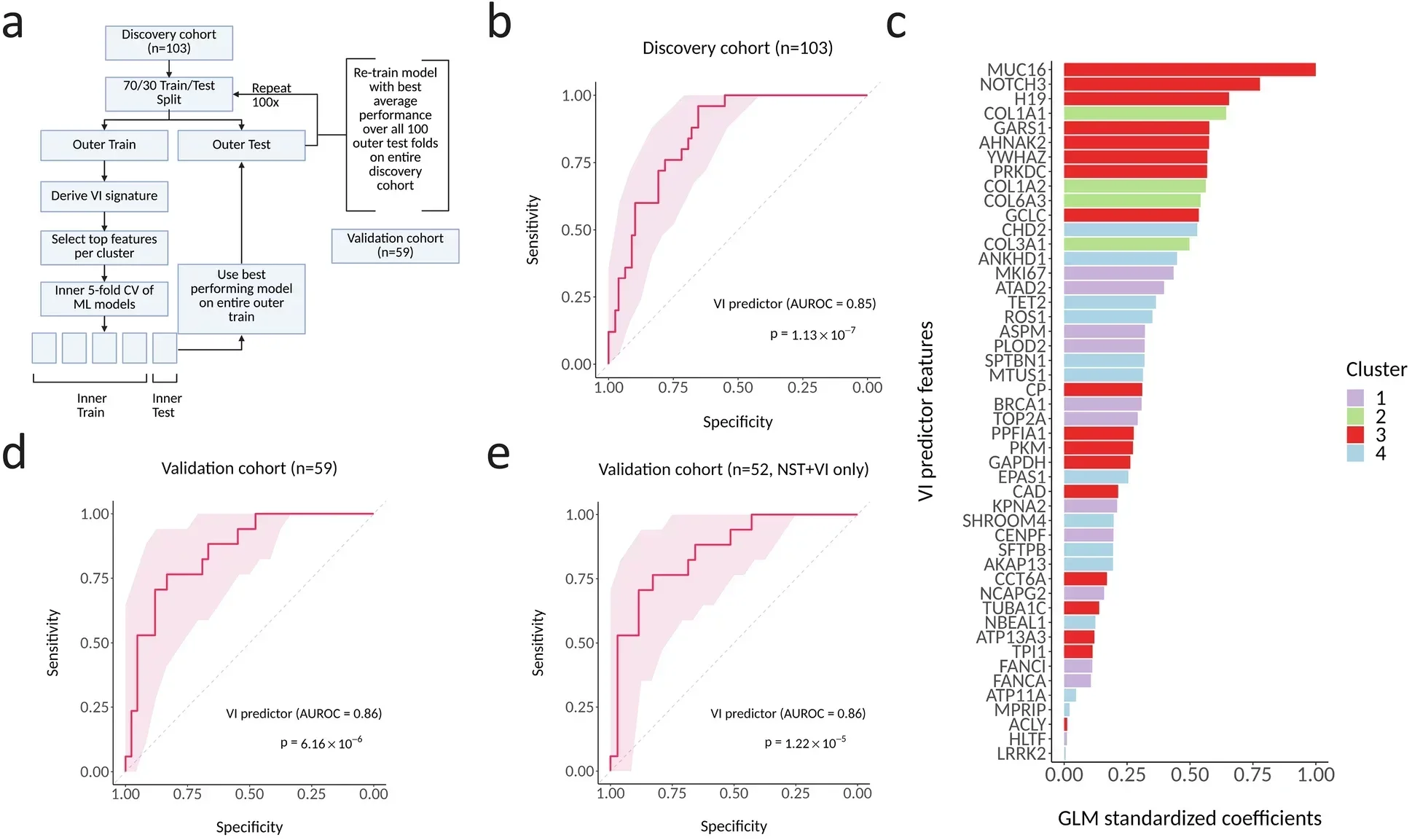
Identifying genes was only the first step. The researchers then built a machine learning model to predict whether vascular invasion is present.
They used a set of 48 genes to create the predictor. The model learned patterns from known cases and tested its accuracy on new data.
The results were strong. The model accurately predicted vascular invasion in multiple datasets. It also showed a clear link to tumor recurrence.
Most importantly, the test worked on small biopsy samples taken before surgery. These samples are usually too limited for traditional methods to detect vascular invasion.
That breakthrough means doctors could assess risk earlier in the treatment process.
“When lung cancer is detected early, there is a higher likelihood it can be cured,” Lenburg said. “We want to get the treatment right: we don’t want to undertreat an aggressive cancer and risk recurrence, but we also don’t want to over-treat a less aggressive cancer. The ability to know this prior to surgery will allow the surgeon to choose the right surgical approach.”
Surgical decisions in early lung cancer often involve trade-offs. Surgeons may choose between removing a small portion of lung tissue or performing a more extensive resection.
A limited surgery may preserve lung function. However, it may not fully address aggressive tumors. A more extensive procedure may reduce recurrence risk but comes with greater physical cost.
Without clear pre-surgical markers, these decisions rely on imaging, tumor size, and clinical judgment. The new genetic test could add a more precise layer of insight.
A patient with a low-risk profile might avoid unnecessary surgery. A patient with a high-risk profile could receive a more aggressive approach from the start.
That shift could improve survival while reducing overtreatment.
The implications may extend beyond lung cancer. Researchers note that vascular invasion is linked to poor outcomes in other cancers, including breast, liver, and gastric cancers.
The next step is to determine whether similar genetic patterns appear in those diseases. If so, the approach could support broader cancer care strategies.
The study also highlights the power of combining clinical insight with advanced data analysis.
“This project began years ago due to the persistent clinical challenge: we need a better way to identify high-risk tumors from just a biopsy,” said Kimberly Rieger-Christ, PhD, from Lahey Hospital & Medical Center, part of Beth Israel Lahey Health. “This multidisciplinary collaboration allowed us to turn that challenge into a new molecular approach that can screen early-stage lung cancer patients for the biology linked to vascular invasion. It’s a powerful example of how clinical insight and advanced analytics can come together to improve patient care.”
This research may reshape how doctors treat early-stage lung cancer. By identifying high-risk tumors before surgery, physicians can tailor treatment plans more precisely. Patients with aggressive tumors may receive more extensive surgery or additional therapy. Those with lower-risk tumors may avoid unnecessary procedures and recover more quickly.
The findings also support the growing role of molecular testing in routine care. A biopsy-based test could become part of standard evaluation for early lung cancer. This would allow earlier intervention and more personalized decisions.
For researchers, the study opens new questions about how tumors spread and evolve. It also provides a framework for applying similar methods to other cancers. Over time, this approach could improve survival rates and reduce uncertainty for patients facing treatment decisions.
Research findings are available online in the journal Nature Communications.
The original story “New genetic test predicts lung cancer spread before surgery” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post New genetic test predicts lung cancer spread before surgery appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.