Pain in the shoulder can linger long after a rotator cuff tear heals on paper. Even after surgery, strength may not return. Movement may stay limited. For many patients, the real problem is not the tear itself, but the scar that replaces what was lost.
Now, new research offers a deeper look into why that scar forms. Scientists from Xiangya Hospital at Central South University used advanced tools to map how tendon cells behave after injury. Their findings show that healing does not simply go wrong. Instead, it follows a complex path where multiple cell types actively build a scar.
“We were struck by how tendon cells that should support healing instead became drivers of fibrosis,” said Prof. Jianzhong Hu. “This helps explain why scarring persists even long after inflammation subsides.”
Rotator cuff injuries are among the most common shoulder problems. They often bring chronic pain and limit daily movement. Surgeons can reconnect torn tendons to bone. Still, recovery outcomes vary widely.
The issue lies in how adult tendons heal. Unlike fetal tendons, which can regenerate cleanly, adult tendons rely on scar formation. This scar tissue lacks the organized structure of healthy tendon. It is weaker, less flexible, and more prone to re-injury.
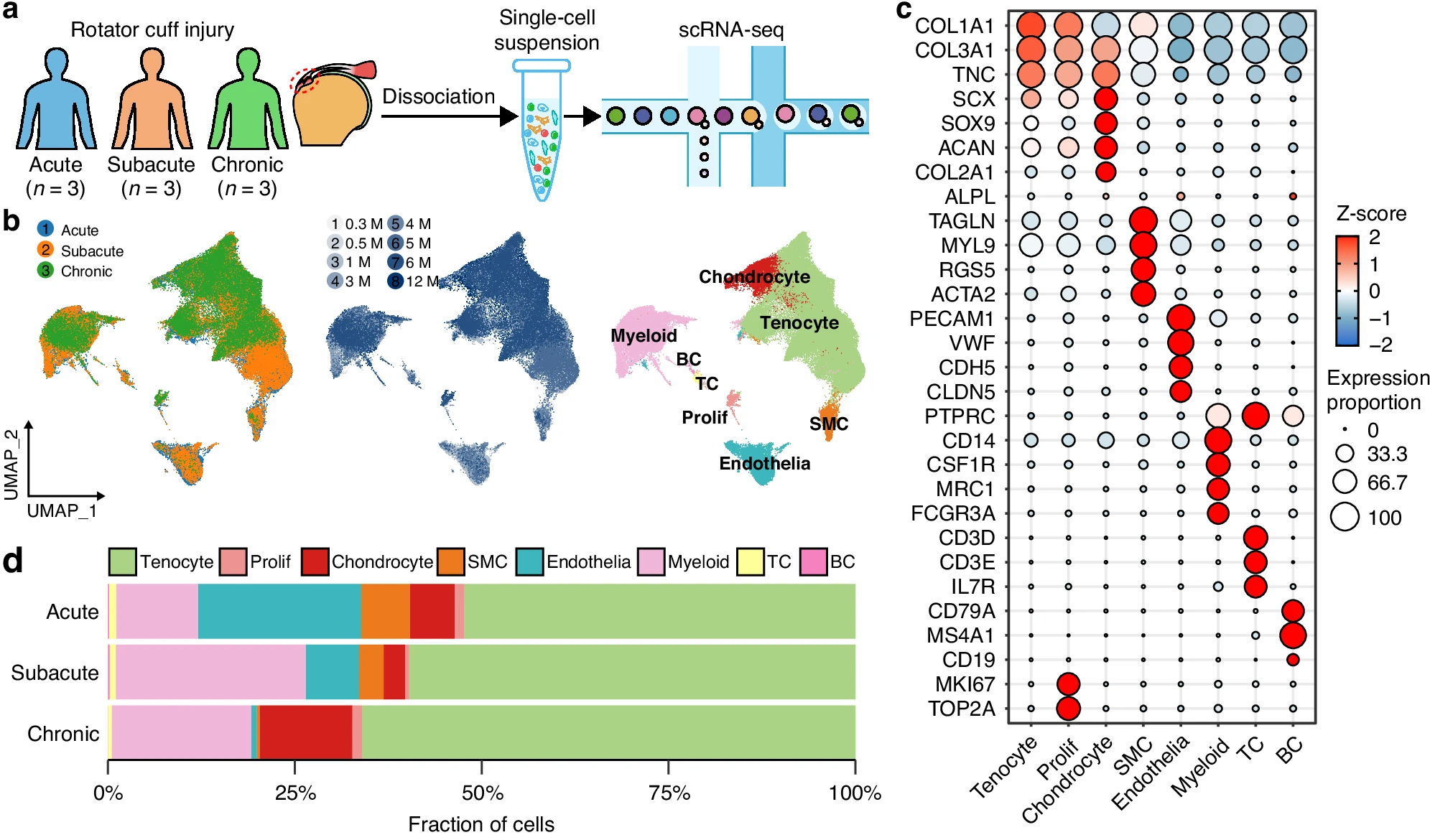
To understand why this happens, researchers examined tendon samples from patients at different stages of injury. They combined traditional imaging with single-cell RNA sequencing. This allowed them to study nearly 90,000 individual cells.
The result was one of the most detailed views of tendon healing to date.
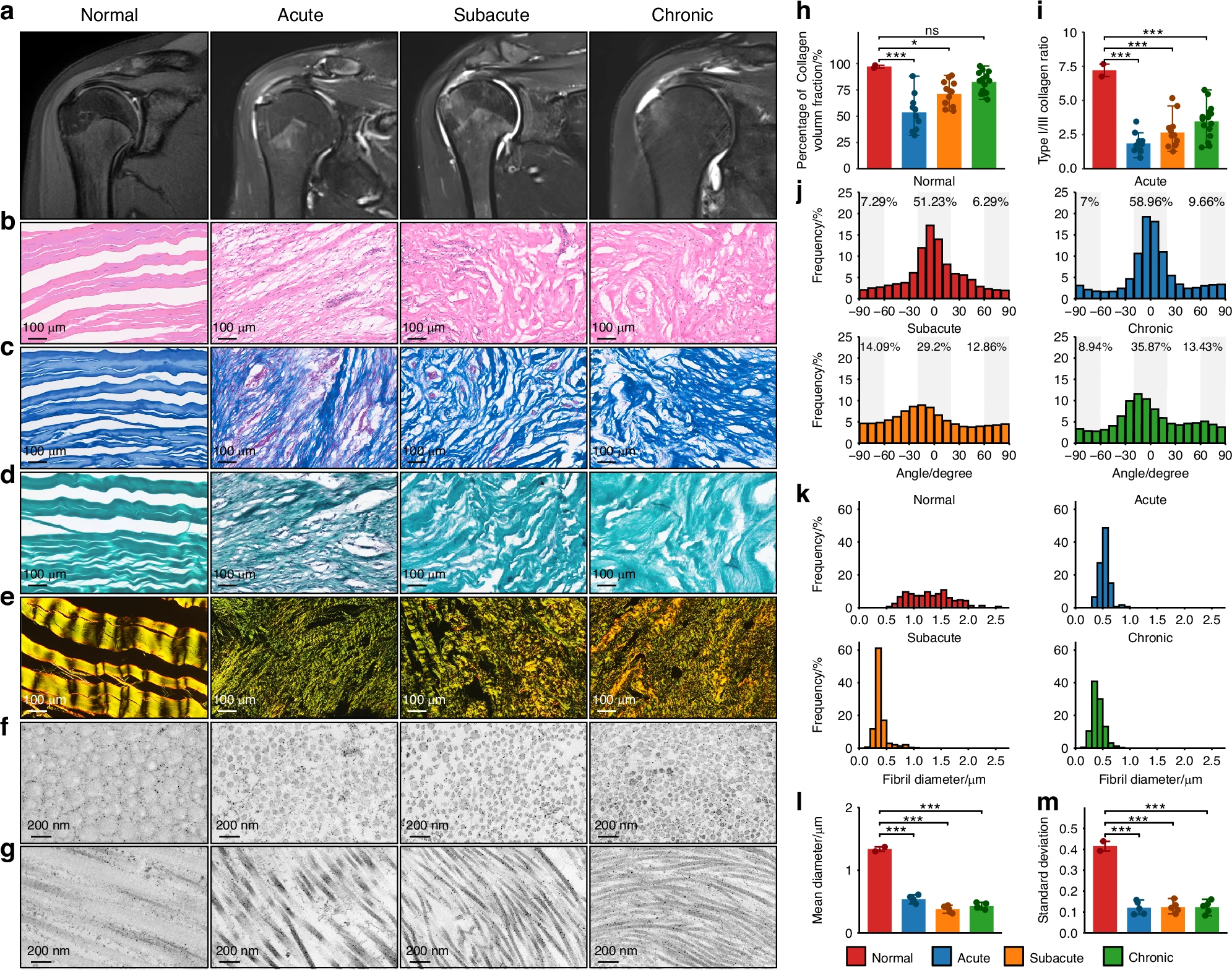
Under the microscope, injured tendons looked very different from healthy ones. In normal tissue, collagen fibers align in tight, parallel patterns. After injury, that structure breaks down.
The researchers found that collagen fibers became disorganized early on. They also saw a shift in collagen types. Healthy tendon contains mostly type I collagen. After injury, type III collagen increased sharply.
This imbalance persisted even in later stages. Collagen fibers also became thinner than normal. These changes weaken the tendon and reduce its ability to handle stress.
Even months after injury, the tissue did not return to its original form. The structure remained altered, mirroring the long-term symptoms many patients experience.
At the center of the study lies a surprising discovery. Cells that should help repair the tendon instead drive scar formation.
Tendon stem cells and progenitor cells usually mature into functional tendon cells. In this study, they failed to complete that process. Instead, they stayed in an active state and kept producing extracellular matrix material.
This continuous production added to scar buildup.
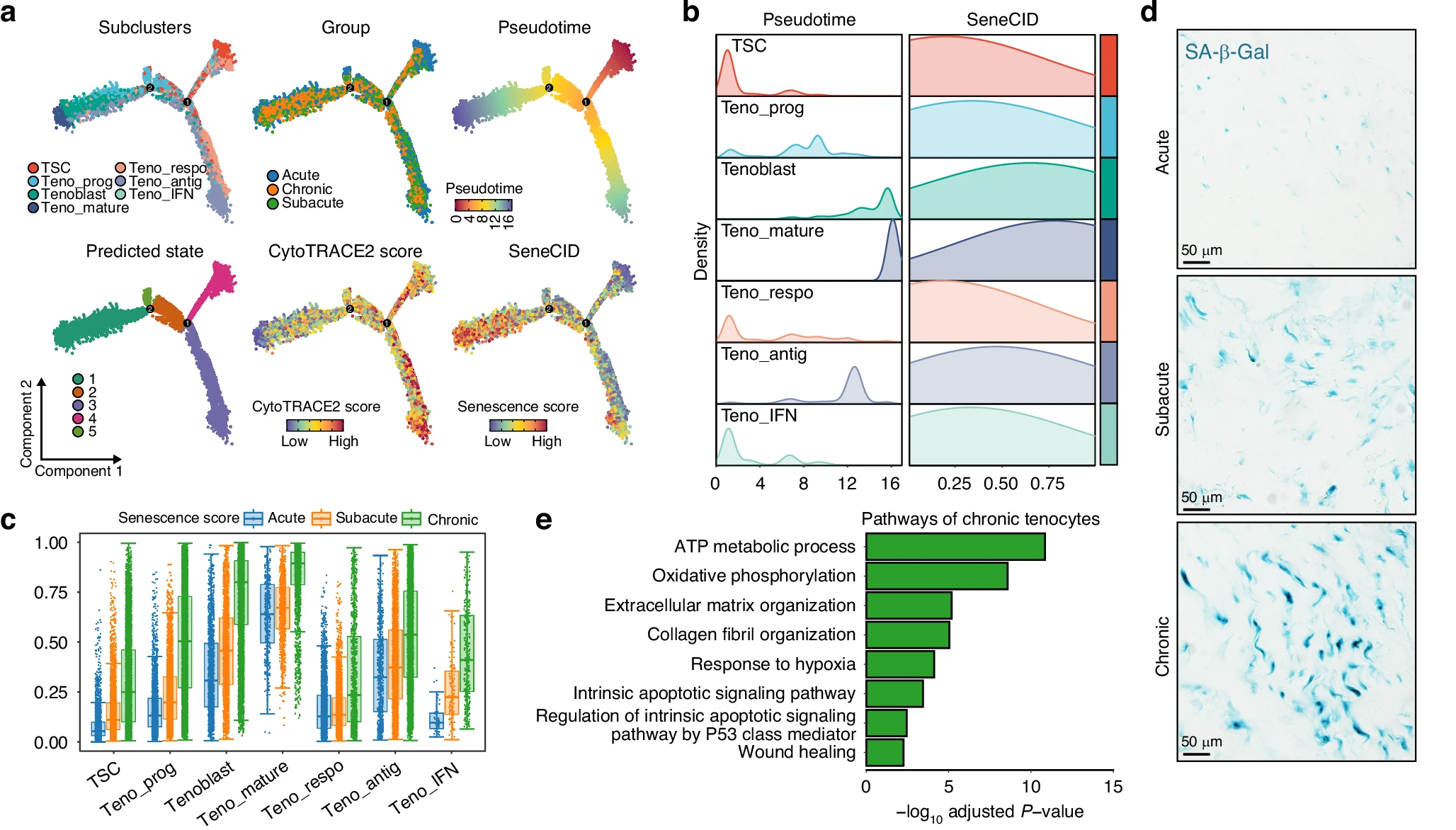
At the same time, many mature tendon cells entered a senescent state. These aging cells stopped dividing and lost their ability to repair tissue. Yet they remained active in other ways.
They released signals that encouraged more collagen production. This created a feedback loop that reinforced scarring.
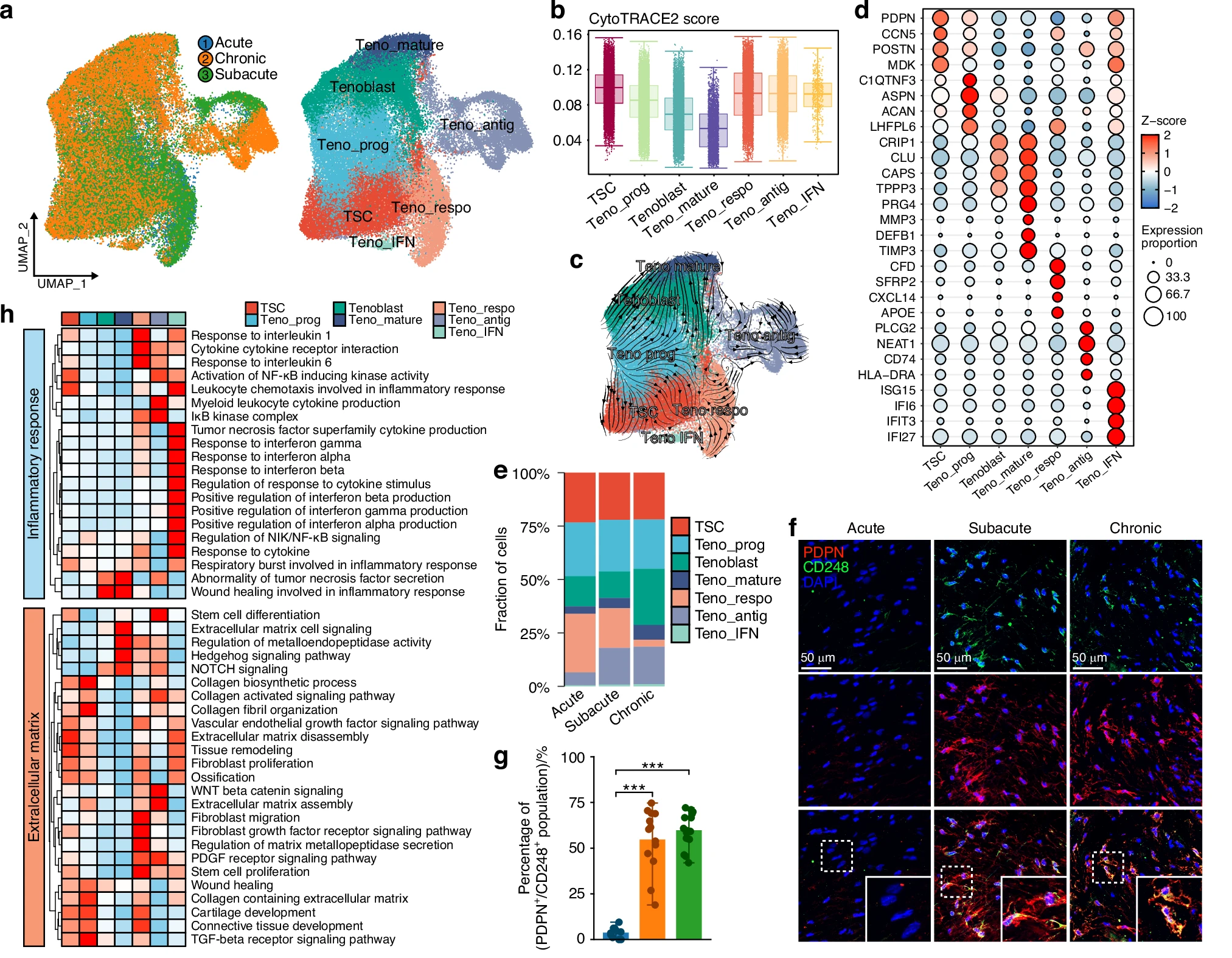
The combination of stalled stem cells and aging tendon cells helps explain why healing becomes stuck in a fibrotic state.
The immune system also plays a key role. In early stages, immune cells respond to injury and inflammation. Over time, most of these cells decline.
Macrophages, however, remain.
The study found that macrophages undergo a major transformation. Instead of simply responding to damage, they begin producing collagen themselves. This makes them direct contributors to scar formation.
“These macrophages are not just bystanders,” said Prof. Hongbin Lu. “They actively build scar tissue, creating a self-sustaining fibrotic environment that is difficult to reverse.”
This shift is driven by a regulatory factor known as SOX9. It reprograms macrophages into cells that produce matrix components. Once this change occurs, scar formation becomes harder to stop.
Blood vessels, which briefly expand after injury, also play a part. Early in healing, new vessels form to support repair. Later, their numbers decline.
During this transition, some vascular cells change identity. They take on features of scar-producing cells. This process adds another layer to the fibrotic environment.
The researchers linked this shift to signaling pathways tied to tissue growth and repair. These pathways, while helpful in early healing, appear to contribute to long-term scarring when left unchecked.
The study also identified two major signaling pathways that drive scar formation. One involves osteopontin, a protein produced by immune cells. The other involves transforming growth factor beta, a well-known regulator of tissue repair.
Together, these signals encourage cells to produce more collagen and matrix material.
In laboratory tests, exposing tendon cells to these signals increased scar-related proteins. Blocking these pathways reduced collagen production and limited scar formation.
In animal models, treatments targeting these signals led to less dense scar tissue. The effect was strongest when both pathways were blocked at the same time.
These findings suggest that fibrosis is not random. It is guided by specific molecular signals that could be targeted in future therapies.
By combining structural analysis with single-cell data, the researchers created a detailed map of tendon healing. The picture that emerges is not simple.
Multiple cell types contribute to scar formation. Stem cells fail to mature properly. Aging cells lose repair function. Immune cells switch roles. Vascular cells join the process. Signaling pathways amplify these effects.
Together, these changes create a self-reinforcing environment that favors scar over regeneration.
The study also highlights limits. The number of patient samples was modest. Some control tissue came from a different tendon type. Still, the findings offer a strong foundation for future research.
This research could change how rotator cuff injuries are treated in the future. Right now, surgery focuses on reconnecting tendon to bone. It does not address the biological processes that lead to scarring. By identifying the cells and signals responsible for fibrosis, this study opens the door to therapies that target healing itself.
In the short term, drugs that block osteopontin or TGF-β signaling could be used alongside surgery. These treatments may reduce scar formation and improve long-term outcomes. Patients could experience stronger tendons, better mobility, and lower risk of re-tear.
Over time, the findings may support regenerative approaches. Instead of forming scar tissue, therapies could guide cells to rebuild healthy tendon. This would represent a major shift in orthopedic care.
The impact may extend beyond shoulder injuries. Similar fibrotic processes occur in organs such as the heart, lungs, and liver. Understanding how cells drive scarring at this level could inform treatments across many diseases.
By mapping healing at single-cell resolution, the study brings science closer to a future where tissues repair themselves fully, not imperfectly.
Research findings are available online in the journal Bone Research.
The original story “New study findings could change how rotator cuff injuries are treated in the future” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post New study findings could change how rotator cuff injuries are treated in the future appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.