Diamonds are famous for their strength, but scientists have long suspected that another form of diamond might be even harder.
Evidence of this was gathered over the past sixty years in meteorite fragments, where the mineral lonsdaleite appeared. Lonsdaleite was theorized to have formed due to immense impact pressures on carbon-rich rocks from cosmic impacts. However, the evidence supporting this theory has generally been inconclusive due to its small size and contamination with other carbon forms.
Additionally, there has been some skepticism about whether hexagonal diamond exists as an actual mineral.
Recent experimental results indicate that lonsdaleite is real and can be manufactured in a pure form. Shoulong Lai and his research team from Henan Key Laboratory of Diamond Materials and Devices created lonsdaleite in the laboratory using extreme temperatures and pressures, allowing them to create a sample large enough to perform chemical and physical characterization.

The results of this research were published in the journal Nature. The information presented in this paper provides supporting evidence for previous theoretical studies that suggested hexagonal diamond could be a stronger form of diamond.
The structure of hexagonal diamonds differs from conventional diamonds in that carbon atoms stack in a hexagonal arrangement rather than a cubic arrangement. This difference results in differing characteristics.
Proving the existence of this new form of diamond has been difficult. Scientists have attempted to produce lonsdaleite using shockwave and high-pressure techniques since its discovery in the Canyon Diablo meteorite. These efforts had only been able to produce a mixture of graphite, cubic diamonds, and other forms of carbon. There was still doubt as to whether researchers had actually detected pure hexagonal diamond because the diffraction patterns were similar.
Some previous studies theorized that researchers were simply observing cubic diamonds containing minor imperfections.
Recreating diamond from graphite has now been accomplished by using a specialized carbon material referred to as highly oriented pyrolytic graphite. This carbon material has flat layers of carbon atoms that are evenly oriented in one direction. This structure allows researchers to employ compression techniques on the material.
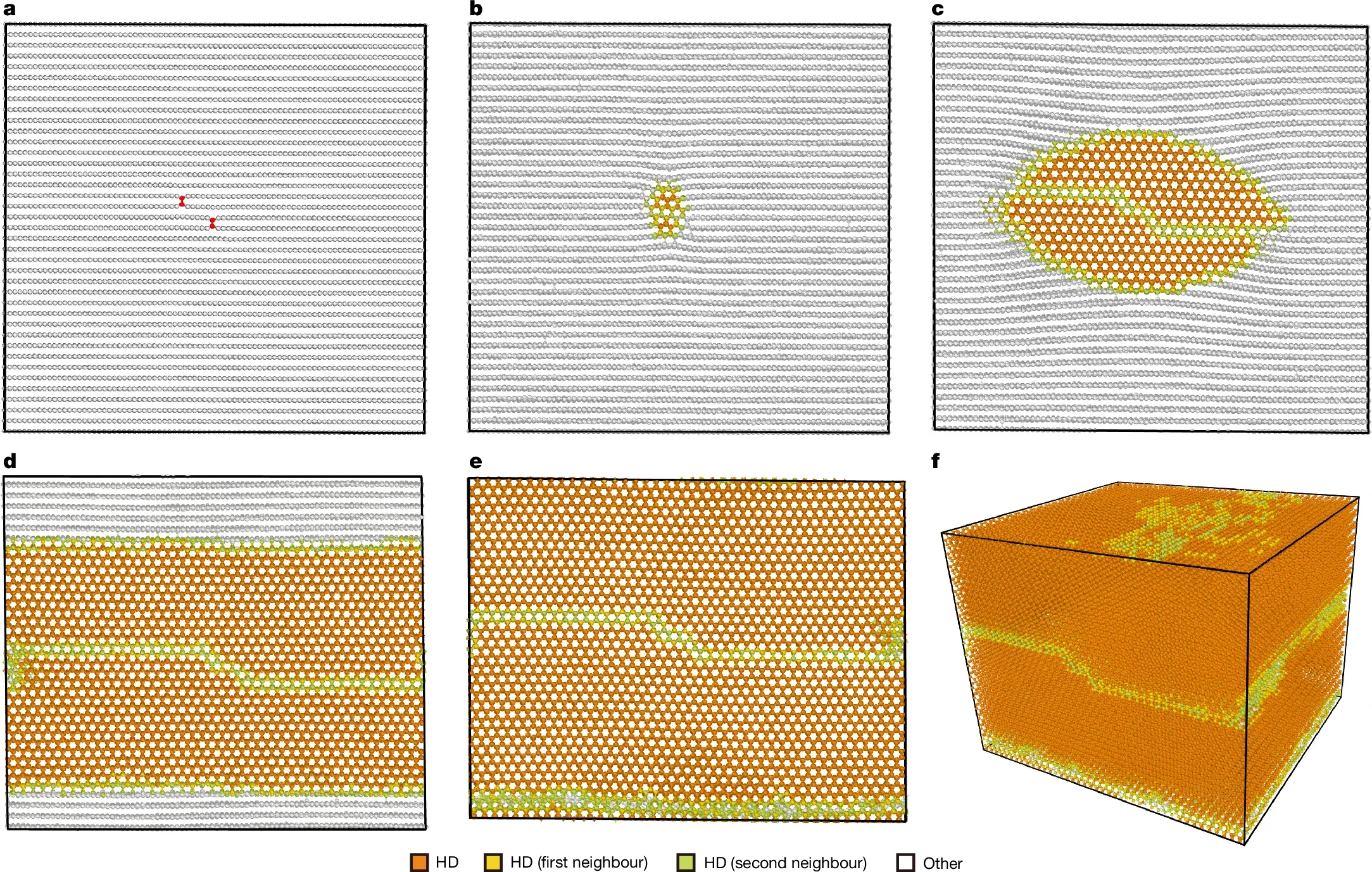
The graphite was subjected to very high temperatures and pressures (20 gigapascals, or about 200,000 times atmospheric pressure). The temperature range was 1,300 °C to 1,900 °C.
The method used to apply pressure was the most important factor in this process. The c-axis direction of graphite was compressed, meaning that the compressive force was applied directly toward the stacked layers of carbon.
Through these high temperatures and pressures, the layers of carbon atoms realigned themselves. The result was the production of millimeter-sized hexagonal diamonds.
There were multiple methods used to verify the nature of this new carbon material.
The first method employed synchrotron X-ray diffraction (XRD) to measure the crystal structure. This method is based on the scattering of X-rays through the atomic planes of the crystal. The intensity of X-rays scattered from a crystal displays specific characteristics, or peaks, according to the crystal structure. The peaks generated from the material matched those of a hexagonal diamond structure.
These peaks match the P63/mmc structure and indicate that this material is hexagonal in nature, rather than a combination of two carbon forms.
Another technique used to observe this new material included atomic-resolution scanning and transmission electron microscopy. The microscope images revealed the ABAB stacking pattern characteristic of hexagonal diamond.

When viewed from another perspective, the hexagonal lattice of atoms exhibited sixfold symmetry.
Another key difference was where the six-membered rings of carbon had empty centers in the hexagonal structure. However, when cubic diamond was viewed along specific directions, a line of atoms could be observed running through its center.
Across all the areas that were analyzed by the researchers, there was no evidence of contamination by either graphite or cubic diamond.
To better understand how graphite was transformed into hexagonal diamond, the researchers performed extensive molecular dynamics simulations.
These simulations indicate that interlayer bonding between neighboring graphite layers occurs first. This process creates nucleation sites for the initial growth of the hexagonal arrangement.
Additionally, interlayer bonding inhibits the individual layers of carbon from moving freely relative to each other.

This relative movement leads to the formation of cubic diamonds. However, prohibiting this movement creates an environment conducive to stabilizing the hexagonal stacking pattern.
The simulations also indicate that when the initial graphite layers become disordered, they typically result in the formation of cubic diamonds rather than the hexagonal phase.
After the material was formed, researchers evaluated its mechanical properties.
Using the Vickers hardness test, a diamond-embedded indenter was pressed into the material to quantify its resistance to deformation.
The hardness value of the hexagonal diamond was determined to be approximately 114 gigapascals. Natural diamonds are reported to have hardness values of approximately 110 gigapascals.
Additionally, researchers measured the stiffness of the newly created material with the aid of ultrasonic evaluations and nanoindentation testing. The Young’s modulus of the hexagonal diamond material was reported at 1,229 gigapascals. A single crystal of cubic diamond was used as a reference for comparison, and its Young’s modulus was measured at 1,087 gigapascals.

The thermal testing indicated that the hexagonal diamond material begins to oxidize above approximately 1,121 kelvin. This suggests a high degree of thermal stability at elevated temperatures.
The capability to produce bulk quantities of hexagonal diamond would present new commercial opportunities in manufacturing.
A material that possesses high levels of hardness combined with significant stiffness and thermal stability may be used to construct high-quality cutting tools and abrasives. It could also be used in high-tech components for high-performance electronics.
Additionally, this research highlights unusual forms of carbon that result from meteorite impacts, where intense pressures can rapidly transform graphite into new crystal structures.
If these findings are confirmed through future research and if bulk production increases, hexagonal diamond may represent one of the hardest and most durable carbon-based materials ever manufactured.
Here are three of the most interesting aspects of lonsdaleite — how it forms, how its structure differs from diamond, and why researchers care about it:
Lonsdaleite is essentially a product of sudden, catastrophic pressure. When a meteorite slams into Earth, pressures can exceed tens of gigapascals in microseconds. Under those conditions, graphite doesn’t have time to slowly rearrange into ordinary diamond. Instead, its carbon atoms are forced into a different configuration almost instantly.
That’s why lonsdaleite is often found in impact-related materials like the Canyon Diablo meteorite. The key idea is speed:
Recent studies suggest it may not always form as a perfectly pure crystal. Instead, it can appear as stacking sequences or intergrowths between diamond and graphite layers. This has led scientists to rethink whether lonsdaleite is always a distinct mineral or sometimes a transitional structure formed under extreme strain.
Both diamond and lonsdaleite are made entirely of carbon atoms bonded in a tetrahedral arrangement. The difference comes down to how those layers stack:
That sounds minor, but it changes how stress moves through the material. In the hexagonal version:
In simple terms, the same atoms are arranged with a slightly different rhythm, and that rhythm affects how the material behaves under pressure, strain, and fracture.
Lonsdaleite has drawn attention because it hints at a new class of engineered supermaterials. If scientists can reliably produce it—or structures like it—it could lead to:
What’s especially interesting is that researchers are no longer just trying to make “pure lonsdaleite.” Instead, they’re exploring hybrid carbon materials that mimic its hexagonal stacking or combine it with diamond at the nanoscale.
There’s also active work on synthesizing it through:
If those methods scale, lonsdaleite-inspired materials could move from rare geological curiosity to something used in real-world engineering.
Research findings are available online in the journal Nature.
The original story “Pure hexagonal diamonds are real and harder than natural diamonds” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Pure hexagonal diamonds are real and harder than natural diamonds appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.