Blood clotting depends on speed, force, and timing. Platelets have to stay quiet while circulating through the body, then switch into action fast when a blood vessel breaks. A new look inside that system shows, in unusually fine detail, how one of its key molecular motors stays shut down until the moment it is needed.
Researchers at the University of Leeds have visualized the structure of platelet myosin, the form of myosin found in platelets, and mapped how it folds into an inactive state. Their findings, published in Science Advances, also point to regions where inherited mutations can upset that balance, helping explain bleeding disorders as well as some forms of deafness and kidney disease.
The work centers on nonmuscle myosin 2A, or NM2A, the only class 2 myosin isoform found in platelets. Platelets are small blood cell fragments that rush to an injury, stick together, and help seal damaged tissue. To do that, they rely on an internal scaffolding, the cytoskeleton, and on NM2A working with actin to pull and compact the growing clot.
What had remained unclear was how that motor keeps itself inactive before clotting begins.
Professor Michelle Peckham, from the University of Leeds School of Molecular and Cellular Biology, put the problem this way: “By understanding how this molecule is normally kept under control, we can begin to see how genetic mutations push it out of balance and lead to disease.”
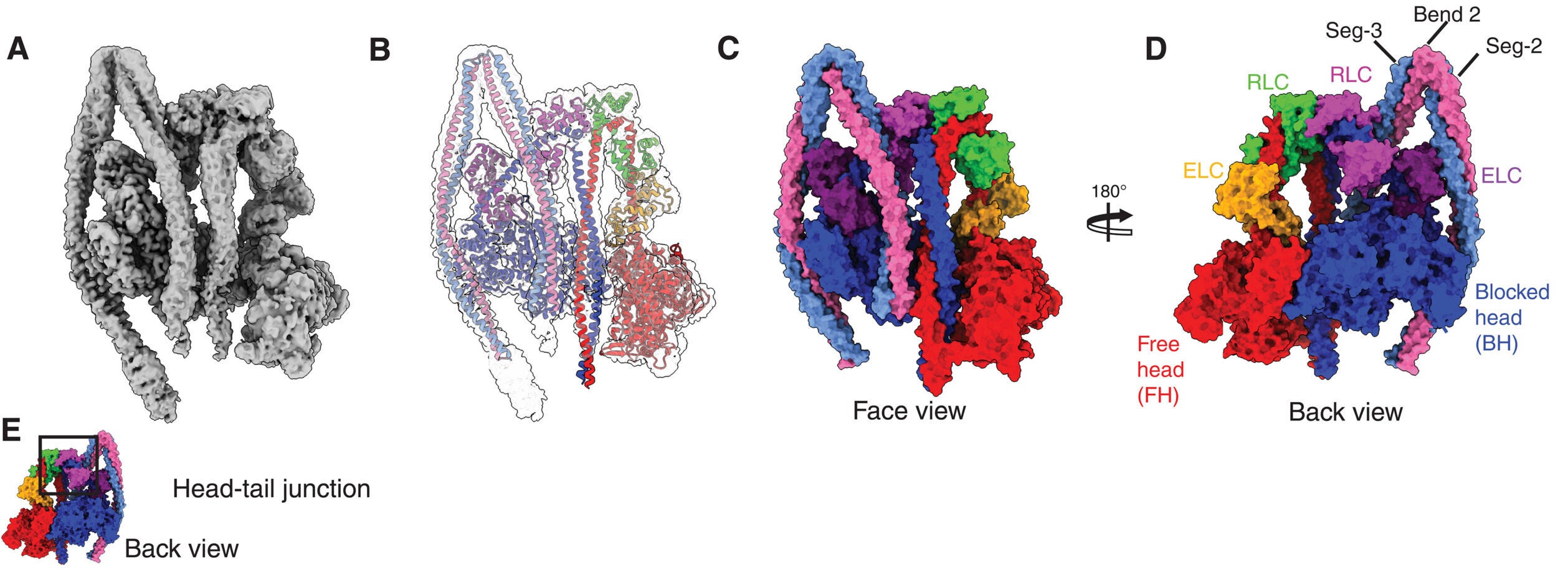
Using cryo-electron microscopy, the Leeds team solved the structure of dephosphorylated human NM2A to a global resolution of 3.0 angstroms in the head region. The overall structure showed the molecule in what the authors describe as a shutdown state, with its tail wrapped around the heads and its activity held very low.
That shutdown form matters. In active platelets, NM2A helps drive filament formation and platelet spreading, both central to clot formation. But in circulating platelets, the molecule must stay restrained until a chemical trigger flips it on.
The new structure shows several parts of the protein working together to create that restraint. The researchers identified regions in the head-tail junction, the regulatory light chains, and multiple segments of the coiled-coil tail that help lock the motor into place. One especially important region, called the “mortar,” helps cement two regulatory light chains together. Another, called the “latch,” appears to help hold part of the tail in its shutdown position.
Glenn Carrington, a postdoctoral researcher at the Astbury Centre for Structural Molecular Biology, said: “What’s exciting is that we can now explain, in structural detail, how this molecule folds in on itself to stay inactive, and how a simple chemical change flips it back on.”
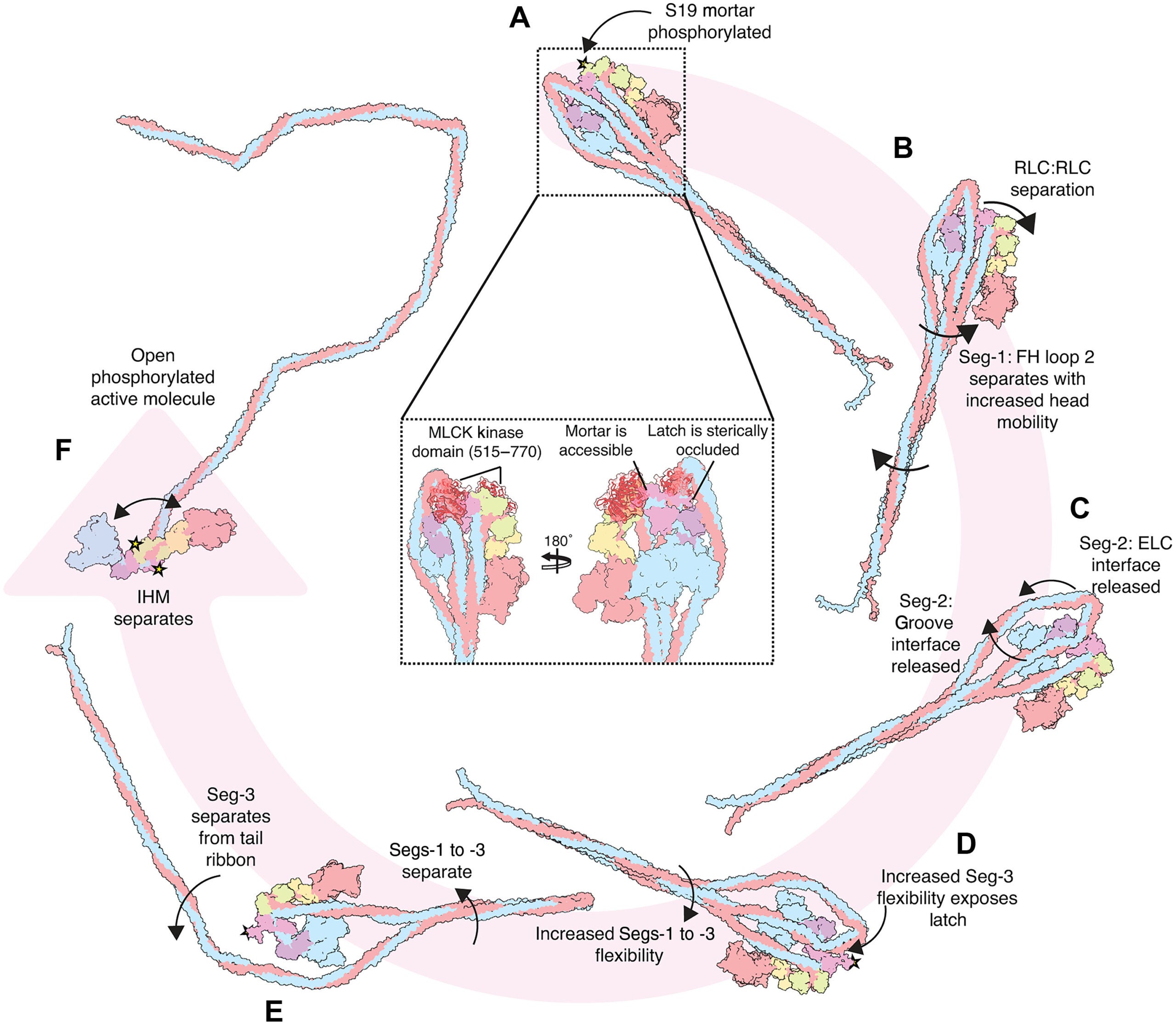
That chemical change is phosphorylation of the regulatory light chain, mainly at a site called Ser19. The analysis suggests that one Ser19 site, on the free head side of the molecule, is more accessible and likely gets phosphorylated first. That first step appears to loosen the mortar, increase motion at the head-tail junction, and begin destabilizing the compact shutdown form. A second phosphorylation event would then help release the latch, allowing the folded molecule to open into an active state.
The structure also gives the disease story sharper edges.
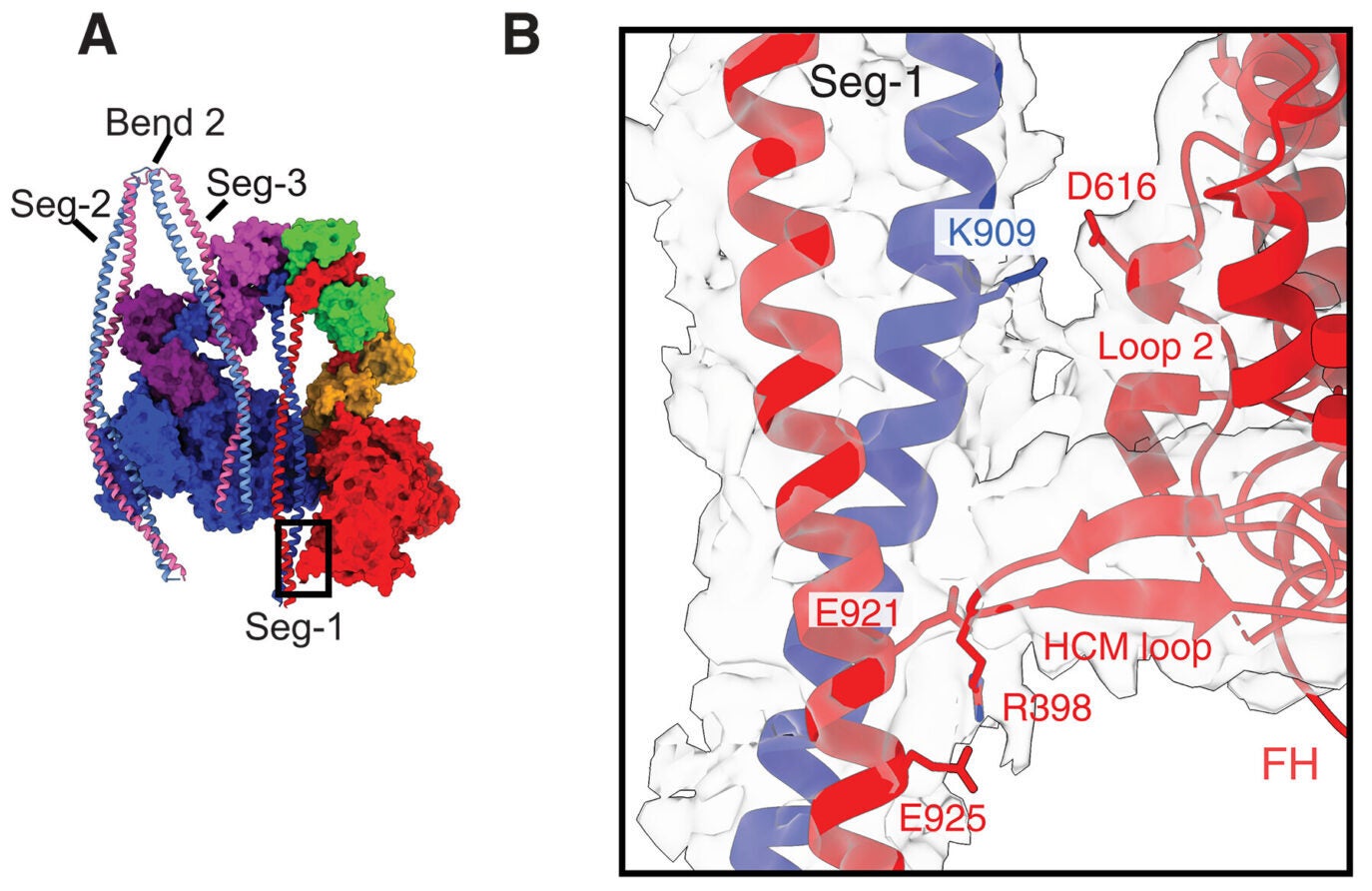
More than 200 autosomal dominant missense mutations have been reported in MYH9, the gene that encodes the NM2A heavy chain. Those mutations are linked to bleeding disorders, kidney abnormalities, hearing defects, and other problems. By solving the shutdown structure, the authors could see where many of those mutations sit and how they may interfere with the motor’s normal control system.
Several of the mutation hotspots appeared in regions that stabilize the inactive form. In one groove of the blocked head motor domain, for example, the team identified 11 interacting residues, 8 in the motor and 3 in the tail, where missense mutations have been tied to MYH9-related disease. According to the analysis, changes in these positions could weaken the shutdown state.
That matters because the clotting problem may not come from a motor that fails to work at all. It may come from one that activates at the wrong time or cannot return cleanly to the state platelets need before they build their cytoskeleton. The authors say such destabilization could make myosin prematurely active, disrupt normal platelet function, reduce the cells’ ability to assemble their internal framework when needed, and weaken clot formation.
In other words, a bleeding disorder may begin with a control problem, not just a force problem.
The study does more than describe a static structure. It proposes a sequence for how the molecule shifts from a folded 10S shutdown state to an open, active form.
In that model, phosphorylation first weakens the mortar. That increases mobility at the head-tail junction and in the tail segments. The added motion helps release another tail segment from a groove in the motor domain and from contacts with the essential light chain. Once the latch becomes exposed, a second phosphorylation event can occur, leading to release of the tail ribbon and full activation of the motor.
The researchers also compared NM2A with other myosin forms, including smooth muscle myosin and beta-cardiac myosin. Some parts of the shutdown design were similar, but the Leeds team also found marked differences in how tail segments run across the molecule and how key junctions are arranged. Those differences help explain why platelet myosin behaves as it does and why models based on other myosins cannot fully stand in for it.
Not every part of the protein could be resolved equally well. The distal region of the full-length molecule was highly mobile, which prevented the team from solving the entire structure directly from the density map. To extend the model, they used AlphaFold to build missing coiled-coil segments and cross-linking mass spectrometry to validate that model. The authors also note that some interpretations, including the sequence of phosphorylation events, come from what their data indicate rather than from direct observation of every intermediate step.
That still leaves the field with something it did not have before: a detailed structural explanation for how platelet myosin stays off, how it turns on, and how disease-linked mutations may throw that process off balance.
This work gives researchers a clearer framework for interpreting inherited mutations in MYH9 and for understanding why some of them lead to bleeding disorders, hearing defects, or kidney problems. It also provides a structural map of the residues and interfaces that keep NM2A in its inactive form, which could guide future efforts to study platelet dysfunction at the molecular level.
For clotting biology, the importance is basic but substantial. Platelets depend on precise timing, and this study shows where that timing is built into the molecule itself. By exposing the protein’s internal brake system, the findings offer a more concrete starting point for studying what happens when that brake fails.
Research findings are available online in the journal Science Advances.
The original story “Research breakthrough reveals the secret behind blood clotting” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Research breakthrough reveals the secret behind blood clotting appeared first on The Brighter Side of News.