Brown fat has long carried a kind of scientific promise. Unlike the more familiar white fat that stores energy, this darker tissue burns it, turning fuel into heat when the body needs to stay warm. That ability has made it a target for researchers searching for new ways to address obesity.
Yet a basic question has lingered. What allows brown fat to actually do its job?
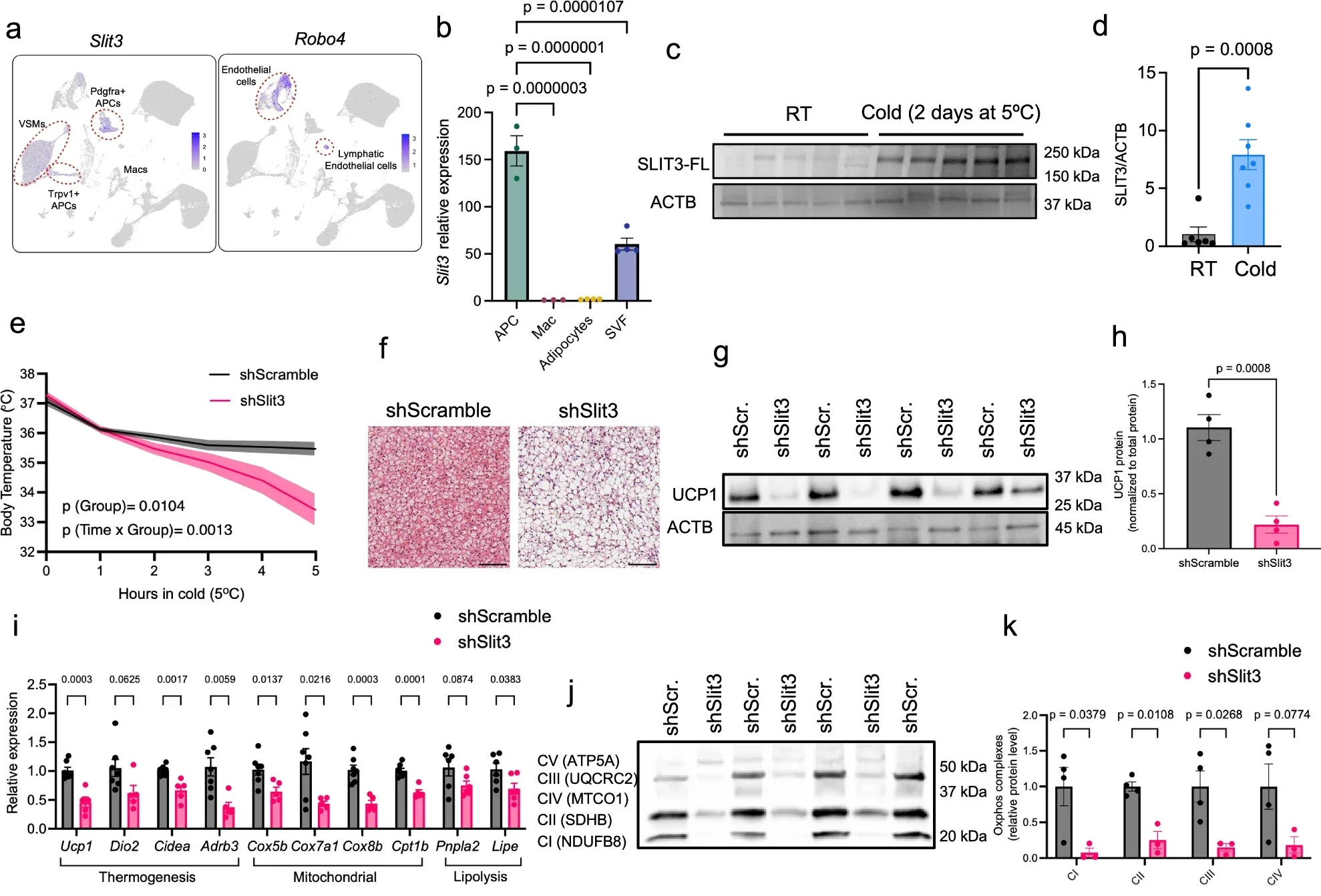
A study involving researchers at NYU College of Dentistry points to an answer that sits outside the fat cells themselves. The work centers on a protein called SLIT3, which appears to build the internal support system that lets brown fat function as a calorie-burning tissue. Without that support, the cells are present, but the process falters.
“By rapidly taking up and using fuel sources from our bodies and the food that we eat, brown fat acts like a metabolic sink that draws in nutrients and prevents them from being stored,” said Farnaz Shamsi, assistant professor of molecular pathobiology and the study’s senior author. The finding shifts attention from what brown fat is to how it is wired.
Most fat in the human body stores excess energy. That role belongs to white fat, which expands when calorie intake exceeds what the body uses. Brown fat behaves differently. It activates under cold conditions, pulling in glucose and lipids and converting them into heat through thermogenesis.
This process depends on speed. Signals from the brain must reach the tissue quickly. Oxygen and nutrients must arrive just as fast. Heat must then circulate through the body. That requires infrastructure.
Brown fat contains dense networks of blood vessels and nerves. The nerves relay signals from the brain, especially when temperature drops. Blood vessels deliver fuel and oxygen, then carry heat away. Earlier research focused largely on the fat cells themselves, asking how to make them burn more energy. The underlying networks received less attention. Shamsi’s group took a different approach. They looked for signals produced within the tissue that might organize these systems.
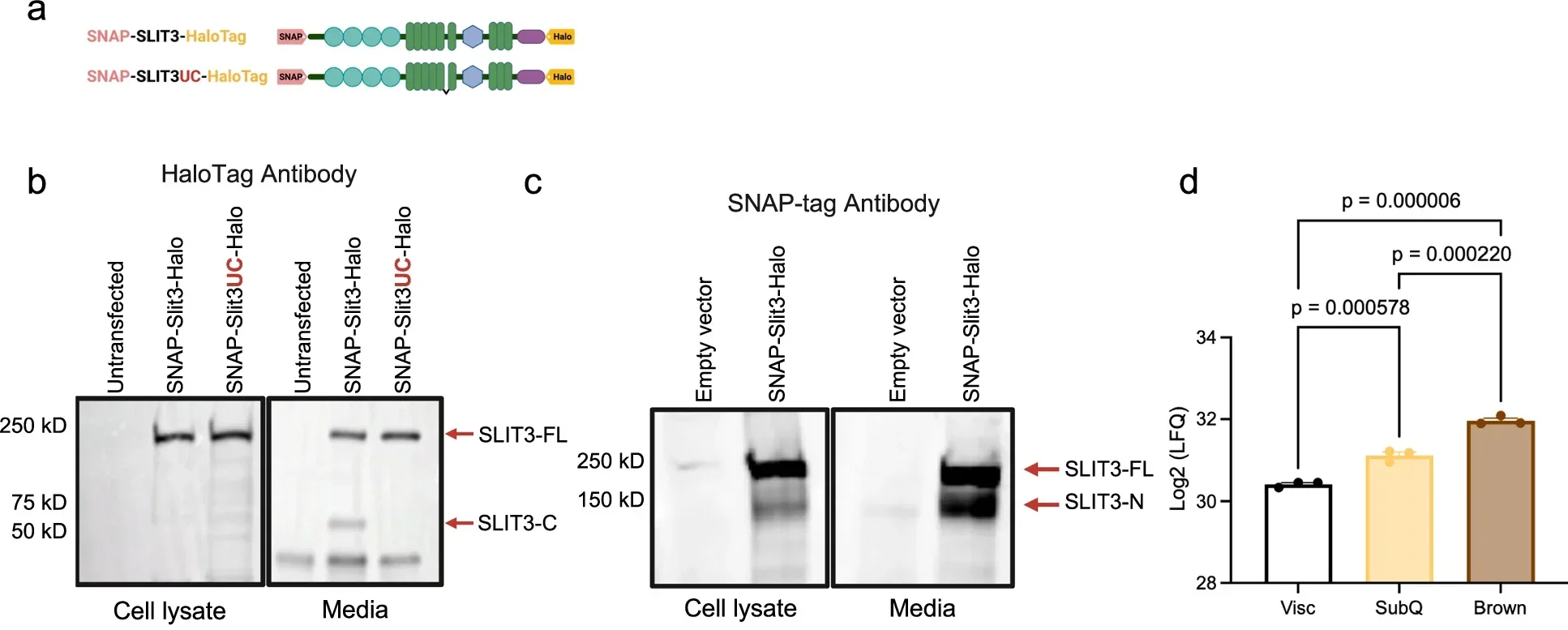
The story begins with SLIT3, a protein secreted by brown fat cells. Earlier work using single cell RNA sequencing had flagged it as a possible player in cell communication. What it actually did remained unclear.
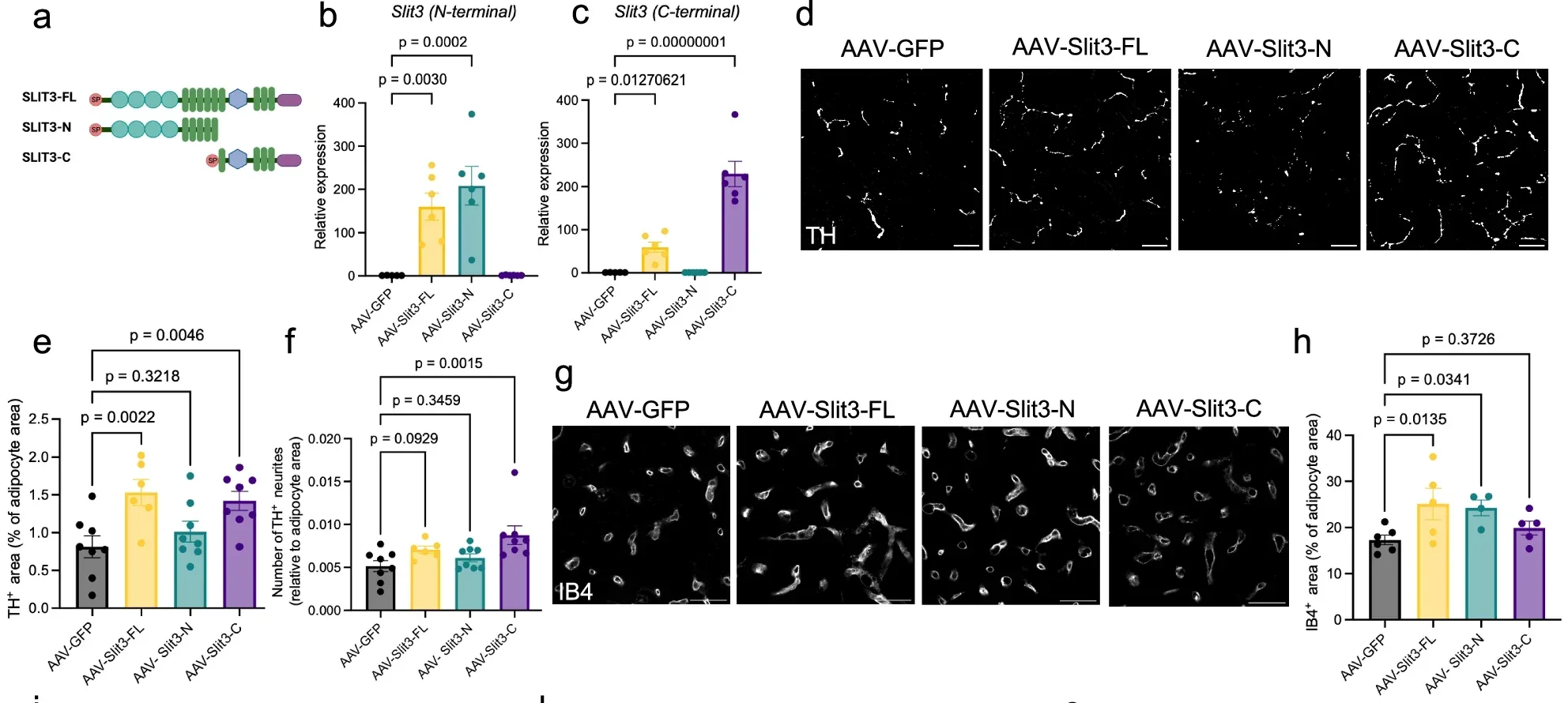
In the new study, the researchers identified the enzyme responsible for modifying it. BMP1 cleaves SLIT3 into two separate fragments. Each fragment takes on a different role. One promotes the growth of blood vessels, a process known as angiogenesis. The other supports the expansion of nerve networks, often referred to as sympathetic innervation. Together, they create the pathways that allow brown fat to receive signals and fuel.
“It works as a split signal, which is an elegant evolutionary design in which two components of a single factor independently regulate distinct processes that must be tightly coordinated in space and time,” Shamsi said. A single molecule, divided into two parts, coordinates two systems that must operate in sync.
The researchers also identified a receptor involved in this process. Known as PLXNA1, it binds to one of the SLIT3 fragments and helps control the growth of nerve connections in brown fat. That interaction matters because nerves trigger activation. When the brain senses cold, it sends signals through these pathways, prompting brown fat to begin generating heat.
Blood vessels handle the other half of the equation. They supply the raw materials for thermogenesis and distribute the resulting heat throughout the body. Without both systems in place, the process breaks down.
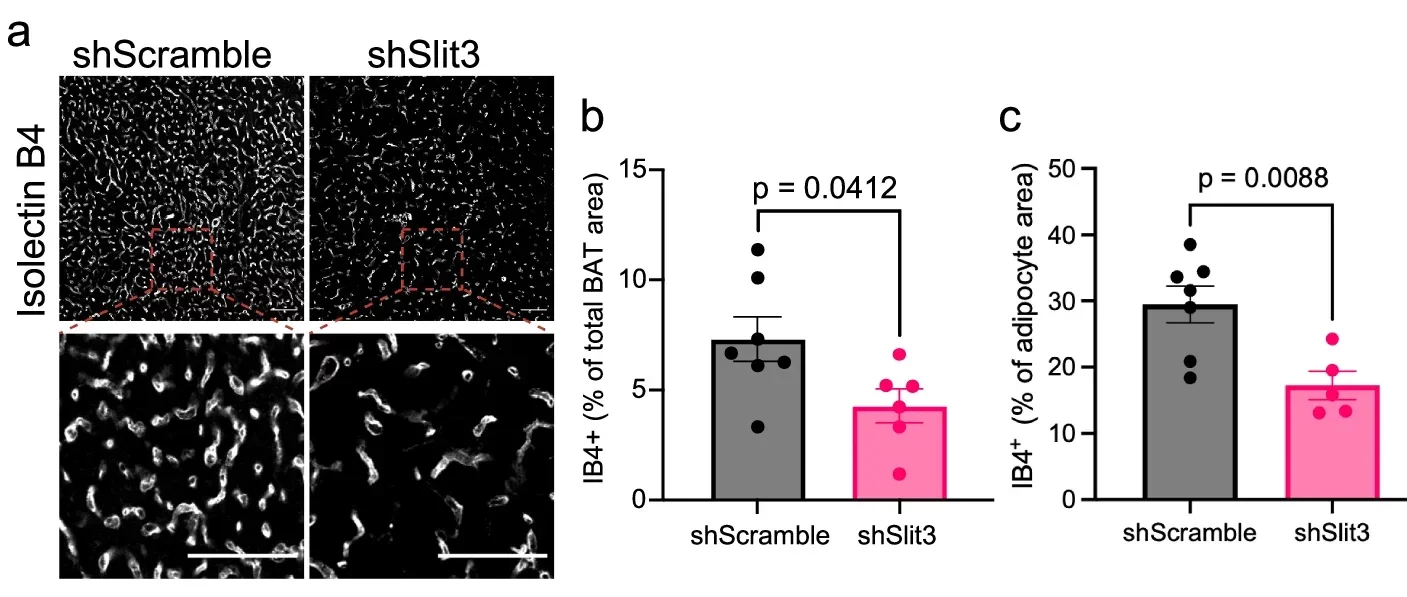
To test the importance of this pathway, the team studied mice, which naturally have active brown fat and can tolerate cold conditions well. They removed either SLIT3 or the PLXNA1 receptor from the animals’ brown fat. The effect was immediate and clear.
The mice became sensitive to cold and struggled to maintain body temperature. Closer examination of their brown fat revealed structural problems. The tissue lacked proper nerve organization and had a reduced density of blood vessels. The fat cells themselves remained, but their surroundings had changed. That distinction proved important. The findings suggest that simply having brown fat is not enough. The tissue must also have the right internal structure to function effectively.
“Our research shows that just having brown fat isn’t enough, you need the right infrastructure within the tissue for heat production,” Shamsi said.
The team also explored whether this pathway might matter in humans. They examined fat tissue samples from more than 1,500 people, including individuals with obesity.
Their focus stayed on the gene that produces SLIT3. Previous research had linked this gene to obesity and insulin resistance. In this analysis, SLIT3 expression appeared to relate to fat tissue health, inflammation, and insulin sensitivity.
“That really got our attention, as it suggests that this pathway could be relevant in human obesity and metabolic health,” Shamsi said. The study does not establish direct clinical applications. It does suggest that the same mechanisms observed in mice could play a role in people.
Most current weight loss drugs work by reducing appetite. GLP-1 medications, for example, help people eat less, lowering the amount of energy stored in the body. Brown fat offers a different path. Instead of limiting intake, it increases energy use.
That distinction matters. A treatment that enhances brown fat activity could shift how the body handles calories, drawing them into heat production rather than storage. The SLIT3 pathway points to a way of strengthening the systems that make this possible. Rather than targeting fat cells directly, the approach would focus on the networks that support them.
The study also highlights how multiple cell types work together in this process. Brown fat cells, endothelial cells that form blood vessels, and nerve cells all interact to build a functional system. The coordination among these components appears essential.
The findings add detail to a broader picture of how tissues operate. Biological systems rarely rely on a single cell type acting alone. Instead, they depend on coordinated activity across different structures. In this case, brown fat behaves less like an isolated group of cells and more like a networked system. Signals, fuel, and structure all intersect.
The SLIT3 mechanism offers a way to see how that coordination emerges. A single protein, split into two parts, directs two separate processes that must align for thermogenesis to work. That arrangement may reflect an efficient design. By linking the growth of nerves and blood vessels, the system ensures that activation and fuel supply develop together.
This work suggests that improving metabolic health may require strengthening the internal networks of brown fat, not just activating the cells themselves.
Therapies that target pathways like SLIT3 could increase how much energy the body uses, offering an alternative to appetite-focused treatments.
Research findings are available online in the journal Nature.
The original story “Scientists are rethinking how the body burns calories” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Scientists are rethinking how the body burns calories appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.