Aging does not arrive all at once. It builds quietly across years, touching cells long before symptoms appear. Scientists have spent decades studying diseases tied to aging. Now, many want to understand aging itself, hoping to slow its effects at the source.
A new study offers one of the clearest views yet. Researchers at The Rockefeller University mapped how aging changes cells across the entire body. They examined nearly 7 million individual cells from mice. These cells came from 21 different tissues and three life stages.
“Our goal was to understand not just what changes with aging, but why,” said Junyue Cao, who heads the Laboratory of Single Cell Genomics and Population Dynamics. “By mapping both cellular and molecular changes, we can identify what drives aging. That opens the door to interventions that target the aging process itself.”
The findings reveal a body in motion. Aging is not a simple decline. It is a coordinated shift across organs, cell types, and biological systems. Some changes begin earlier than expected, and many differ between males and females.
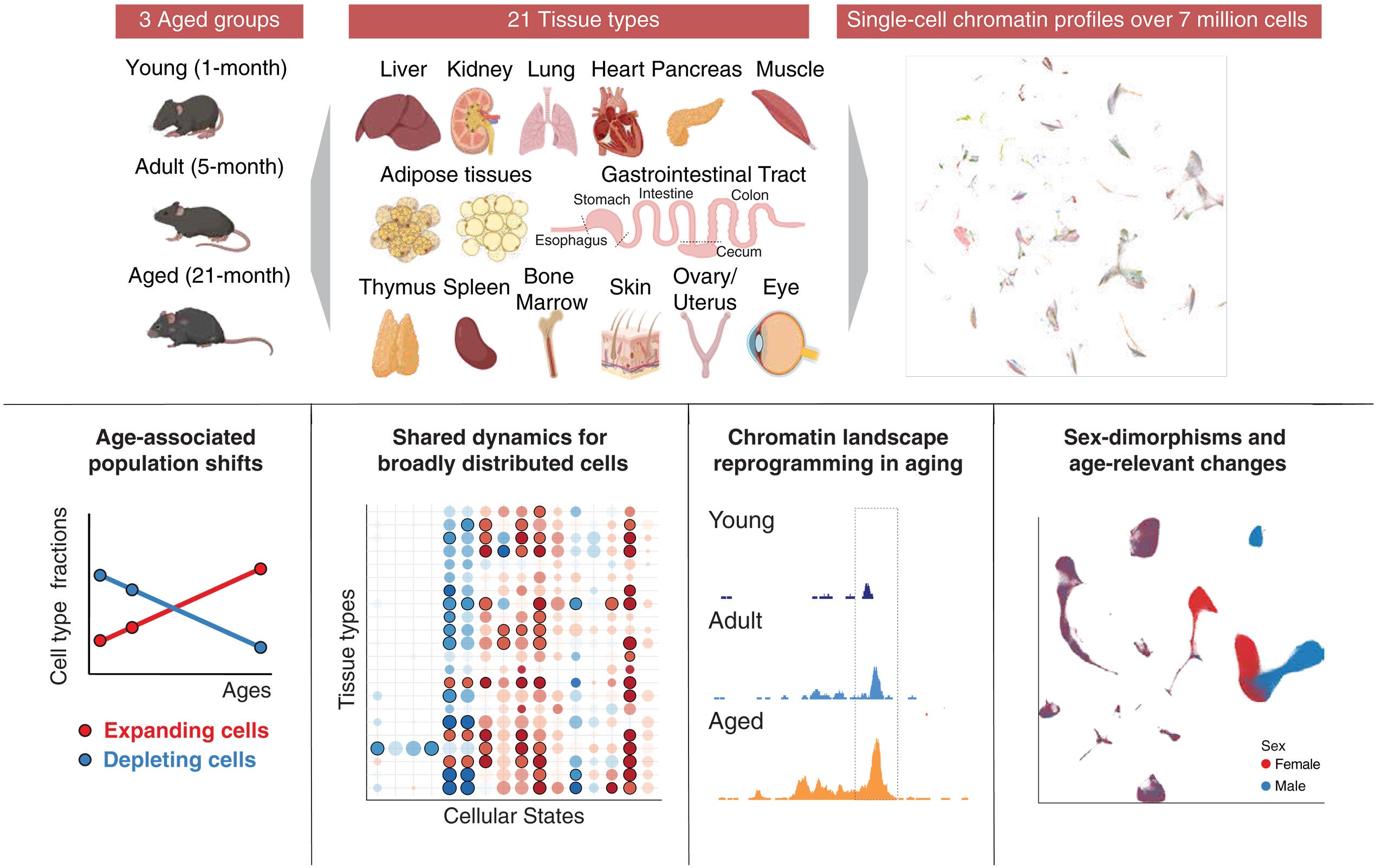
To build this atlas, the research team used a method called single-cell ATAC-seq. This technique shows which parts of DNA are open and active in each cell. When DNA is open, genes can be used. When it is closed, genes remain silent.
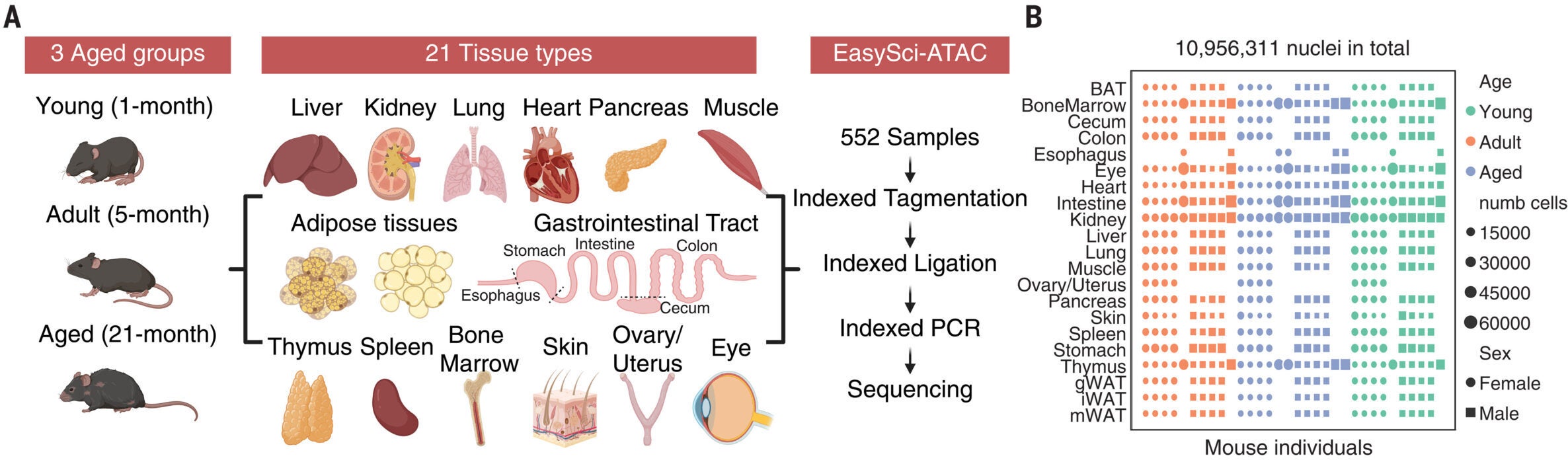
By studying how DNA packaging changes, scientists can track how cells function and respond to stress. The team applied this method to millions of cells collected from 32 mice. They examined animals at one month, five months, and 21 months of age.
“What’s remarkable is that this entire atlas was generated by a single graduate student,” Cao said. “Most large atlases like this require large consortia with dozens of laboratories but our method is far more efficient than other approaches.”
The dataset is massive. Researchers identified more than 1,800 cell subtypes, including rare types not previously described. They also mapped over 1.3 million regions of the genome that regulate gene activity.
This level of detail allows you to see aging in a new way. Instead of focusing on one organ, the study tracks patterns across the whole body.
For years, scientists believed aging mainly changed how cells functioned. This study shows it also changes how many of each type exist.
About one quarter of all cell types showed clear shifts in number over time. Some declined sharply. Others expanded.
Muscle cells and kidney cells often decreased with age. These cells play key roles in movement and filtration. Their loss may help explain why physical strength and organ function decline over time.
At the same time, immune cells expanded across many tissues. Plasma cells and macrophages increased in number. These changes suggest a rising state of inflammation, a common feature of aging.
“The system is far more dynamic than we realized,” Cao said. “And some of these changes begin surprisingly early. By five months of age, some cell populations had already begun to decline. This tells us that aging isn’t just something that happens late in life; it’s a continuation of ongoing developmental processes.”
Some declines were steep. Certain cell types dropped by more than half by middle age in mice. These included stem-like cells that help repair tissue. Their loss may reduce the body’s ability to heal.
One of the most striking findings is how synchronized these changes appear. Cells in distant organs showed similar patterns over time.
The same types of cells rose or fell together, even when they lived in different tissues. This suggests that signals move through the body to coordinate aging.
These signals may include molecules in the bloodstream. They could act as messengers, telling cells when to shift into new states.
This idea challenges the view that aging is purely local. Instead, it points to a shared program that unfolds across the entire body.
If that program can be understood, it may be possible to slow or redirect it.
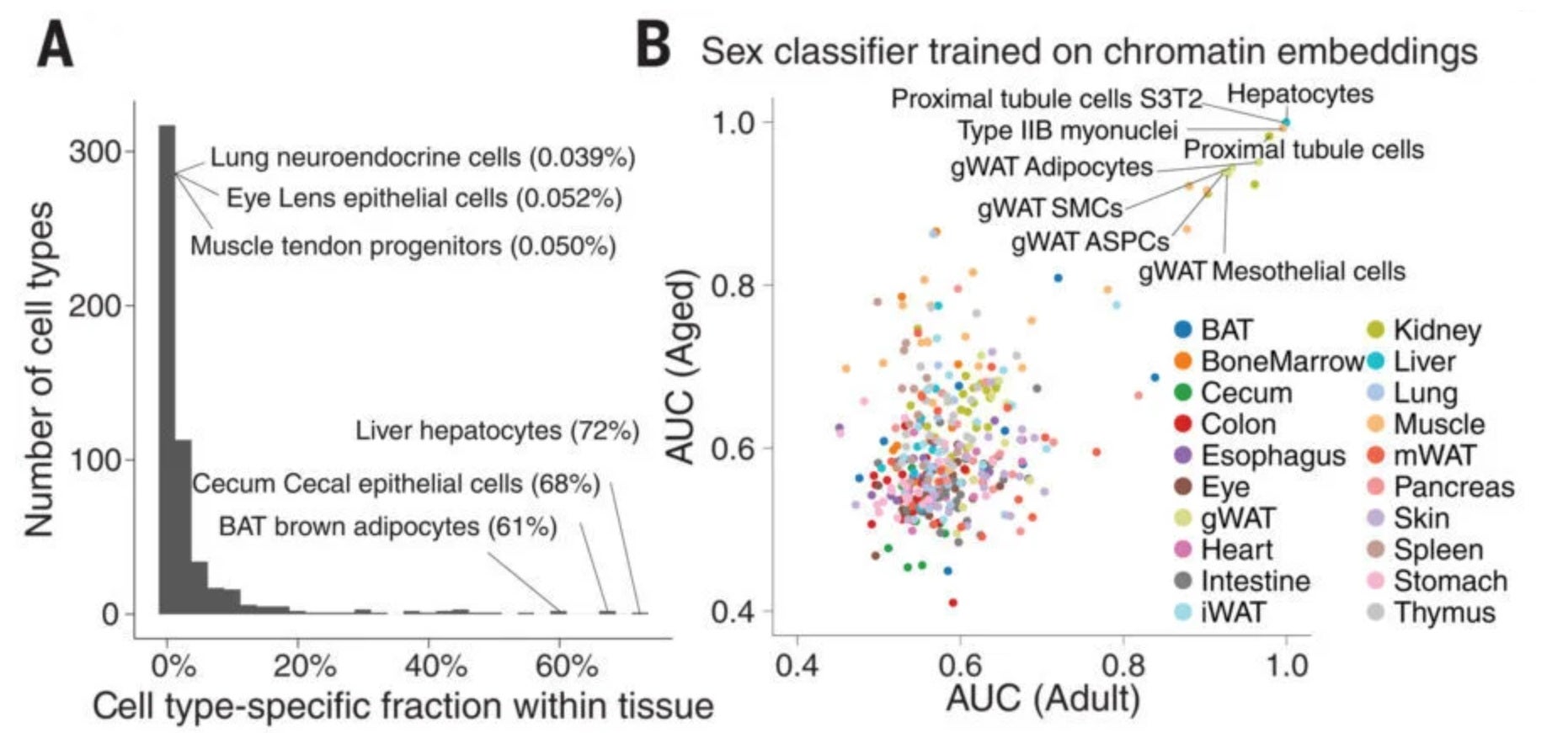
The study also uncovered strong differences between males and females. About 40 percent of aging-related changes differed by sex.
Many of these differences involved the immune system. Females showed broader immune activation as they aged. This may help explain why autoimmune diseases are more common in women.
“It’s possible this could explain the higher prevalence of autoimmune diseases in women,” Cao said.
In some cases, the same cell type behaved in opposite ways in males and females. One kidney cell type increased in older females but decreased in males. Another showed the reverse pattern.
These findings highlight the need for sex-specific approaches in aging research. Treatments that work for one group may not work the same way for another.
Looking at broad cell categories can miss important details. When researchers examined smaller subtypes, they found even more change.
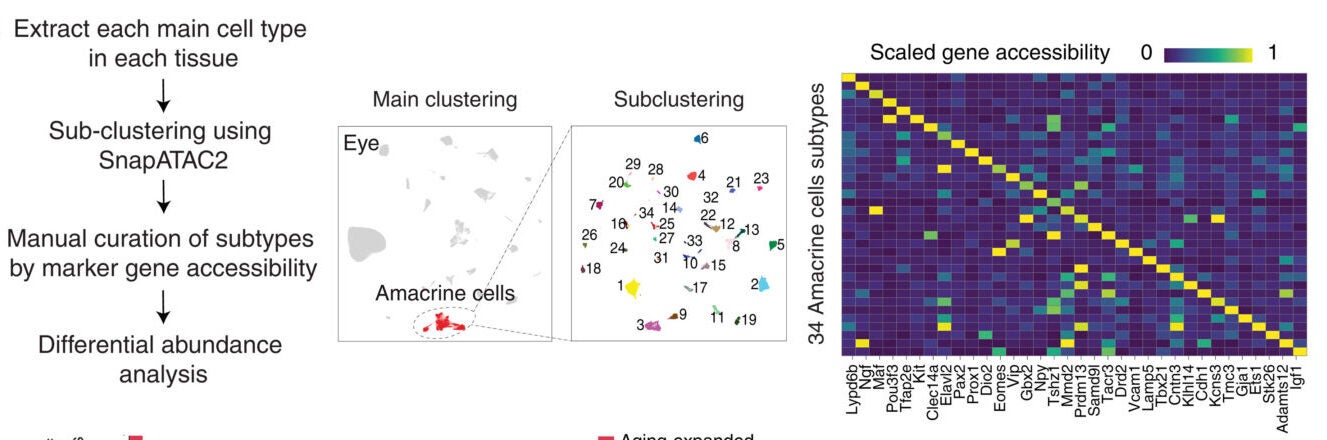
Nearly one third of cell subtypes showed significant aging-related shifts. Some of these changes were invisible at the larger scale.
For example, total immune cell numbers might rise. But within that group, younger, less active cells declined. More activated or stressed cells took their place.
In bone marrow, early immune cells decreased while others increased. This shift may alter how blood cells form over time.
In the kidney, certain endothelial cell subtypes declined while others stayed stable. Some new subtypes appeared that showed signs of stress and inflammation.
These patterns show that aging reshapes not just the number of cells, but their identity and behavior.
The immune system stood out across the dataset. Changes appeared in nearly every tissue.
Naïve T cells, which respond to new threats, declined with age. Early B cell progenitors also decreased. This suggests the body becomes less able to produce fresh immune responses.
At the same time, more activated immune cells increased. These cells often produce inflammatory signals.
This shift may lead to chronic inflammation, a condition linked to many age-related diseases. It may also weaken the ability to fight new infections.
The study found that many of these immune changes appeared across multiple organs. This supports the idea of a system-wide aging process.
Beyond cell numbers, the study tracked changes in DNA accessibility. These changes reveal how genes turn on or off over time.
Out of 1.3 million regions studied, about 300,000 showed significant changes with age. Around 1,000 of these changes appeared across many cell types.
“These findings challenge the idea that aging is just random genomic decay,” Cao said. “Instead, we see specific regulatory hotspots that are particularly vulnerable, and these are precisely the regions we should be studying if we want to understand what drives the aging process.”
Many of these hotspots relate to the immune system, inflammation, and stem cell maintenance. These areas may hold the key to understanding why aging progresses.
The team also found that inflammatory signals, such as cytokines, can trigger many of the same changes seen in aging cells.
This suggests that chronic inflammation may drive much of the aging process at the molecular level.
By combining cell counts with DNA data, the study offers a powerful framework for future research.
Scientists can now identify which cells are most vulnerable and which genetic regions change most. This creates clear targets for new therapies.
Drugs that control cytokines may help reduce harmful inflammation. Other treatments may aim to preserve stem cells or restore healthy cell balance.
“This is really a starting point,” Cao said. “We’ve identified the vulnerable cell types and molecular hotspots. Now the question is whether we can develop interventions that target these specific aging processes. Our lab is already working on that next step.”
The study focuses on mice, so more work is needed to confirm these patterns in humans. Still, the scale and detail provide a strong foundation.
This research shifts how scientists think about aging. Instead of viewing it as a slow breakdown, it shows a coordinated process across the body. That insight could change how diseases are treated.
If aging drives many conditions, slowing it could delay several diseases at once. This includes cancer, heart disease, and neurodegenerative disorders.
The study also highlights the role of inflammation. If cytokines drive aging signals, therapies that control immune activity may help protect tissues over time.
The discovery of early changes is also important. Some cell populations begin to decline much earlier than expected. This suggests that prevention strategies may need to start sooner in life.
Sex differences add another layer. Future treatments may need to be tailored to male and female biology to be effective.
For researchers, the atlas serves as a roadmap. It helps identify which cells and genes to target in future studies. It also provides a reference for testing new anti-aging therapies.
In the long term, this work may lead to treatments that extend healthy years of life, not just lifespan. By understanding how cells change with age, scientists move closer to slowing the process itself.
Research findings are available online in the journal Science.
The original story “Scientists map millions of cells to decode the biology of aging” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Scientists map millions of cells to decode the biology of aging appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.