A once-daily dose of a medication, which averaged 195 mg, produced blood levels significantly lower than those typically pursued by psychiatrists. However, it succeeded in generating a weak signal concerning the potential influence on memory performance.
This is the essence of a two-year placebo-controlled clinical trial studying the effects of low-dose lithium therapy on memory performance in older adults who have mild cognitive impairment. The University of Pittsburgh School of Medicine conducted the study, which utilized repeated cognitive assessments, neuroimaging data, and blood-based biomarkers to monitor participants over time. The results were published in JAMA Neurology.
Lithium has been used for many years as a medication for patients with bipolar disorder. This study examined a different question. It asked how effective long-term therapy with low doses of lithium might be in slowing the decline of memory and cognitive function in individuals diagnosed with mild cognitive impairment, which is often identified before the onset of Alzheimer’s disease.
Ariel Gildengers, PhD, was the principal investigator of this clinical trial. He is a professor and geriatric psychiatrist at the University of Pittsburgh Medical Center (UPMC). Previous research conducted by Gildengers in older adults with bipolar disorder provided the basis for the current trial.

“In prior studies of older patients diagnosed with bipolar disorder who were treated with chronic administration of lithium, we observed increased measures of brain integrity,” stated Gildengers. “Thus, the new study’s objective was to evaluate whether lithium’s apparent protective effects on neurons may extend beyond mood disorders. We also wanted to assess the efficacy of long-term therapy on cognitive decline in a formal longitudinal clinical trial.”
The clinical trial was named Lithium as Therapy for Prevention of Cognitive Impairment in Elderly Patients, or LATTICE. It was a randomized clinical trial conducted at one institution, and the Data and Safety Monitoring Board conducted interim analyses every six months throughout the trial. The enrollment period began September 1, 2017, and continued through the COVID-19 pandemic. This required the research team to adapt methods used for screening and recruiting subjects.
Between February 2, 2018, and August 6, 2022, researchers evaluated 170 individuals for eligibility. A total of 80 participants began the trial. Of these, 41 received lithium and 39 received placebo.
Participants were required to be at least 60 years old with mild cognitive impairment and to function normally in their daily lives. They could not have any significant psychiatric or neurological illness. The amount of amyloid plaque buildup in the brain was measured using amyloid PET scans with Pittsburgh Compound B.
The cutoff point for determining whether someone was considered amyloid-positive was when the subject’s standardized uptake value ratio (SUVR) exceeded 1.346. This equated to a score of +1.0000 on the general index.
At the time of enrollment, there was no significant difference between the two groups except for age. The average age of the lithium group was 72.93 years, while the average age of the placebo group was 71.22 years. There was also a roughly equal number of females in both groups (56%). Most participants were Caucasian. Of the 80 total subjects, roughly 25% were considered amyloid-positive: 11 participants from the lithium group and 10 from the placebo group.
Lithium carbonate was administered in 150 mg or 300 mg doses in identical-looking capsules. This allowed titration to the maximum tolerated dose for each participant. The average final daily lithium dose for completers was 195 mg, with a mean serum level of 0.17 mEq/L and a maximum of 0.5 mEq/L. Adherence to the medication was 98% for the lithium group and 97% for the placebo group.
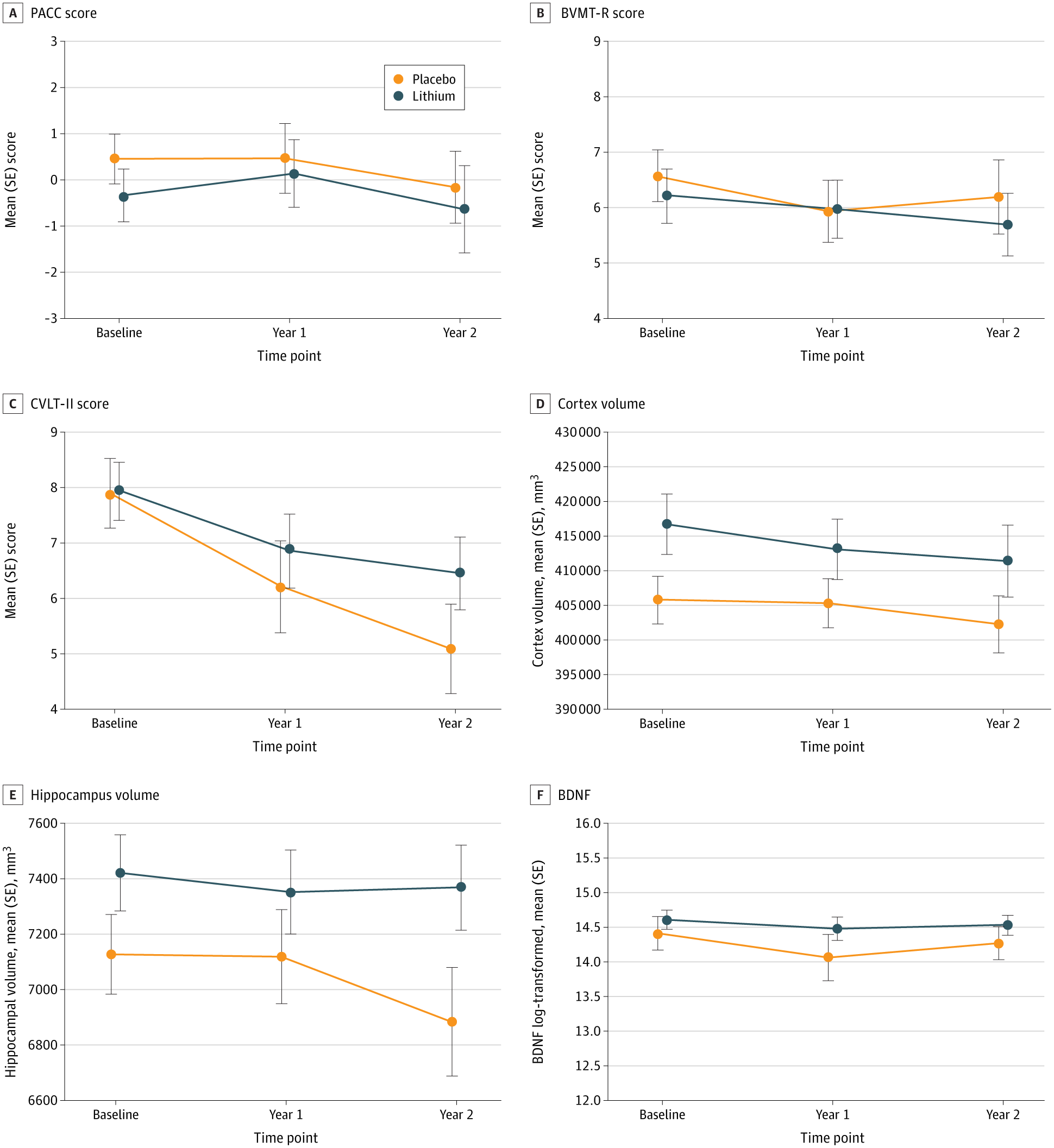
The study defined six coprimary outcomes: three cognitive measures, two brain volume measures, and one blood biomarker. The delayed recall subtest of the California Verbal Learning Test Second Edition (CVLT-II) was used as the primary cognitive outcome measure for memory.
CVLT-II scores decreased in both the lithium and placebo groups between time point one and time point two. However, the decrease in the lithium group was less pronounced than in the placebo group. The placebo group had an average annual decline of 1.42 points, while the lithium group had an average decline of 0.73 points.
The difference in annual decline was 0.69 points. This was considered statistically significant with a 95% confidence interval of 0.01 to 1.37. The P value was .05.
In addition to memory, no other cognitive domains demonstrated a significant benefit for lithium over placebo at either time point. The Preclinical Alzheimer Cognitive Composite (PACC) or the Brief Visuospatial Memory Test Revised (BVMT-R) showed no treatment-by-time interactions.
Results from imaging were consistent with these findings. Both groups showed decreases in hippocampal volume and cortical gray matter volume over time. The reduction in hippocampal volume between treatments did not reach statistical significance (P = .09).
Additionally, no significant difference between treatments was observed for total cortical gray matter volume (P = .78). There was also no difference in levels of Brain-Derived Neurotrophic Factor (BDNF) (P = .98).
One biomarker, Glycogen Synthase Kinase-3 (GSK-3), did not produce usable results. The assay failed to pass quality control.
Participants in this trial were selected based on their experience of mild cognitive impairment (MCI), not because they had biological evidence of Alzheimer’s disease-type pathology. This reflects the state of the field when the study was developed in 2016–2017. At that time, confirming Alzheimer’s disease required costly diagnostic methods.
These methods included radioactive imaging of amyloid levels using PET scans or a lumbar puncture to analyze cerebrospinal fluid. Blood-based biomarkers for detecting amyloid pathology were not yet widely available.
Because of the study design, many participants had the amnestic form of MCI, meaning memory problems were prominent. However, a large percentage were not necessarily experiencing Alzheimer’s disease pathology. Many were recruited from the general population.
The authors point out that this may have affected the strength of any potential treatment effects in the study.
According to exploratory analyses among participants who completed the study, amyloid-positive participants appeared to show stronger effect sizes than amyloid-negative participants. For the verbal memory measure, the effect size was Hedges’ g = 0.74 among amyloid-positive participants.
For hippocampal volume, the effect size was Hedges’ g = 0.82 in the amyloid-positive group. Amyloid-negative participants showed smaller effect sizes. The authors emphasized that the small sample sizes limit the conclusions that can be drawn from these analyses.
“The major takeaway from the study is that lithium cannot restore previous memories that have been lost,” says Gildengers. “However, if the results continue to show this effect, lithium will help to slow the rate of decline. This distinction is key to the design and interpretation of trial results.”
Because safety was a primary goal, all subjects were monitored for serum lithium concentration throughout the study. Researchers also monitored serum creatinine levels and thyroid hormones.
There were 41 serious adverse events during the study. Twenty-five occurred in the lithium group, and 16 occurred in the placebo group.
All events were determined to be unrelated to the study medication, except for one instance of increased creatinine levels or diarrhea. Two additional cases involved tiredness and tremor in the lithium group.
Of the participants who died during the study, all were in the placebo group. None of the deaths were related to the study.
More participants discontinued taking the medication than completed the full course. However, discontinuation rates were similar between groups. Thirty-four percent of the lithium group discontinued by two years, compared with 38 percent of the placebo group.
Both groups maintained more than 80 percent retention of outcome assessments at two years. This was consistent with the study’s feasibility goals.
The authors also identified a dose-related limitation. Older adults had difficulty tolerating lithium doses above 300 mg per day. As a result, they recommended that future studies limit dosing to 150–300 mg per day.
Research findings are available online in the journal JAMA Neurology.
The original story “Low doses of lithium may slow verbal memory decline from Alzheimer’s ” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Low doses of lithium may slow verbal memory decline from Alzheimer’s appeared first on The Brighter Side of News.