The current path to CAR-T cell therapy is, by any measure, a logistical ordeal. A patient’s immune cells must be drawn out of the body, shipped to a specialized facility, genetically reprogrammed, quality-checked, shipped back, and then infused, all while the cancer continues. The process takes weeks. It costs between $400,000 and $500,000. Some patients don’t survive the wait. Many never get access at all.
Scientists at UC San Francisco have now demonstrated something that could change that calculus entirely: a method to reprogram cancer-fighting immune cells while they remain inside the body, using a single injection. The research, published in Nature, describes the first time researchers have successfully inserted a large DNA sequence at a precise location in human T cells that were never removed from a living organism.
In mouse models engineered with human immune systems, a single dose cleared aggressive leukemia in 18 out of 20 animals. It also worked against multiple myeloma and, in a result the researchers call particularly significant, against a solid sarcoma tumor.
“I think this is just the beginning of a big wave of new therapies that will be truly transformational and save a lot of lives,” said Justin Eyquem, PhD, associate professor of medicine at UCSF and the paper’s senior author.
CAR-T therapy works by giving T cells, the immune system’s frontline fighters, a new set of molecular instructions. Those instructions take the form of chimeric antigen receptors, proteins that protrude from the cell surface and recognize specific markers on cancer cells. When a CAR latches onto one of those markers, it triggers the T cell to attack. Seven such therapies have been approved by the FDA for blood cancers, and they can produce dramatic, durable remissions in patients who had no other options.
The problem is access. Manufacturing these therapies requires infrastructure most hospitals don’t have, timelines that some patients can’t afford, and costs that most health systems struggle to cover. Patients also typically require intensive preparatory chemotherapy to clear bone marrow before receiving the engineered cells, a step that older or frailer individuals often can’t tolerate.
“It’s become a global access issue; many patients who would benefit from CAR-T cells either can’t afford them or can’t get them fast enough,” Eyquem said. “There has been a big push in the field to try to move to directly producing these cells in the body.”
In vivo manufacturing, as this approach is called, could eliminate the need for that preparatory chemotherapy as well.
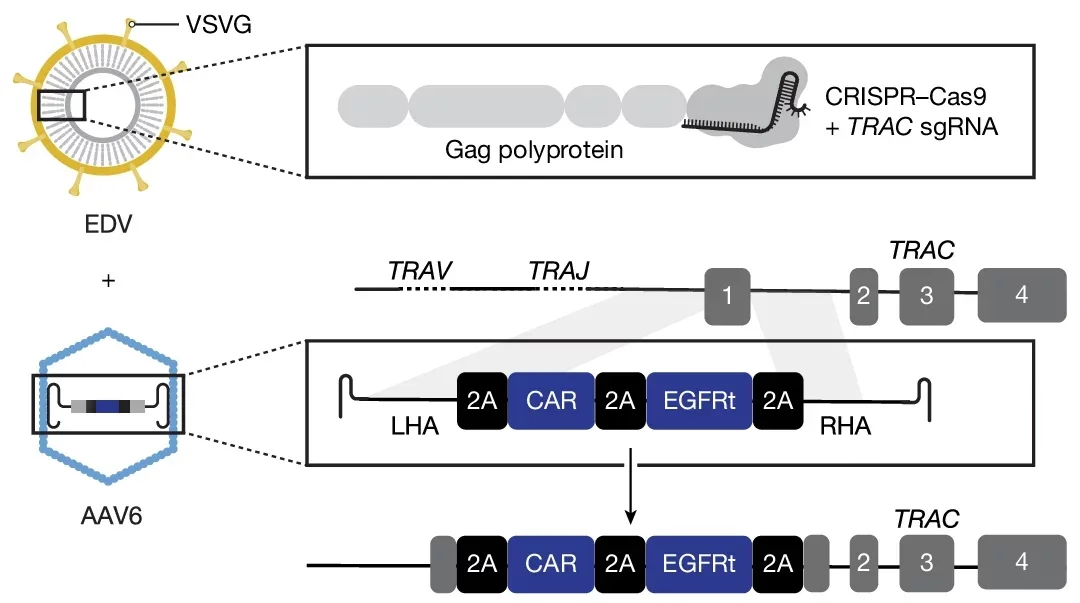
The UCSF team, working with collaborators at the Gladstone Institutes, Duke University, and the Innovative Genomics Institute, engineered a dual-particle system to carry gene-editing machinery directly to T cells circulating in the bloodstream.
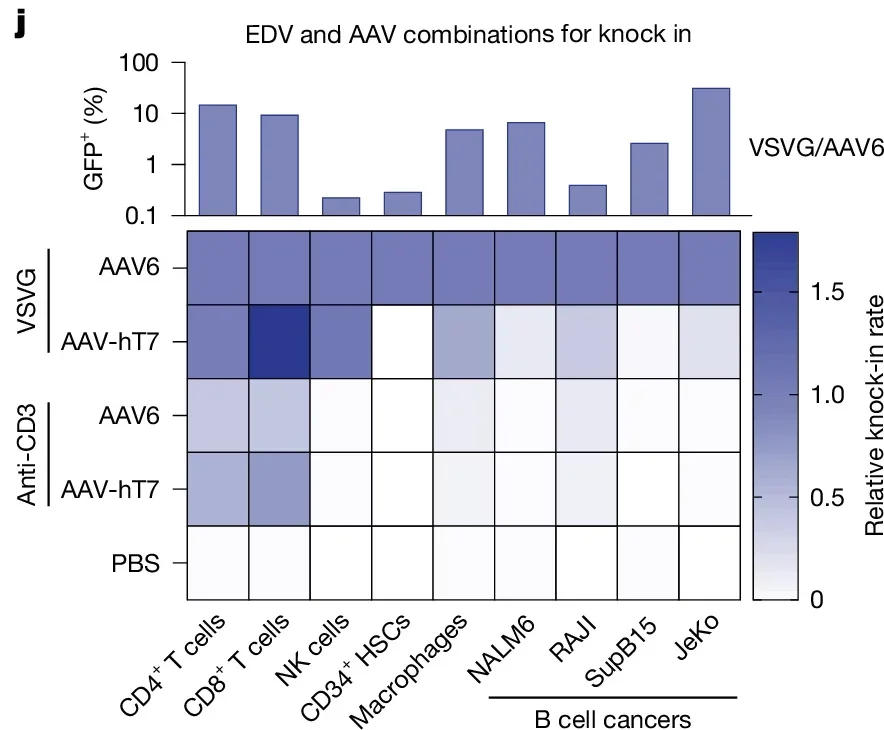
The first particle acts as a targeting vehicle. Coated with antibodies that bind exclusively to a protein called CD3, found only on T cell surfaces, it ensures the editing tools reach their intended destination and nothing else. This particle also carries CRISPR-Cas9 machinery, the molecular tool used to cut DNA at a specific location in the T cell’s genome.
The second particle is an engineered virus-like carrier that delivers new DNA, specifically the genetic instructions for building the cancer-fighting CAR protein. Critically, the DNA is designed to insert at a precise site within the T cell genome, one that functions as a molecular switch active only in T cells. The CAR gene only activates after landing in that exact spot, which means even if the DNA ends up in a cell it wasn’t meant for, it won’t produce a functional protein there.
The research team also evolved the viral carrier through multiple selection cycles to make it resistant to antibodies already present in human blood, a significant hurdle for earlier gene therapy approaches, and confirmed that it preferentially targets T cells while leaving stem cells and cancer cells largely unaffected.
“When you manufacture these cells outside the body, you can do a lot of quality control to make sure you only end up with re-engineered T cells,” Eyquem said. “Inside the body, we can’t do that post-manufacturing quality control, so we really needed to optimize the approach upfront to avoid altering any other cells.”
Co-first authors William Nyberg, PhD, and Pierre-Louis Bernard, PhD, both UCSF postdoctoral fellows, led the experiments. In mice engrafted with aggressive leukemia, a single injection cleared all detectable cancer in nearly all animals within two weeks. The engineered CAR-T cells made up as much as 40 percent of immune cells in some organs and successfully eliminated cancer from both the bone marrow and spleen. When researchers rechallenged recovered mice with fresh cancer cells weeks later, the engineered cells responded again and maintained control.
Against multiple myeloma, all eight treated mice achieved complete responses. Against a solid sarcoma, a tumor type that has historically resisted CAR-T therapy because of how the solid tumor environment suppresses immune cells, the approach produced complete responses in the majority of animals.
One finding surprised even the researchers. The T cells reprogrammed inside the body appeared to outperform those manufactured in the lab by conventional means, expanding faster, expressing the cancer-targeting protein more consistently, and reaching peak activity earlier.
“What was especially remarkable was that the cells we’re generating in vivo actually look better than what we make in the lab,” Eyquem told The Brighter Side of News. “We think that when cells are taken out of the body and grown in the lab, they lose some of their ‘stemness’ and proliferative capacity and that doesn’t happen here.”
The working hypothesis is that removing T cells from the body and culturing them in laboratory conditions strips away some of their youthful, highly adaptable qualities. Cells reprogrammed in place retain those characteristics.
The study has important caveats. All experiments were conducted in mice, and the path from mouse models to human clinical trials is long and often humbling. The humanized mouse models used here, while valuable, operate under different conditions than a fully intact human immune system. Safety questions about where the delivery particles go in the body and how a human immune system responds to them over time will require studies in larger animals before clinical testing can begin.
There are also technical questions about redosing. Antibodies generated against the viral carrier after a first injection could limit the effectiveness of subsequent treatments, a challenge the team acknowledges as an active area of work.
Eyquem and his collaborators have founded a company called Azalea Therapeutics to advance the dual-particle platform through clinical development. No timeline for human trials has been specified.
If this approach translates to humans, the implications extend well beyond laboratory metrics. CAR-T therapy would no longer require the infrastructure of a major academic cancer center. A community hospital with refrigeration and an infusion suite could theoretically offer it. The weeks-long manufacturing wait would be gone. The need for preparatory chemotherapy might be eliminated. The six-figure price tag could fall substantially.
Beyond cost and logistics, the technology’s underlying architecture is not limited to CAR-T. The same dual-particle approach could theoretically be adapted to insert other types of genetic instructions into T cells, including engineered T cell receptors and other immune-reprogramming tools not yet in clinical use.
The researchers also note that because the system targets only T cells through both the delivery vehicle and the gene’s own activation switch, the off-target engineering risks that have complicated earlier in-body gene therapy efforts are substantially reduced.
“If we can translate this to humans, we could dramatically reduce costs, eliminate waiting times, and potentially allow community hospitals, not just major cancer centers, to offer these life-saving therapies,” Eyquem said. “That would truly democratize access to CAR-T cell therapy.”
Research findings are available online in the journal Nature.
The original story “New injectable system reprograms cancer-fighting T cells inside the body” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post New injectable system reprograms cancer-fighting T cells inside the body appeared first on The Brighter Side of News.