When a person loses a leg above the knee, the nerves that once moved that leg don’t simply go quiet. They keep firing. The brain still sends signals down through what remains, still attempts to flex the ankle, extend the knee, curl the toes, even when none of those structures exist anymore. The signals travel to the end of what’s left and stop there, carrying movement instructions that have nowhere to go.
For decades, those signals were essentially inaccessible, too faint and too difficult to interpret reliably. Prosthetic legs, unlike their arm counterparts, have largely operated on autopilot, using mechanical systems and built-in sensors to approximate walking without any direct input from the user’s own nervous system.
A research team led by scientists at Chalmers University of Technology in Sweden has now changed that.
In a study published in Nature Communications, the researchers describe the first successful decoding of leg movement signals recorded directly from the remaining nerves of people with above-knee amputations. Using hair-thin implantable electrodes and an AI system designed to process signals the way biological neurons do, they extracted not just broad movement categories but precise, detailed intentions, including the will to wiggle toes.

“When you tell your body to move, signals travel through the nerves to the muscles which carry out the action, even if the limb is no longer there,” said Giacomo Valle, assistant professor at Chalmers and one of the study’s senior authors. “This means you can find all the information needed within those nerves. The major challenge is extracting that information and understanding the neural code behind it.”
The gap between the aspiration and the reality of neural prosthetic control is considerable. Arm and hand prostheses have made real progress in muscle-based control, using signals from residual muscles that are still activated by nerves from the brain. That approach works reasonably well when the relevant muscles survive amputation.
For above-knee amputees, the problem is more severe. The amputation removes not just the leg but the muscles that would otherwise serve as the interface. Most prosthetic legs on the market today operate passively, adapting automatically to movement but never responding to what the user actually intends. Some systems use surface electrodes on the skin to pick up muscle signals, but these are too noisy and imprecise for fine-grained control, particularly when multiple joint movements are involved.
The more direct route, reading signals from the nerves themselves rather than from muscles, had been attempted before, but only in the arms. Nerves produce weak electrical signals that are easily drowned out by noise, and extracting them requires electrodes implanted inside the nerve tissue itself, along with algorithms capable of making sense of what they capture. Applying this to the legs, where amputations are far more common and far more severe, remained an unsolved problem.
The Chalmers team, working with collaborators at the University of Zurich, ETH Zürich, and the University of Freiburg, attempted it with two participants who had above-knee amputations.
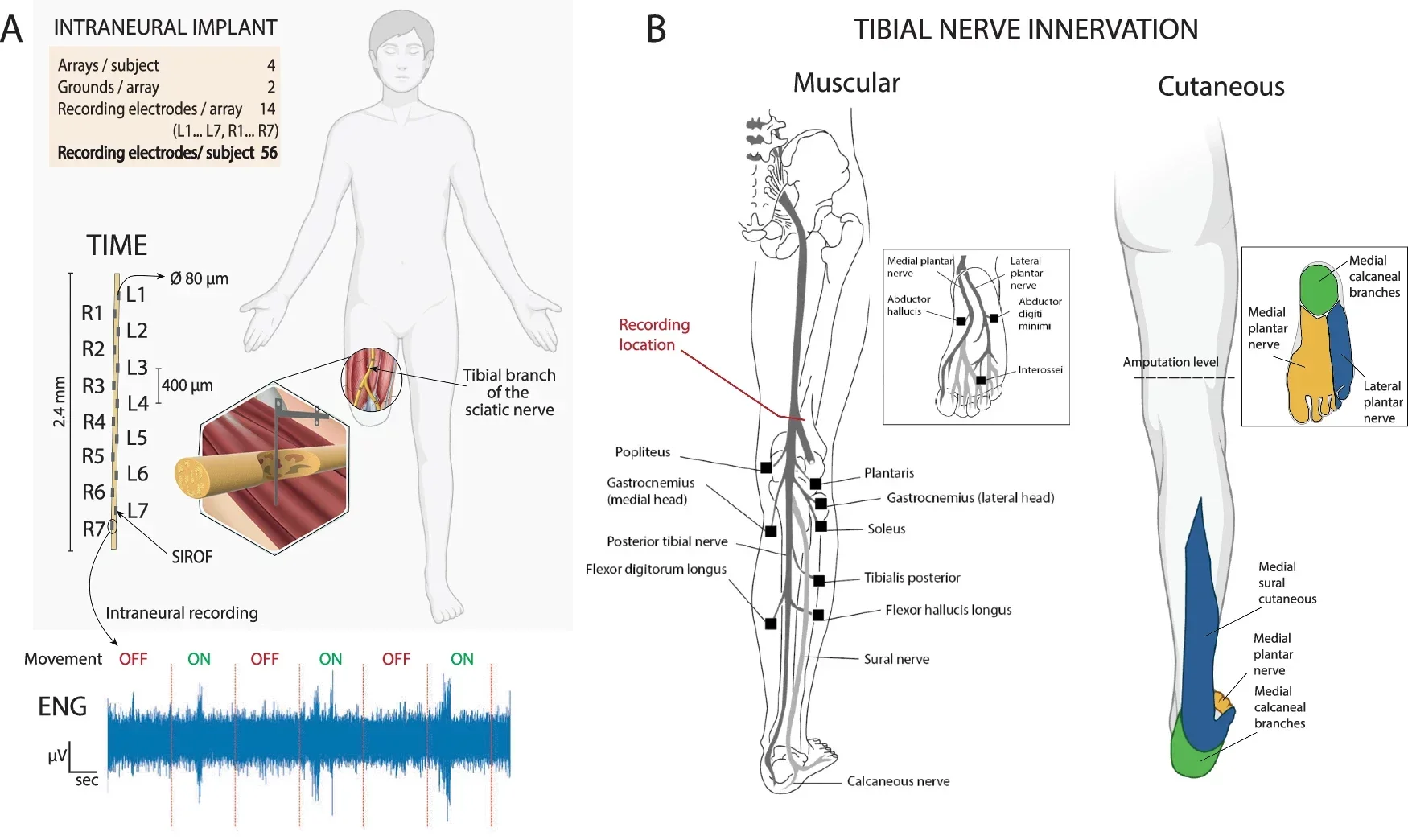
The implants used in the study are called transversal intrafascicular multichannel electrodes, a name that describes their design: thin, flexible, and inserted across the width of a nerve rather than along it. Each electrode is roughly the diameter of a human hair, carries 14 recording and stimulation sites, and is flexible enough to move with the nerve without damaging it.
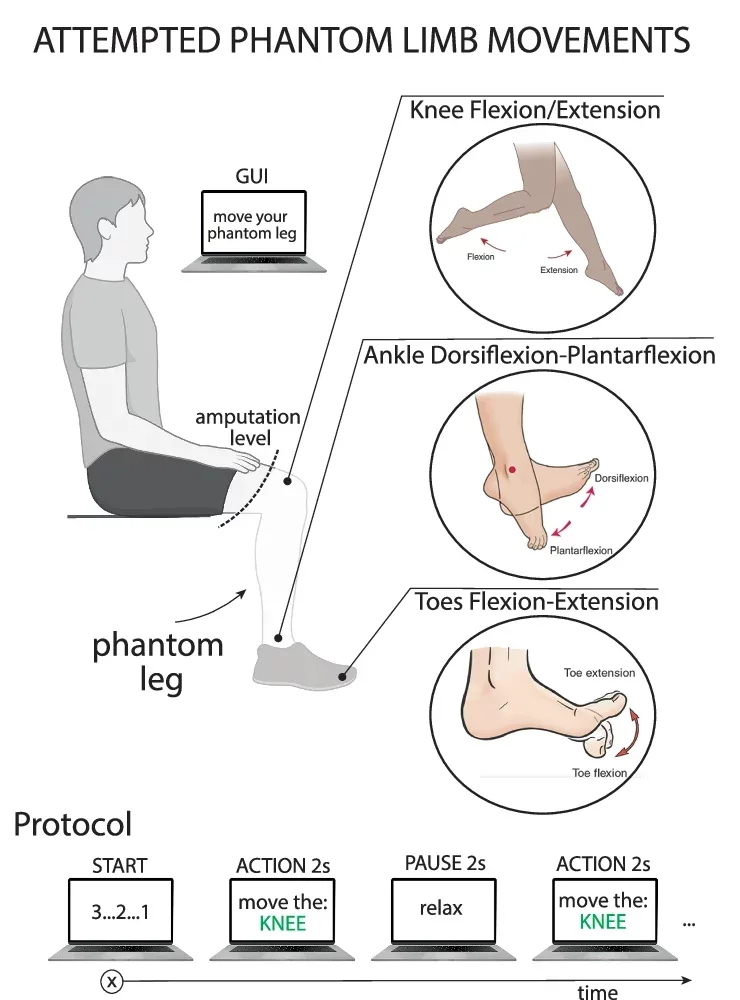
Four of these were implanted in the tibial branch of the sciatic nerve in each participant, the nerve that controls movement and sensation through the lower leg, ankle, and foot. Participants were then asked to attempt movements with their phantom limb: flexing and extending the knee, the ankle, and the toes, one at a time, while the implants recorded whatever signals they could capture.
Ninety-one percent of the recording sites in the first participant showed meaningful responses to at least one movement type. The signals varied by joint and by direction, with different electrode sites responding to knee movement versus ankle movement versus toe movement. Even toes, controlled by muscles that had been amputated entirely, produced detectable signals in the remaining nerve.
“It was amazing to see how electrodes placed high up in what remains of a leg could decode attempts to wiggle the toes,” Valle said.
Recording the signals was only half the challenge. The other half was interpreting them, and this is where the study breaks new ground in a technical sense.
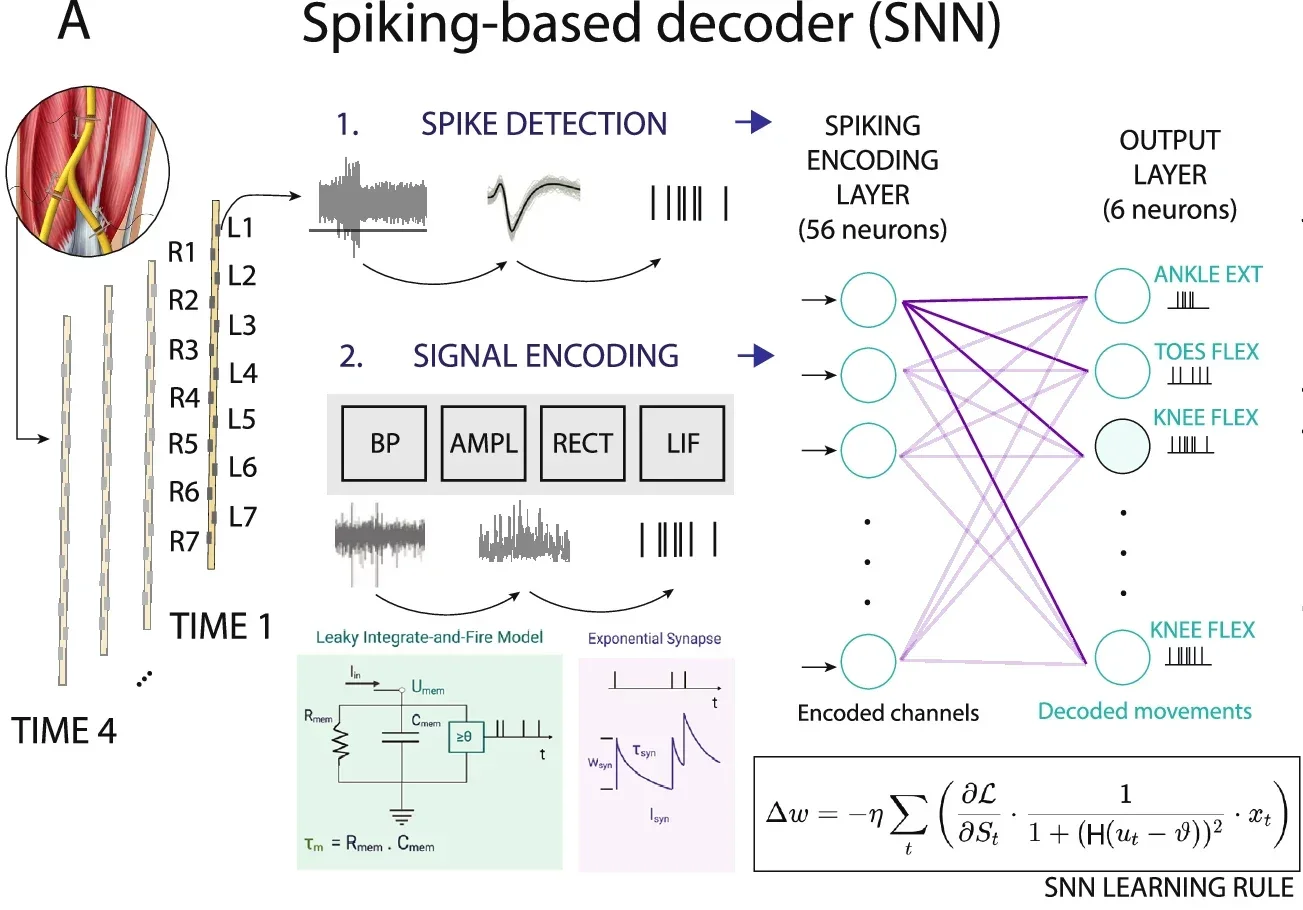
Conventional AI systems, including the kind used in language models and image recognition, process continuous streams of numerical data. Nerves don’t communicate that way. They send discrete electrical pulses called spikes, and timing and pattern carry information as much as raw magnitude does.
The research team used a class of algorithm called a spiking neural network, which processes inputs as sequences of spikes rather than continuous values. The choice was deliberate: matching the computational architecture to the nature of the biological signal.
“Peripheral nerves communicate through discrete electrical impulses, or spikes, and spiking neural networks are therefore naturally suited to processing this type of signal,” Elisa Donati, professor at the University of Zurich and ETH Zürich and the other senior author told The Brighter Side of News. “By aligning our computational models more closely with biology, we can extract movement intent efficiently, using compact models and relatively limited data.”
The spiking neural network significantly outperformed conventional classifiers in both participants when tested on the same data. And the algorithm’s design carries a practical advantage beyond accuracy: it is compact and energy-efficient enough to potentially run on a chip embedded directly in a prosthetic device, without requiring an external computer to process the signals.
A further finding from the study concerns the bidirectional potential of the technology. Previous work on restoring sensation to amputees used the same type of electrode, but only to deliver electrical stimulation outward to the nerve, creating tactile sensations. This study used the same implants in both directions simultaneously: recording motor signals going out from the brain and delivering sensory signals coming back.
The two types of nerve fibers, those carrying motor commands and those carrying sensory information, turned out to be spatially segregated within the nerve itself at the implantation site. Motor electrodes and sensory electrodes occupied different areas. That separation meant a single device could serve both functions without the channels interfering with each other.
“Once electrodes are implanted inside the nerve, they can be used to communicate bidirectionally with the nervous system,” Valle said. “So, for the first time, a single neurotechnology can provide both natural neural control and sensory feedback in the same implantable device.”
The study is explicit about its limitations. Two participants is a small sample, and the decoding was done offline rather than in real time. Accuracy, while significantly better than previous approaches, was not perfect; the system sometimes confused flexion for extension of the same joint. Long-term signal stability after implantation remains unknown. Applying this in daily life, with a real prosthesis responding to decoded signals in real time, has not yet been tested.
The researchers also note a fundamental difficulty in verifying that participants were imagining specific movements cleanly. Without sensory or visual feedback confirming the movement, the brain’s motor commands may be less precise than they would be with a functioning limb attached.
The next step is integrating the technology into an actual prosthetic leg and testing it in use. Valle said he believes the method could eventually extend beyond leg prosthetics to other applications, though no specific timeline is projected.
Lower-limb amputations account for roughly 69 percent of all limb loss worldwide, and above-knee amputations carry the highest rates of disability, reduced walking speed, and energy cost. The vast majority of people in this group currently use prosthetic legs that require no neural connection at all, operating mechanically with no input from the user’s intentions.
The gap between that reality and what the nervous system is still trying to do is the opening this research addresses. A leg that responds to what the user actually intends, rather than what sensors predict based on body position and gait patterns, could meaningfully change daily life for millions of people.
Beyond mobility, the bidirectional capability points toward something more fundamental: a prosthesis that a user can not only control but feel. Sensation from the foot while walking, feedback when a surface changes, the experience of the prosthesis as part of the body rather than a tool attached to it. The signals for both are already present in what remains of the leg’s nervous system. What this research demonstrates is that those signals can now be read.
Research findings are available online in the journal Nature Communications.
The original story “New implant can read leg movement signals from amputated nerves” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post New implant can read leg movement signals from amputated nerves appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.