Getting light deep inside the body has always come with a catch. Tissue scatters and absorbs it, which means doctors and researchers often have to cut into the body or thread in optical fibers to reach the places they want to target.
A team at Stanford University now says it has found a way around that problem, at least in mice. Instead of trying to shine light through layers of tissue, the researchers used ultrasound to trigger tiny particles circulating in the bloodstream, making them emit light exactly where the sound waves were focused. The result is a noninvasive method for creating small, controlled pockets of light inside living tissue.
That matters because light has become an increasingly useful tool in biology and medicine. It can stimulate cell activity, influence neural signals, and help treat certain cancers. The problem has never been what light can do. It has been how to get it where it needs to go without physically entering the body.
“Ultrasound is very convenient to use, and it penetrates much deeper into the body than light,” said Guosong Hong, an assistant professor of materials science and engineering at Stanford and the senior author on the paper. “With these materials, we can produce light emission in the brain, in the gut, in the spinal cord, in the muscle, virtually anywhere, without needing a physical implant.”

The approach starts with particles that are not the sort of material most people would associate with medicine. The researchers began with large ceramic particles that emit light when they are under mechanical stress. Ultrasound waves can create that stress.
To make them usable in the body, the team processed those particles into nanoparticles and added a biocompatible coating so they could stay suspended in solution. After injection into mice, the particles traveled through the animals’ blood vessels, effectively turning the circulatory system into a delivery network.
Hong put it simply: “Wherever there is live soft tissue, there’s going to be vasculature providing nutrients, oxygen, and blood cells. We can also use that to deliver light.”
The particles stayed mostly dark until focused ultrasound hit them. Then they lit up at the chosen spot. According to the researchers, the system could do more than activate one fixed point. It could create light in multiple places at once, and it could also scan through tissue, generating light as the ultrasound focal point moved.
That scanning feature is one of the study’s more unusual elements. Rather than placing a permanent device in one location, the method treats light delivery as something mobile, something that can be redirected in three dimensions as needed. In principle, that could make the technique more flexible than implanted tools that stay locked in place.
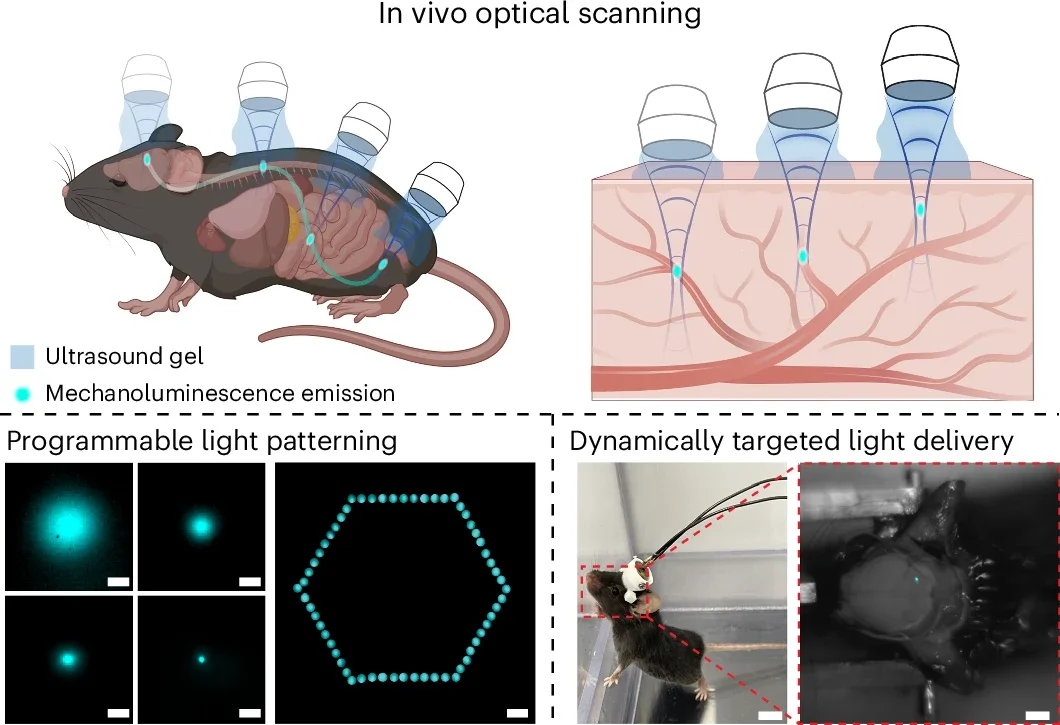
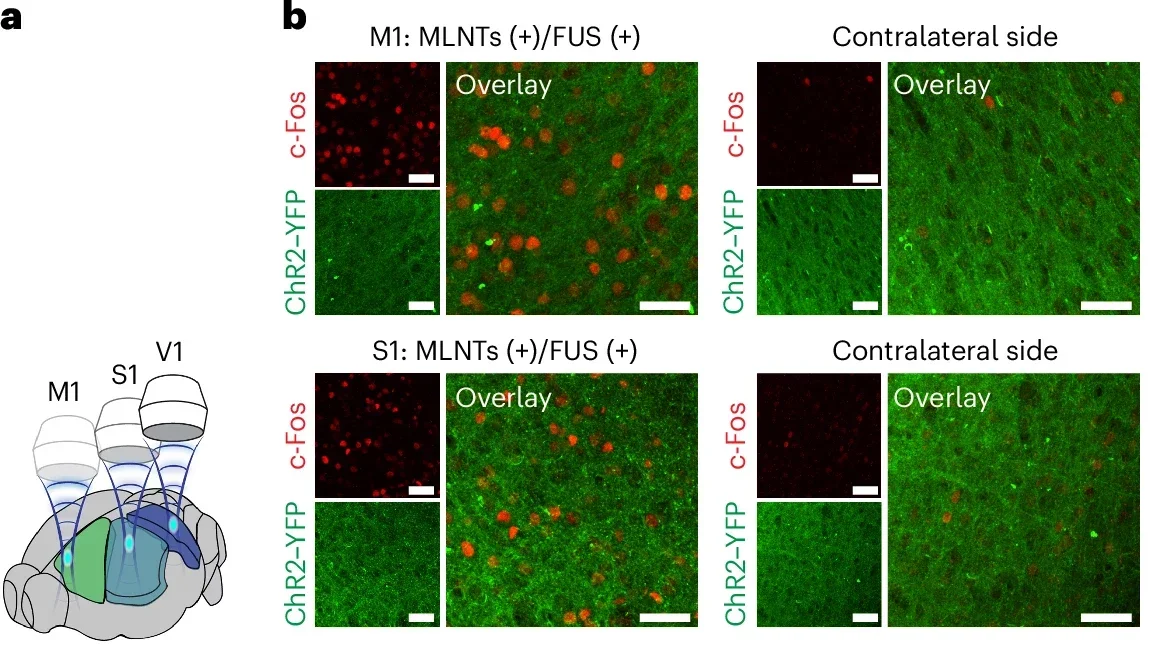
To test whether the method really worked deep inside the body, the team moved to the brain. Since light generated there cannot always be seen from the outside, the researchers built a small ultrasound-producing hat for mice and used it to create light in different parts of the brain.
The light then activated neurons in those regions. Depending on which area was stimulated, the mice turned left or right. The study also reported validation in opsin-expressing neurons using electrophysiological recordings and immunostaining in both the brain and the spinal cord. In freely moving animals, the researchers said they achieved dynamic three-dimensional brain targeting and temporally resolved behavioral control.
“We can noninvasively tune this emission in different brain regions to produce a variety of behavioral outcomes,” Hong said.
That sentence points to the larger promise here. The work is not only about making tissue glow. It is about using sound to switch biological activity on and off in places that normally require invasive access. The researchers describe it as a general method for applications that need light in deep tissue.
The particles used in this study emit blue light at a wavelength of 490 nanometers. That wavelength is useful for exciting neurons, which the team demonstrated, and it also has relevance for photodynamic therapy in cancer treatment. The same basic strategy, the researchers said, could be adapted with other nanomaterials to produce different wavelengths.

Hong and his colleagues are already experimenting with a material that emits ultraviolet light, which could potentially be used to kill bacteria and viruses.
The work also points toward gene editing. Hong is collaborating with Michael Lin, a professor of neurobiology and bioengineering at Stanford, to explore whether this light-producing system could be paired with a light-activated gene-editing platform.
One of the persistent worries in gene editing is that edits can happen in the wrong place. The idea here is to use ultrasound-guided light as a kind of local switch, turning editing on only in specific parts of the body rather than everywhere the editing machinery travels.
That remains a future goal, not something the researchers say they have already achieved. And the study comes with a clear limitation that the team itself acknowledged: safety.
While the particles did not appear to cause adverse effects in mice, they do not break down quickly. That raises the possibility that they could accumulate in organs such as the liver. For now, the work remains a proof of concept, not a ready-made medical treatment.
“What we’re demonstrating here is a proof of concept showing that you can produce light emission in a programmable manner deep within the body,” Hong said. “If we can replace the material with one that is safer to be used in humans, that will start to pave the way for clinical applications.”
That caveat is important. The advance is not that doctors can suddenly use this in patients tomorrow. It is that a longstanding technical barrier, how to place controllable light deep inside the body without implants, may now have a workable path around it.
If the safety challenge can be solved, this method could widen the reach of light-based medicine.
It could help researchers study brain and spinal cord circuits without surgery, support more precise cancer treatments, and offer a way to activate gene-editing tools only in selected tissues.
The central appeal is flexibility: instead of putting a light source into the body, the body’s own blood vessels carry the ingredients, and ultrasound decides where the light appears.
Research findings are available online in the journal Nature Materials.
The original story “Scientists create light inside the body using ultrasound” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Scientists create light inside the body using ultrasound appeared first on The Brighter Side of News.
Getting light deep inside the body has always come with a catch. Tissue scatters and absorbs it, which means doctors and researchers often have to cut into the body or thread in optical fibers to reach the places they want to target.
A team at Stanford University now says it has found a way around that problem, at least in mice. Instead of trying to shine light through layers of tissue, the researchers used ultrasound to trigger tiny particles circulating in the bloodstream, making them emit light exactly where the sound waves were focused. The result is a noninvasive method for creating small, controlled pockets of light inside living tissue.
That matters because light has become an increasingly useful tool in biology and medicine. It can stimulate cell activity, influence neural signals, and help treat certain cancers. The problem has never been what light can do. It has been how to get it where it needs to go without physically entering the body.
“Ultrasound is very convenient to use, and it penetrates much deeper into the body than light,” said Guosong Hong, an assistant professor of materials science and engineering at Stanford and the senior author on the paper. “With these materials, we can produce light emission in the brain, in the gut, in the spinal cord, in the muscle, virtually anywhere, without needing a physical implant.”

The approach starts with particles that are not the sort of material most people would associate with medicine. The researchers began with large ceramic particles that emit light when they are under mechanical stress. Ultrasound waves can create that stress.
To make them usable in the body, the team processed those particles into nanoparticles and added a biocompatible coating so they could stay suspended in solution. After injection into mice, the particles traveled through the animals’ blood vessels, effectively turning the circulatory system into a delivery network.
Hong put it simply: “Wherever there is live soft tissue, there’s going to be vasculature providing nutrients, oxygen, and blood cells. We can also use that to deliver light.”
The particles stayed mostly dark until focused ultrasound hit them. Then they lit up at the chosen spot. According to the researchers, the system could do more than activate one fixed point. It could create light in multiple places at once, and it could also scan through tissue, generating light as the ultrasound focal point moved.
That scanning feature is one of the study’s more unusual elements. Rather than placing a permanent device in one location, the method treats light delivery as something mobile, something that can be redirected in three dimensions as needed. In principle, that could make the technique more flexible than implanted tools that stay locked in place.

To test whether the method really worked deep inside the body, the team moved to the brain. Since light generated there cannot always be seen from the outside, the researchers built a small ultrasound-producing hat for mice and used it to create light in different parts of the brain.
The light then activated neurons in those regions. Depending on which area was stimulated, the mice turned left or right. The study also reported validation in opsin-expressing neurons using electrophysiological recordings and immunostaining in both the brain and the spinal cord. In freely moving animals, the researchers said they achieved dynamic three-dimensional brain targeting and temporally resolved behavioral control.
“We can noninvasively tune this emission in different brain regions to produce a variety of behavioral outcomes,” Hong said.
That sentence points to the larger promise here. The work is not only about making tissue glow. It is about using sound to switch biological activity on and off in places that normally require invasive access. The researchers describe it as a general method for applications that need light in deep tissue.
The particles used in this study emit blue light at a wavelength of 490 nanometers. That wavelength is useful for exciting neurons, which the team demonstrated, and it also has relevance for photodynamic therapy in cancer treatment. The same basic strategy, the researchers said, could be adapted with other nanomaterials to produce different wavelengths.

Hong and his colleagues are already experimenting with a material that emits ultraviolet light, which could potentially be used to kill bacteria and viruses.
The work also points toward gene editing. Hong is collaborating with Michael Lin, a professor of neurobiology and bioengineering at Stanford, to explore whether this light-producing system could be paired with a light-activated gene-editing platform.
One of the persistent worries in gene editing is that edits can happen in the wrong place. The idea here is to use ultrasound-guided light as a kind of local switch, turning editing on only in specific parts of the body rather than everywhere the editing machinery travels.
That remains a future goal, not something the researchers say they have already achieved. And the study comes with a clear limitation that the team itself acknowledged: safety.
While the particles did not appear to cause adverse effects in mice, they do not break down quickly. That raises the possibility that they could accumulate in organs such as the liver. For now, the work remains a proof of concept, not a ready-made medical treatment.
“What we’re demonstrating here is a proof of concept showing that you can produce light emission in a programmable manner deep within the body,” Hong said. “If we can replace the material with one that is safer to be used in humans, that will start to pave the way for clinical applications.”

That caveat is important. The advance is not that doctors can suddenly use this in patients tomorrow. It is that a longstanding technical barrier, how to place controllable light deep inside the body without implants, may now have a workable path around it.
If the safety challenge can be solved, this method could widen the reach of light-based medicine.
It could help researchers study brain and spinal cord circuits without surgery, support more precise cancer treatments, and offer a way to activate gene-editing tools only in selected tissues.
The central appeal is flexibility: instead of putting a light source into the body, the body’s own blood vessels carry the ingredients, and ultrasound decides where the light appears.
Research findings are available online in the journal Nature Materials.
The original story “Scientists create light inside the body using ultrasound” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Scientists create light inside the body using ultrasound appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.