A small cluster of damaged immune cells can do outsized harm.
That is the picture emerging from new research at UCLA, where scientists identified a rogue population of macrophages, the tissue-patrolling immune cells that normally clean up debris and help maintain order. In aging livers, and in livers damaged by fatty liver disease, those cells can slip into a senescent state. They stop dividing, refuse to die and begin sending out inflammatory signals that disturb the tissue around them.
“Senescent cells are fairly rare, but think of them like a broken-down car on the 405,” said Anthony Covarrubias, senior author of the study and a member of the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA. “Just one stalled car can back up traffic for miles. Now imagine five or ten of them slowly accumulating. That’s what these cells do to a tissue: even a small number causes enormous disruption.”
For years, scientists argued over whether macrophages could truly become senescent at all. Part of the problem was that healthy macrophages already carry some of the same molecular features linked to senescence, making it hard to tell a damaged cell from one simply doing normal immune work.

The UCLA team says it found a cleaner way to spot the difference.
The researchers identified a two-part signature involving the proteins p21 and TREM2. Together, they marked macrophages that were no longer functioning normally and were instead feeding inflammation in surrounding tissue.
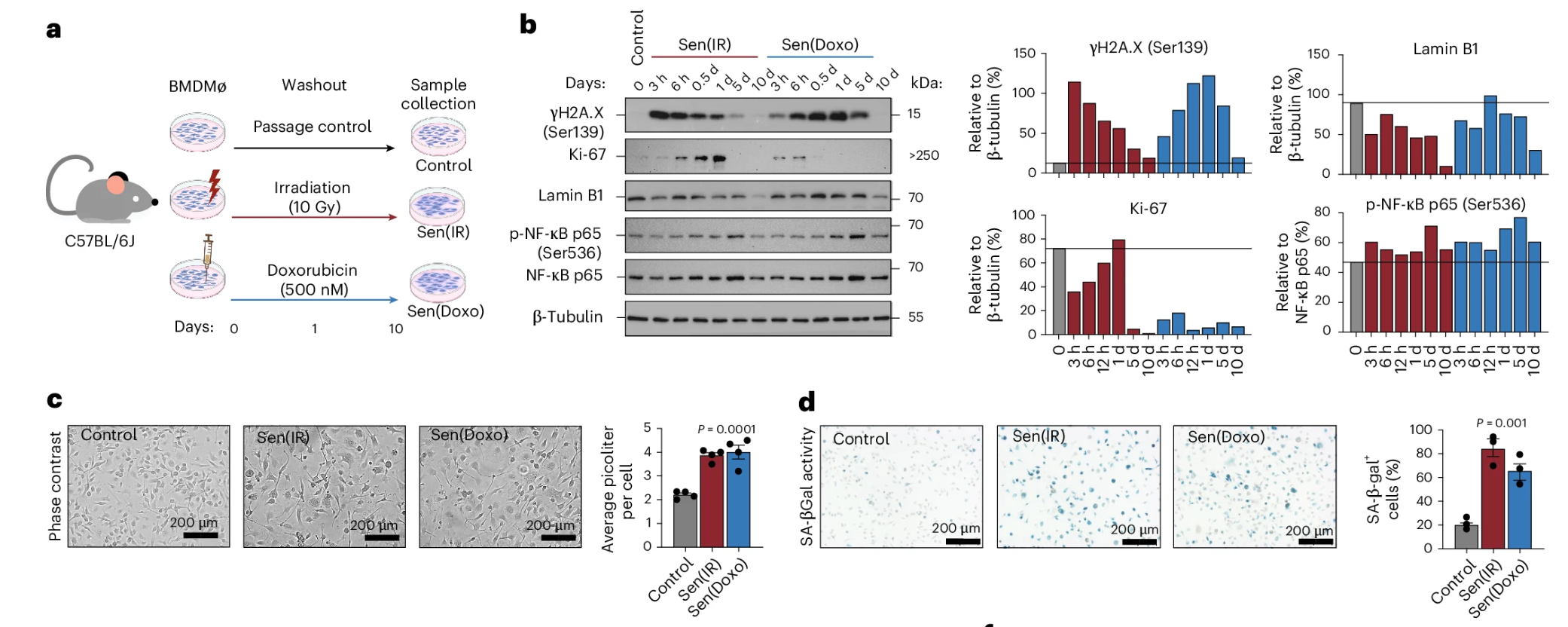
That combination stood out in the liver. In young mice, about 5% of liver macrophages fit the senescent pattern. In old mice, the proportion climbed to roughly 60% to 80%, closely tracking the rise in chronic liver inflammation that comes with age.
The team also found that aging is not the only force pushing macrophages in this direction. Excess cholesterol appears to do it too.
When healthy macrophages were exposed in the lab to high levels of LDL cholesterol, they began acting like senescent cells. They stopped dividing. They produced inflammatory proteins, and they developed the same p21-TREM2 signature seen in aging liver tissue.
“Physiologically, macrophages can handle cholesterol metabolism,” said Ivan Salladay-Perez, first author of the study and a graduate student in the Covarrubias lab. “But in a chronic state, it’s pathological. And when you look at fatty liver disease, which is driven by overnutrition and too much cholesterol in the blood, that excess cholesterol appears to be a major driver of the senescent macrophage population.”
The work helps explain a long-standing problem in liver disease. Fatty liver does not just involve fat piling up inside the organ. It also involves a slow-burning inflammatory environment, and these altered immune cells may be one of the key reasons that damage persists.
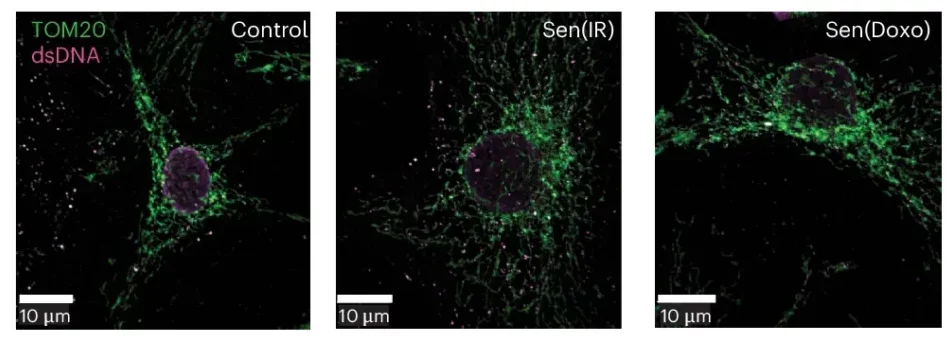
The researchers describe senescence as a stress response. A cell under enough strain stops dividing, but instead of shutting down quietly, it lingers and starts releasing inflammatory molecules, sometimes called a toxic secretory mix.
In these macrophages, the stress seemed tied not only to cholesterol overload but also to deeper signs of dysfunction. The cells developed lipid droplets, shifted their metabolism and activated inflammatory pathways linked to type I interferon signaling, a response more often associated with viral defense. They also became worse at clearing dying cells, a task healthy macrophages usually perform.
That matters because the liver depends on resident macrophages, called Kupffer cells, to keep the tissue stable. In the study, these cells appeared to be a major senescent population in aging liver, more so than hepatocytes or endothelial cells.
The researchers also looked beyond mice. In human macrophages grown from blood cells, DNA damage drove a similar senescent state, again marked by increased p21 and TREM2. Then, using publicly available genomic data from human liver biopsies, the team found the same senescent macrophage signature elevated in diseased livers.
That does not prove the process causes human disease on its own. But it does suggest the pattern is not confined to mouse experiments.
To see whether these macrophages were merely a sign of damage or an active driver of it, the team used ABT-263, a drug that can trigger death in senescent cells.
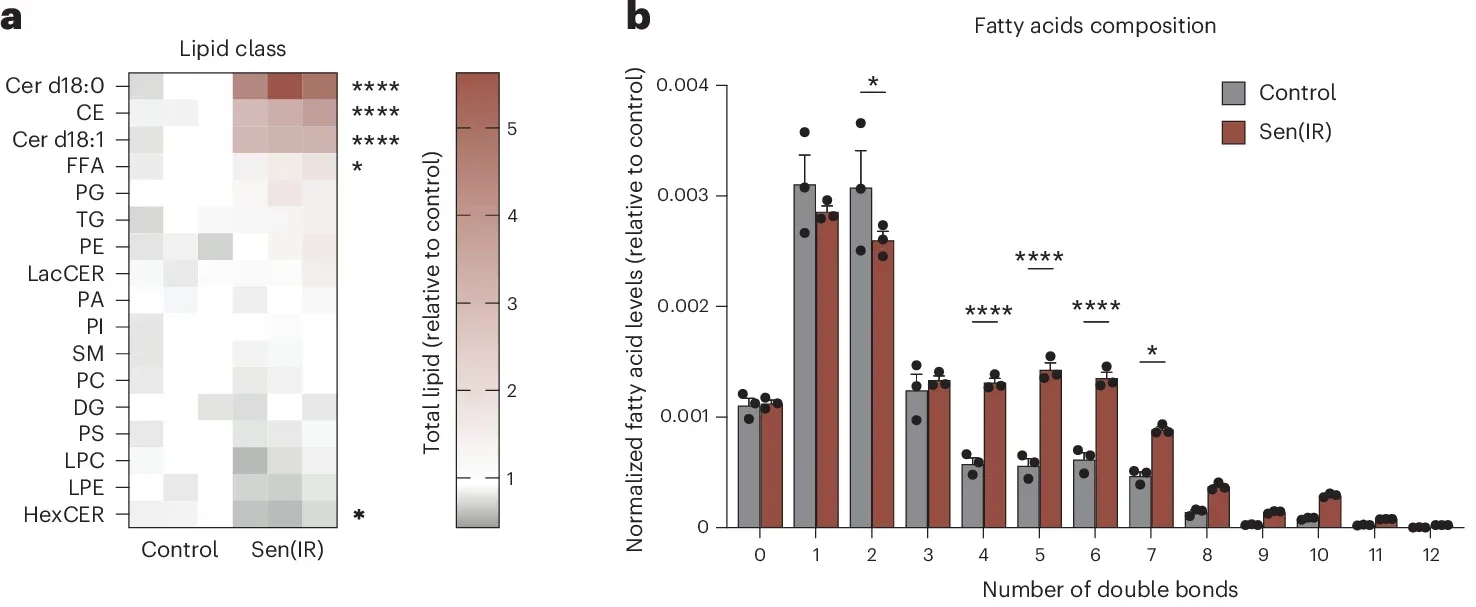
In mice with diet-driven fatty liver disease, the effect was striking. Liver weight dropped from about 7% of body weight back toward a healthier 4% to 5%. Overall body weight fell by about 25%, from an average of 40 grams toward 30 grams, even though the animals remained on a high-fat, high-cholesterol diet. Their livers looked different too, smaller and darker red rather than enlarged and yellowish.
“That’s what wowed me,” Salladay-Perez said. “Eliminating senescent cells doesn’t just slow the fatty liver, it actually reverses it.”
The treatment also reduced inflammatory signals and lowered the burden of senescent macrophages in the liver. In older mice, the share of liver macrophages carrying the senescent pattern dropped sharply after treatment. In a separate mouse model designed to mimic metabolic dysfunction-associated steatotic liver disease, the drug reduced body weight, spleen enlargement, liver fat and inflammatory markers, while raising liver NAD+ levels by about 30%.
Even so, the improvement had limits. The treatment reduced steatosis, or fat buildup, but did not significantly change fibrosis grade in that model. So the drug appeared better at easing some aspects of liver damage than reversing all of them.
The findings fit with a wider idea in geroscience, the notion that one core aging mechanism may help drive several major diseases at once. If senescent macrophages are helping fuel chronic inflammation in the liver, similar cells may also be contributing to disorders in other organs.
The researchers are already exploring that possibility in cancer and neurodegeneration. In the brain, for instance, microglia, the resident macrophages of the central nervous system, may face similar stress in diseases marked by accumulating debris, such as Alzheimer’s.
“If you really understand the basic mechanisms driving inflammation with aging, you can target those same mechanisms to treat not just fatty liver disease, but atherosclerosis, Alzheimer’s and cancer,” Salladay-Perez said. “It all goes back to understanding how these cells arise in the first place.”
Still, the study comes with clear limitations. Much of the animal work relied on male mice. The researchers say sex chromosomes may influence senescent macrophage behavior, but that remains under study. The in vivo work also centered mainly on liver macrophages, not the full range of tissue-resident macrophages found in organs such as the brain, skin, heart and fat. And while ABT-263 worked in mice, the drug is too toxic for broad use in people.
So the immediate takeaway is not that a treatment is ready.
It is that a specific kind of damaged immune cell, once hard to define and easy to dismiss, may be doing far more to age tissues than scientists realized.
The work points to a possible new treatment strategy for fatty liver disease, one that targets harmful senescent macrophages rather than focusing only on diet, weight loss or general inflammation.
It also gives researchers a more precise way to identify these cells using the p21-TREM2 signature. That could improve both drug development and early disease tracking.
For now, the most practical next step is finding safer compounds that can remove these cells without the side effects seen with ABT-263.
Research findings are available online in the journal Nature Aging.
The original story “UCLA researchers discover key driver of fatty liver disease, inflammation and aging” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post UCLA researchers discover key driver of fatty liver disease, inflammation and aging appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.