Sunlight hits tiny particles of plastic floating in a clear water solution. Slowly, they begin to disappear, leaving behind a familiar household chemical: acetic acid, the main ingredient in vinegar. At the University of Waterloo, engineers have developed a process that transforms plastic waste into something useful using sunlight and a specially designed catalyst.
“This method allows abundant and free solar energy to break down plastic pollution without adding extra carbon dioxide to the atmosphere,” said Dr. Yimin Wu, a professor of mechanical and mechatronics engineering and the Tang Family Chair in New Energy Materials and Sustainability.
The research team, led by Wu and PhD student Wei Wei, demonstrates a way to tackle plastic pollution while simultaneously producing valuable chemicals. Their findings, supported in part by the Waterloo Institute for Nanotechnology and the Water Institute, offer a glimpse into how solar-driven chemical reactions could upcycle plastics in the future.
Plastics have a notorious reputation for durability. Everyday materials like polyethylene terephthalate (PET), polypropylene (PP), and polyethylene (PE) can linger for decades in oceans, rivers, and landfills. These polymers fragment into microplastics, which accumulate in ecosystems and even enter human food chains. Traditional recycling methods—often melting plastics back into raw forms—struggle with efficiency, especially when waste streams are mixed or contaminated.
“Microplastic waste is everywhere, and it poses serious threats to both terrestrial and aquatic life,” Wu explained. The Waterloo team wanted to address this problem while producing something economically valuable.
The breakthrough relies on a bio-inspired approach. Certain fungi naturally digest tough plant material using enzymes. Inspired by these organisms, the researchers designed a cascade photocatalysis system—a step-by-step chemical reaction process activated by sunlight.
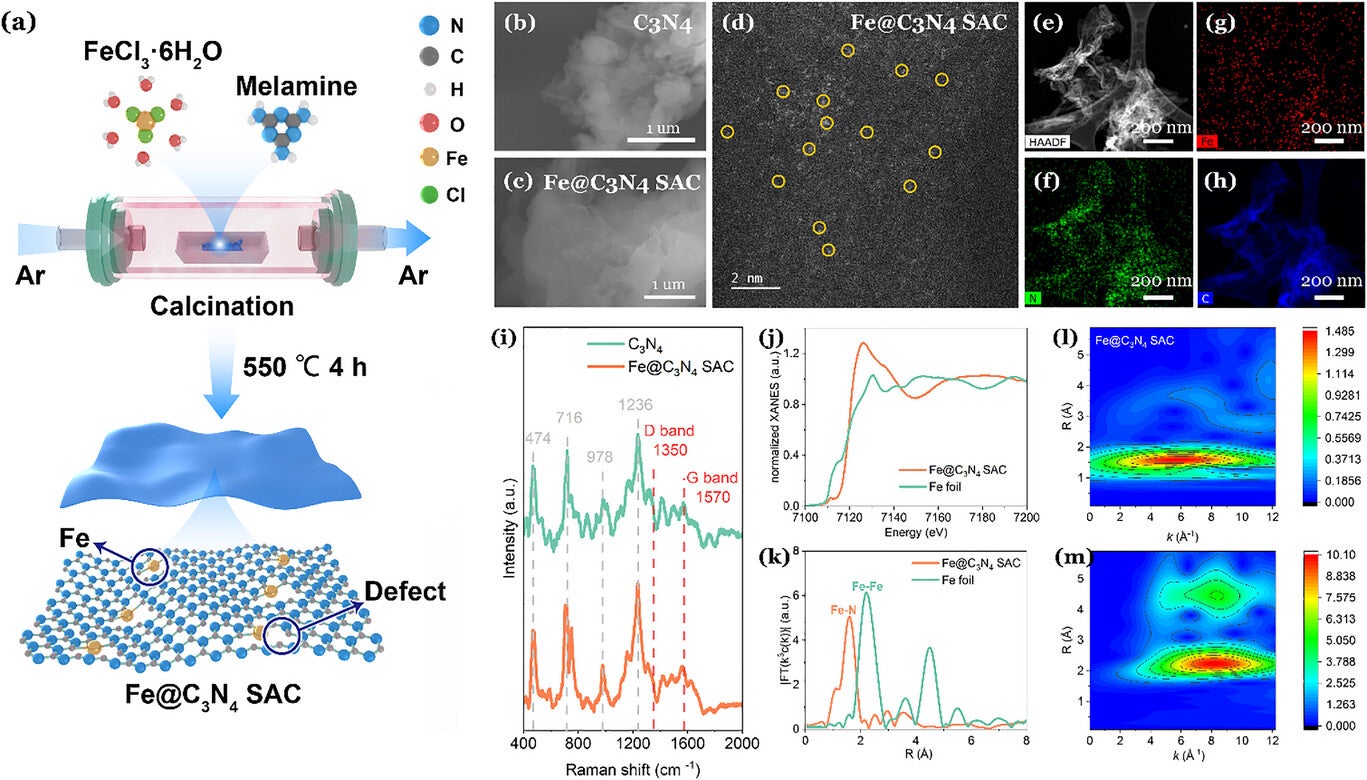
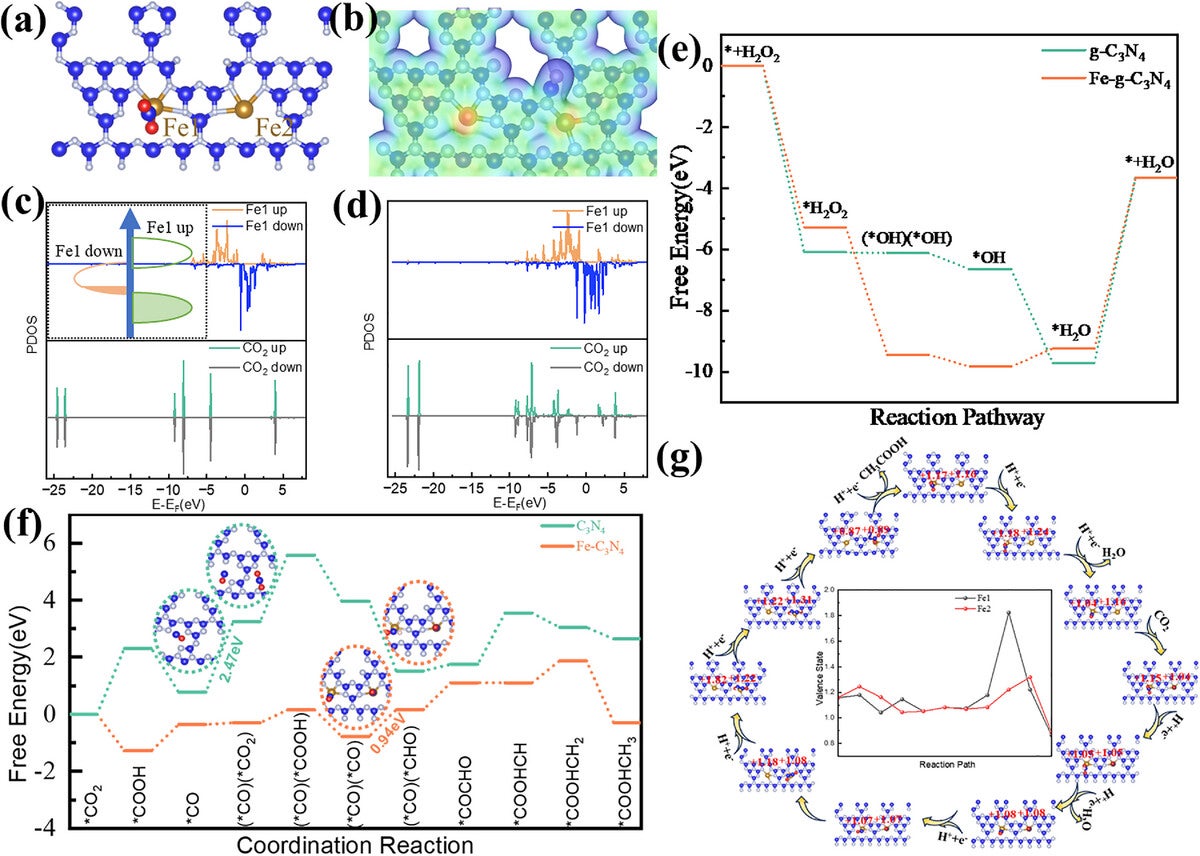
At the core of the system is a single-atom catalyst made of iron atoms embedded in carbon nitride, referred to as Fe@C₃N₄ SAC. The material simultaneously generates reactive species and promotes carbon-based transformations, functioning as a bi-functional system. “Our goal was to solve the plastic pollution challenge by converting microplastic waste into high-value products using sunlight,” Wu said.
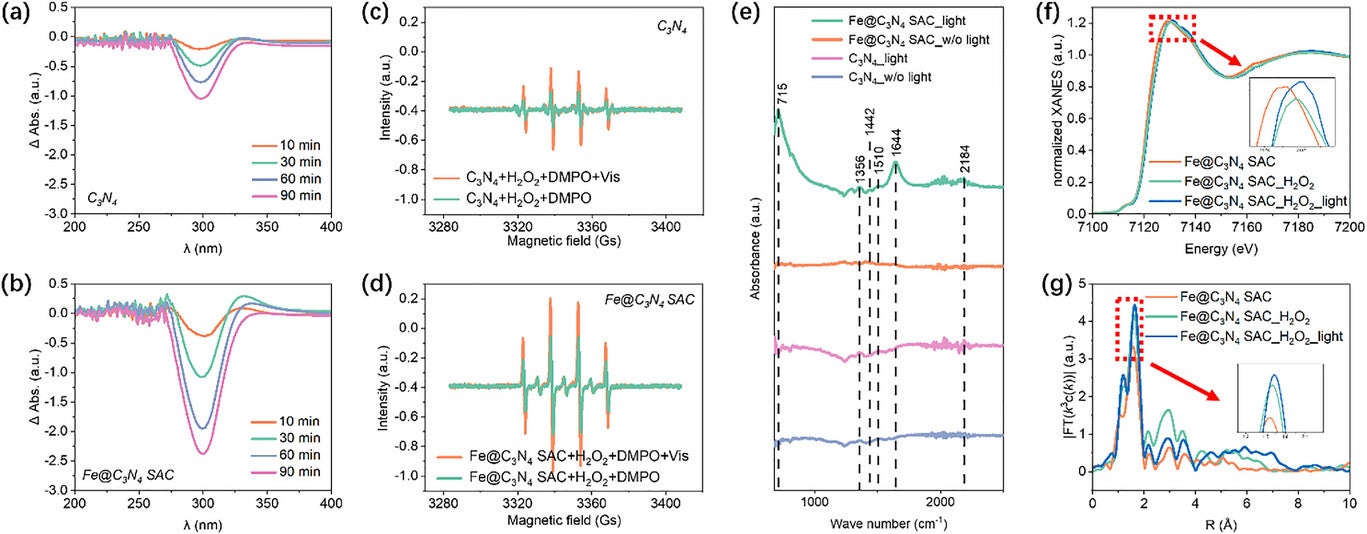
When sunlight hits the catalyst, electrons are excited and reactive oxygen species form. These reactive molecules attack the strong bonds in plastic polymers, breaking them into smaller fragments. A series of oxidation and reduction reactions then transforms the fragments into acetic acid with high selectivity.
Unlike conventional chemical recycling, this approach doesn’t rely on high temperatures, harsh solvents, or added energy from fossil fuels. The reactions occur in water under ambient conditions, making the system particularly relevant for aquatic environments where plastic pollution accumulates.
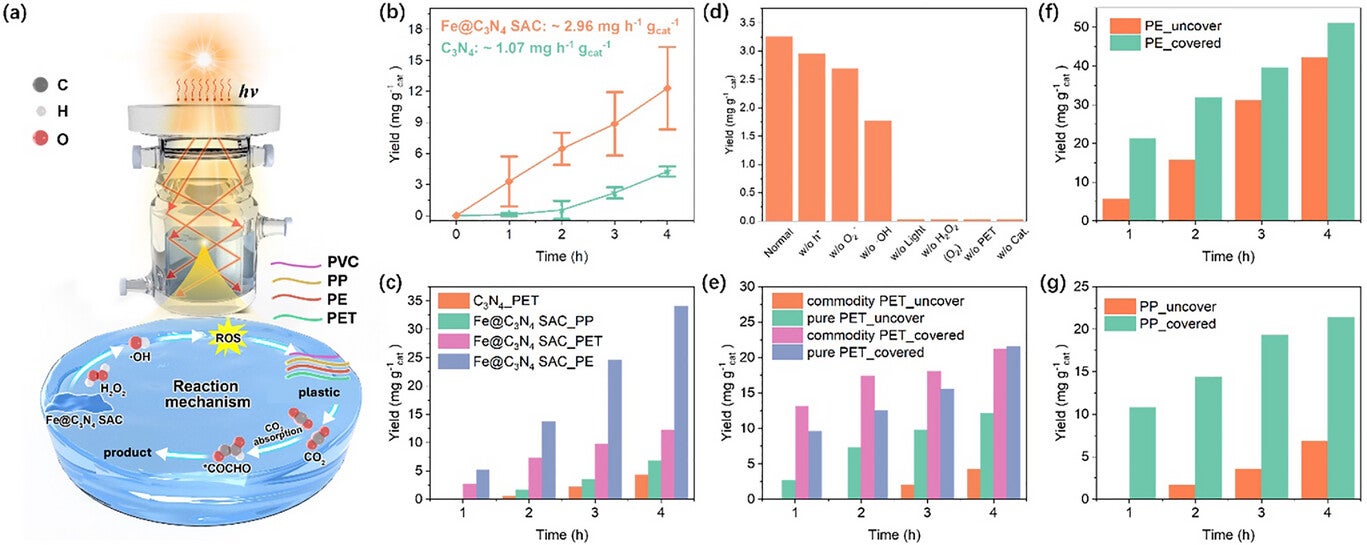
The researchers tested their method on a range of common plastics—PVC, PP, PE, and PET—and found that it worked even with mixed compositions. This versatility could simplify recycling in real-world conditions, where pre-sorting plastics is often costly and labor-intensive.
“The financial and economic benefits associated with this innovation seem promising,” said Roy Brouwer, executive director of the Water Institute and coauthor of the study, referring to both societal and industrial advantages.
What makes this approach stand out is its output. Instead of generating low-value material, the cascade photocatalysis produces acetic acid, widely used in vinegar, adhesives, solvents, and industrial chemicals. By turning a pollutant into a commodity chemical, the process creates both environmental and economic incentives.
The single-atom iron catalyst plays a critical role. Its dispersed atomic structure maximizes active sites for reactions, maintains stability under repeated use, and controls how the chemical transformation proceeds. In lab tests, the catalyst efficiently converted plastic waste into acetic acid over multiple cycles without losing activity—a key factor for potential large-scale applications.
The cascade system involves two main reaction types in sequence. First, the plastic polymers break into smaller organic fragments. Then, these fragments undergo oxidation and reduction reactions, eventually producing acetic acid as the dominant product. Analytical techniques confirmed that the sequence of reactions—one feeding into the next—was responsible for high selectivity.
In experiments, plastic particles gradually disappeared from water as sunlight-driven reactions proceeded. Acetic acid accumulated steadily, illustrating the system’s ability to upcycle waste into a useful chemical. Because the reaction occurs at normal temperature and pressure, it avoids the energy-intensive processes common in conventional recycling.
The system also tolerated mixed plastics, further demonstrating real-world practicality. Many existing methods require separation of plastic types before processing, increasing complexity and cost. By contrast, the Waterloo approach could handle a mixture of PET, PP, and PE without pre-sorting, simplifying operations and potentially improving adoption rates.
The significance of this study extends beyond a single lab-scale achievement. It offers a pathway for chemical recycling that uses renewable energy, reduces carbon emissions, and produces value-added products. Microplastics could be degraded before accumulating in water systems, reducing environmental and public health risks.
Additionally, the use of iron—an abundant and inexpensive metal—as a single-atom catalyst makes the system more scalable and sustainable than methods relying on rare or precious metals. Producing acetic acid directly from waste provides an economic incentive, potentially supporting circular business models where plastics are transformed rather than discarded.
The research remains at the laboratory stage. Scaling up will require reactors that efficiently capture sunlight, handling large volumes of mixed waste, and maintaining long-term catalyst stability under real-world conditions. Researchers also need to optimize material design and manufacturing processes to make industrial deployment viable.
Still, the concept demonstrates the potential for sunlight-driven chemical recycling to integrate into larger environmental cleanup strategies and industrial chemical production. Wu and colleagues envision a future where solar-powered plastic upcycling contributes to a circular economy, reducing environmental harm while generating commercially useful chemicals.
This innovation offers more than a lab curiosity.
By converting plastic waste into acetic acid using sunlight, the system could support cleaner water, reduced landfill accumulation, and lower reliance on fossil-fuel-based chemical production.
Industrial-scale adoption could transform waste management, creating new economic opportunities while tackling a pressing global environmental problem.
Research findings are available online in the journal Wiley Advanced.
The original story “New chemical process uses sunlight to turn plastic pollution into vinegar” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post New chemical process uses sunlight to turn plastic pollution into vinegar appeared first on The Brighter Side of News.