The worst damage from COVID-19 was first measured in fevers, oxygen levels and death counts. Now, years later, researchers are still tracking what the virus may leave behind in the lungs, and one new analysis points to a troubling possibility: conditions that could raise the risk of lung cancer over time.
A team from Marshall University’s Joan C. Edwards School of Medicine and The Hebrew University of Jerusalem reports that people with a history of COVID-19 showed a higher rate of later lung cancer in a large clinical dataset, with the strongest signal appearing in current and former smokers. In lab and animal experiments, the same group also traced a possible biological route for that risk, centering on a protein called thymidine phosphorylase, or TYMP, that appears to help drive inflammation, fibrosis and tumor-friendly changes in lung tissue.
The work was published in Frontiers in Immunology.
“Our findings suggest that COVID-19 may do more than cause acute illness, it may also create biological conditions in the lung that could contribute to increased cancer risk over time,” said Wei Li, a professor of biomedical sciences at Marshall and a co-corresponding author of the research. “Understanding these pathways is critical as we continue to study the long-term health impacts of the virus.”
COVID-19 has infected more than 778 million people worldwide and caused 7.1 million deaths as of September 2025, according to the researchers. Even after recovery, many survivors face lasting complications. Among them is interstitial lung fibrosis, seen in about 25% of patients three months after infection and 14% after one year. That matters because pulmonary fibrosis is already known to raise lung cancer risk.
To look for patterns in people, the investigators used de-identified electronic health records from the TriNetX Research Network. They compared matched groups by age, sex, race and ethnicity, and separated patients by smoking status.
The pattern stood out most clearly in the lung.
Among current smokers, lung cancer developed in 1.7% of people with prior COVID-19, compared with 1.4% of those without it. In former smokers, the numbers were 1.5% versus 1.2%. In never smokers, the absolute risk was far lower, 0.21% versus 0.18%, but the difference still reached statistical significance. By contrast, the study did not find the same consistent pattern for oral or bladder cancer.
That organ-specific pattern mattered to the authors. If the signal had appeared broadly across many cancers, it might have suggested a more general bias in the data. Instead, the increase was concentrated in the organ most directly affected by the virus.
The study also looked at vaccinated and unvaccinated people who had COVID-19. In former smokers with COVID-19, unvaccinated patients had a higher lung cancer risk than vaccinated ones. In current smokers, the time-to-event analysis did not show a significant difference.
The human data alone could not answer why this might be happening. So the team turned to experiments in mice and cells, focusing on the SARS-CoV-2 spike protein, the part of the virus that first interacts with host cells.
Earlier work had already suggested that the spike protein can damage tissues even without full viral replication. In this study, researchers delivered spike-containing material directly into the lungs of mice engineered to express human ACE2, the receptor the virus uses to enter cells.
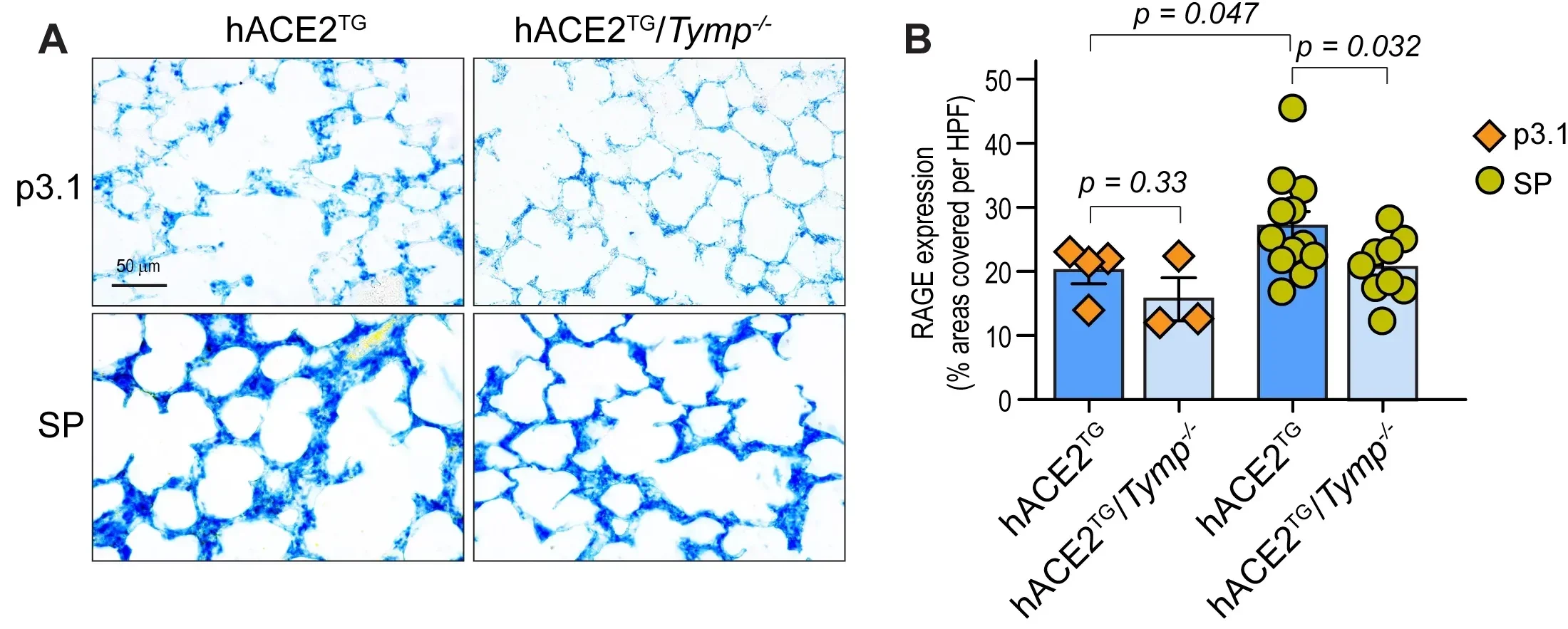
Within 24 hours, the mice showed signs of acute lung injury. Lung tissue had stronger staining for RAGE, a marker linked to lung inflammation. Neutrophils, a type of immune cell that floods damaged tissue, also increased. So did microthrombi, tiny blood clots that have become a familiar feature of severe COVID-19.
But the effect weakened sharply in mice that lacked TYMP.
Those TYMP-deficient mice had less lung injury, fewer infiltrating neutrophils and less extensive clotting after exposure to the spike protein. Macrophage levels, however, did not differ much between groups, suggesting TYMP may shape some parts of the inflammatory response more than others.
That protein has drawn attention before. TYMP is elevated in several diseases, including cancer, diabetes and atherosclerosis, and higher levels have been linked to worse cancer outcomes. It is also increased in the blood, immune cells and lungs of patients with COVID-19, where it tracks with inflammation, thrombosis and respiratory symptoms.
Then came the long-term question.
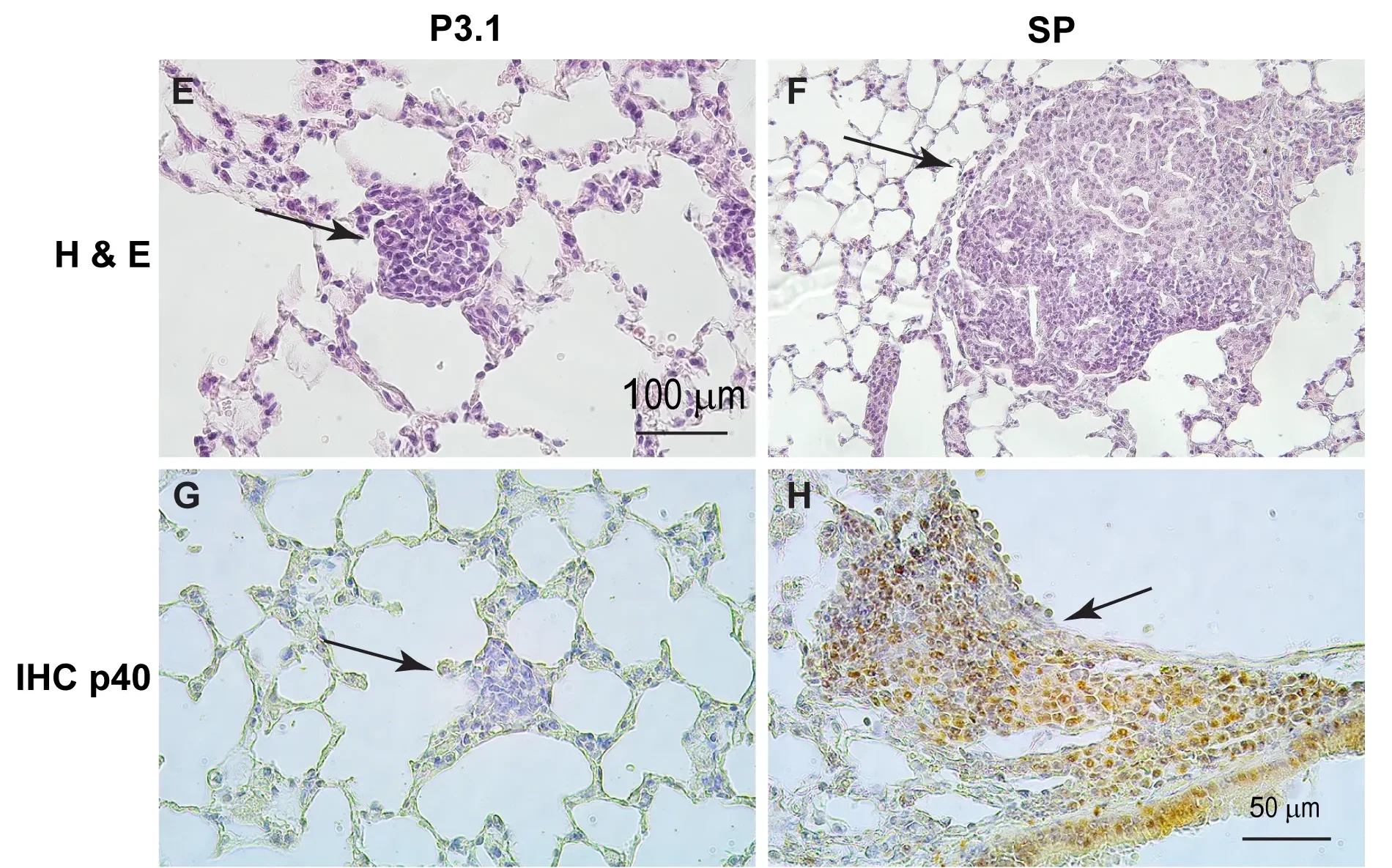
To test whether early lung injury might feed later tumor growth, the researchers used a urethane-induced mouse model of lung cancer after exposing the animals to spike protein. In mice that received the spike treatment, tumors were found in 10 of 20 lung lobes examined. In control mice, only one tumor appeared among 10 lobes.
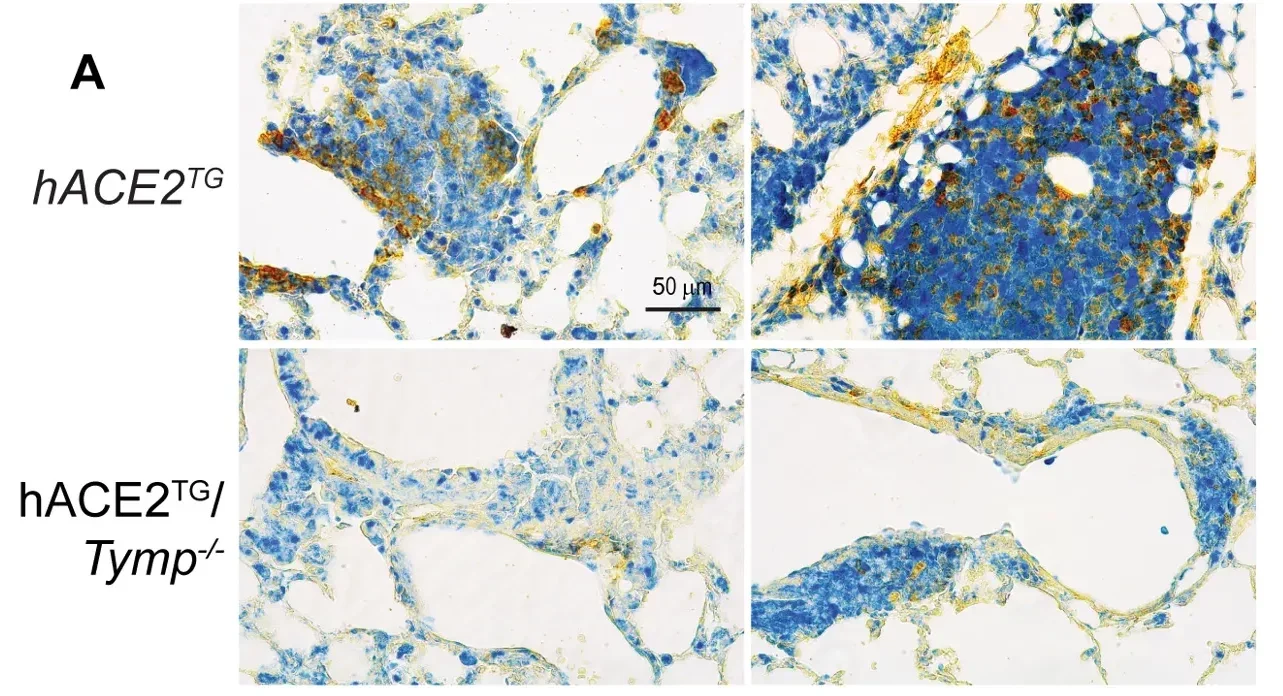
When the scientists repeated the experiment in mice lacking TYMP, tumor burden dropped sharply. Tumors were found in 18% of examined lobes, compared with 50% in the mice with intact TYMP.
The tumors also looked unusual. All tumors found in spike-treated mice stained positive for p40, a marker of lung squamous cell carcinoma. That is not the usual pattern in this urethane model, which more often produces bronchioloalveolar adenoma-like tumors. The finding suggests that spike-related injury may not just increase tumor number, but may also alter the kind of tumor that emerges.
Another clue came from fibrosis. In the mice with TYMP, spike exposure significantly increased collagen-rich fibrotic remodeling in the lung. That fibrotic response was much lower in TYMP-deficient animals.
The research also pointed to STAT3, a signaling pathway tied to inflammation, fibrosis and cancer progression. The team found that spike-related signaling and TYMP appeared to amplify STAT3 activity. In tumor sections, total STAT3 was concentrated mainly within tumors, while activated phospho-STAT3 was spread more widely through the tissue.
Cytokine profiling added one more layer. Mice with TYMP showed a pattern associated with a myeloid-heavy, tumor-promoting inflammatory state, including higher levels of G-CSF, CCL2, CXCL1, TIMP-1 and CXCL13. TYMP-deficient mice, in contrast, showed more of a T-cell-inflamed, anti-tumor profile, with higher IFN-γ, IL-7, CXCL9, CXCL11, IL-16 and CCL1.
Taken together, the findings describe a lung environment that may become more hospitable to cancer after viral injury: inflamed, fibrotic, clot-prone and immunologically altered.
“The collaboration between basic and translational scientists and clinicians enabled the identification of mechanisms underlying epidemiological evidence linking COVID-19 infection to the potential increased risk of lung cancer,” said David Gozal, vice president for health affairs and dean of the Joan C. Edwards School of Medicine at Marshall University.
The findings are provocative, but they do not prove that COVID-19 directly causes lung cancer.
The clinical part of the research was retrospective, meaning it looked backward through existing medical records. That kind of design can show associations, not firm cause and effect. The dataset also lacked patient-level details on vaccine doses, so the authors could not study that variable closely. And in the animal work, the team used intratracheal delivery of spike protein rather than live SARS-CoV-2 infection, which isolates the effect of spike but does not capture the full biology of viral infection.
The authors say prospective studies will be needed to define long-term cancer risk after infection and to better understand how TYMP works downstream in the damaged lung.
Still, the work offers a concrete lead in a murky area. Instead of treating long COVID and cancer risk as separate questions, it connects them through a specific molecular pathway in injured lung tissue.
The study suggests that COVID-19 survivors, especially current and former smokers, may deserve closer attention when doctors think about long-term lung health. It also points to TYMP as a possible target for future therapies aimed at limiting fibrosis, inflammation or tumor-friendly changes after infection.
The researchers note that tipiracil, an FDA-approved TYMP inhibitor already known for anti-thrombotic effects, could be worth studying in this context.
For now, the findings do not justify alarm, but they do strengthen the case for watching post-COVID lung damage as something that may carry consequences well beyond the original infection.
Research findings are available online in the journal Frontiers in Immunology.
The original story “Scientists find concerning link between COVID-19 and lung cancer” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Scientists find concerning link between COVID-19 and lung cancer appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.